Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
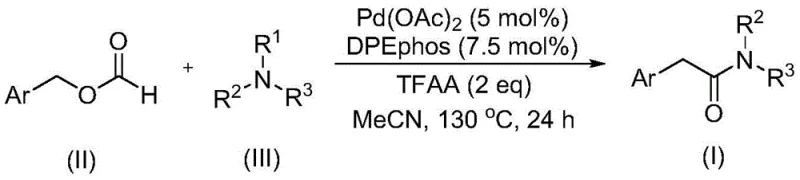
The landscape of organic synthesis for pharmaceutical and agrochemical intermediates is constantly evolving, driven by the need for safer, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN111978194B, which discloses a novel preparation method for aryl acetamide compounds. This technology represents a paradigm shift from traditional amidation strategies by utilizing a palladium-catalyzed carbonylation reaction that employs benzyl formate as both a carbon monoxide source and a reactant. For R&D directors and procurement specialists seeking reliable aryl acetamide suppliers, this innovation offers a robust pathway to high-purity intermediates. The process is characterized by its operational simplicity, mild reaction conditions, and the ability to utilize readily available starting materials, thereby addressing critical bottlenecks in the manufacturing of complex amide scaffolds found in numerous bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamide compounds has relied heavily on the direct amidation of phenylacetic acid and its derivatives. While conceptually straightforward, these classical methods often suffer from significant drawbacks, including harsh reaction conditions, poor atom economy, and the generation of substantial waste streams. Furthermore, transition metal-catalyzed carbonylation reactions have emerged as alternative strategies; however, they predominantly utilize primary and secondary amines as nitrogen sources. The activation of tertiary amines remains a formidable challenge in organic chemistry due to the strength and stability of the C-N bond. Conventional approaches to cleaving these bonds often necessitate the use of excessive oxidants or dangerous explosive mixtures of carbon monoxide and oxygen, posing severe safety hazards and complicating the purification process. These limitations restrict the designability of substrates and increase the overall cost of production, making them less attractive for large-scale commercial applications.
The Novel Approach
In stark contrast to these legacy techniques, the method described in patent CN111978194B introduces a highly efficient route that circumvents the need for hazardous gas handling and aggressive oxidants. By employing benzyl formate as a liquid CO surrogate, the reaction proceeds under much safer conditions while maintaining high efficiency. Crucially, this methodology enables the cleavage of the C-N bond in tertiary amines without the addition of external oxidants, a feature that significantly streamlines the reaction profile. The use of a palladium catalyst in conjunction with specific ligands allows for broad substrate compatibility, tolerating a wide range of functional groups such as halogens, ethers, and trifluoromethyl groups. This versatility ensures that the process can be adapted for the synthesis of diverse aryl acetamide derivatives required in modern drug discovery and development pipelines.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the palladium complex. The reaction utilizes palladium(II) acetate as the precatalyst and DPEphos (bis(2-diphenylphosphinophenyl) ether) as the ligand, forming a highly active species capable of facilitating the necessary bond transformations. The mechanism likely involves the oxidative addition of the benzyl formate to the palladium center, followed by decarbonylation or migratory insertion steps that generate the acyl-palladium intermediate. Simultaneously, the system activates the tertiary amine, promoting the cleavage of the C-N bond to release the amine fragment which then couples with the acyl species. This intricate dance of coordination and bond breaking occurs seamlessly at temperatures around 130°C in polar aprotic solvents like acetonitrile, ensuring high conversion rates without degrading sensitive functional groups on the aromatic ring.
From an impurity control perspective, the absence of external oxidants is a game-changer for R&D teams focused on purity profiles. Traditional oxidative carbonylation often leads to over-oxidation byproducts or complex mixtures that are difficult to separate. By relying on the intrinsic reactivity of the benzyl formate and the specific electronic properties of the Pd-DPEphos complex, the reaction minimizes side reactions. The resulting aryl acetamide compounds, such as those depicted in the patent examples with various aryl substitutions (phenyl, furyl, naphthyl, thienyl), are obtained with high selectivity. This mechanistic precision translates directly to simplified downstream processing, as the crude reaction mixture contains fewer impurities, reducing the burden on purification units and enhancing the overall yield of the target high-purity pharmaceutical intermediates.
How to Synthesize Aryl Acetamide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific parameters to maximize efficiency and reproducibility. The protocol involves charging a reactor with the palladium catalyst, ligand, trifluoroacetic anhydride, benzyl formate, and the chosen tertiary amine in an organic solvent. The mixture is then heated to facilitate the carbonylation and C-N bond cleavage. Detailed standardized operating procedures regarding stoichiometry, temperature ramping, and workup protocols are essential for consistent results. For a comprehensive guide on executing this transformation, please refer to the step-by-step instructions below.
- Combine palladium acetate catalyst, DPEphos ligand, trifluoroacetic anhydride, benzyl formate, and tertiary amine in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 120°C and 140°C, preferably 130°C, and maintain stirring for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target aryl acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical novelty. The shift towards using benzyl formate and tertiary amines addresses several critical pain points in the sourcing and manufacturing of fine chemicals. By eliminating the reliance on high-pressure carbon monoxide infrastructure, facilities can reduce capital expenditure on specialized safety equipment and lower insurance premiums associated with hazardous gas storage. Furthermore, the use of commercially available and inexpensive starting materials ensures a stable supply chain, mitigating the risk of raw material shortages that often plague the production of specialty intermediates. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the simplification of the reaction setup and the elimination of expensive oxidants. Since benzyl formate acts as both the reactant and the CO source, the need for complex gas feeding systems is removed, leading to significant operational cost savings. Additionally, the high reaction efficiency and broad functional group tolerance mean that fewer batches are rejected due to quality issues, effectively lowering the cost per kilogram of the final product. The streamlined post-treatment process, which typically involves simple filtration and chromatography, further reduces labor and solvent consumption costs compared to multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: Sourcing reliability is bolstered by the use of commodity chemicals like formic acid, benzyl alcohol, and common tertiary amines, all of which have robust global supply chains. Unlike specialized reagents that may have single-source suppliers, the inputs for this reaction are widely produced, reducing the risk of supply disruptions. This diversity in sourcing options allows procurement teams to negotiate better terms and secure long-term contracts with confidence. Moreover, the scalability of the reaction from gram to ton scale ensures that supply can be rapidly ramped up to meet surges in demand without compromising on the quality or consistency of the aryl acetamide intermediates supplied to downstream manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The avoidance of toxic oxidants and explosive gas mixtures reduces the environmental footprint of the manufacturing process, simplifying waste treatment and disposal compliance. The reaction generates fewer hazardous byproducts, easing the burden on effluent treatment plants and lowering the costs associated with environmental permits. As regulatory scrutiny on chemical manufacturing intensifies globally, adopting such cleaner technologies positions suppliers as preferred partners for environmentally conscious multinational corporations seeking to reduce their Scope 3 emissions and enhance their sustainability credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the feasibility and advantages of the method for potential partners and technical evaluators.
Q: What is the primary advantage of using benzyl formate over carbon monoxide gas?
A: Benzyl formate serves as a safe, liquid carbon monoxide surrogate, eliminating the need for high-pressure gas equipment and mitigating the safety risks associated with handling explosive CO/O2 mixtures typically required in traditional carbonylation reactions.
Q: Can this method activate the C-N bond of tertiary amines without oxidants?
A: Yes, the patented process uniquely facilitates the cleavage of the C-N bond in tertiary amines without requiring additional external oxidants, which simplifies the reaction system and reduces impurity formation compared to conventional oxidative carbonylation strategies.
Q: What is the substrate scope for the aryl group in this synthesis?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituted aryl groups including those with alkyl, alkoxy, trifluoromethyl, cyano, and halogen substituents, as well as heteroaryl systems like furan and thiophene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111978194B for the production of high-value aryl acetamides. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aryl acetamide delivered meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging cutting-edge chemistry to drive value for our partners.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in palladium-catalyzed transformations can accelerate your path to market.
