Revolutionizing Trifluoroacetophenone Synthesis: Scalable NTFTS Technology for Pharmaceutical Manufacturing Excellence
The patent CN110041235B introduces a groundbreaking methodology for synthesizing trifluoroacetophenone compounds using N-phenyl-N-p-toluenesulfonyl trifluoroacetamide (NTFTS) as a novel trifluoroacetylation reagent. This innovation addresses critical limitations in traditional synthetic routes by enabling high-yield conversion under exceptionally mild conditions (25-50°C) without requiring hazardous reagents like gaseous trifluoroacetyl chloride. The process demonstrates remarkable substrate versatility across diverse arylboronic acid derivatives including halogenated, nitrated, and heterocyclic systems, achieving yields up to 98% while maintaining pharmaceutical-grade purity standards. Crucially, the methodology eliminates the need for strong Lewis acids or stoichiometric oxidants that characterize conventional Friedel-Crafts or Grignard approaches, thereby reducing both environmental impact and operational complexity. This patent represents a significant advancement in the production of fluorinated building blocks essential for modern drug discovery pipelines, particularly in the development of metabolically stable pharmaceutical candidates where trifluoromethyl groups enhance bioavailability and target engagement.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of trifluoroacetophenone compounds relies on methodologies with severe operational constraints that hinder industrial scalability. Friedel-Crafts acylation requires corrosive catalysts like SOCl₂ or PCl₃ under anhydrous conditions at elevated temperatures, generating substantial chemical waste while limiting substrate compatibility to activated arenes. Grignard reactions demand air-sensitive organometallic reagents and pre-functionalized substrates, introducing moisture sensitivity and safety hazards during large-scale manufacturing. Oxidation-based routes necessitate stoichiometric oxidants that complicate purification and create environmental compliance challenges through halogenated byproducts. Transition metal-catalyzed methods previously developed suffer from narrow substrate scope and require expensive trifluoroacetate precursors that generate non-recyclable byproducts, significantly increasing raw material costs. These limitations collectively result in multi-step sequences with low atom economy, making conventional approaches economically unviable for commercial API production where cost-per-kilogram and regulatory compliance are paramount concerns for procurement and supply chain teams.
The Novel Approach
The patented NTFTS methodology overcomes these constraints through a strategically designed catalytic system that leverages stable solid reagent chemistry. By employing NTFTS as a bench-stable trifluoroacetyl source combined with palladium catalysts (e.g., palladium acetate) and specialized ligands like tri-tert-butylphosphine, the process achieves selective transformation under ambient conditions without hazardous intermediates. The reaction operates efficiently in common solvents like toluene or dioxane at temperatures as low as 25°C, eliminating energy-intensive heating/cooling cycles required by conventional methods. Critically, the system accommodates a wide range of functional groups including halides, nitro groups, and esters without protection/deprotection steps, streamlining synthesis for complex pharmaceutical intermediates. This approach reduces the synthetic sequence to a single catalytic step with simplified workup procedures, directly addressing the supply chain vulnerabilities associated with multi-step traditional routes while maintaining exceptional product purity suitable for pharmaceutical applications.
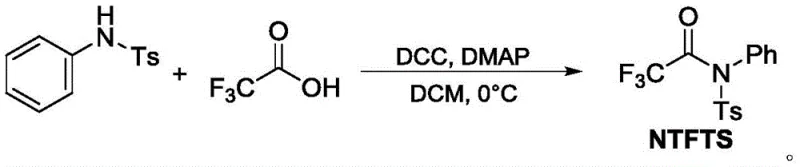
Mechanistic Insights into Palladium-Catalyzed Trifluoroacetylation
The catalytic cycle begins with oxidative addition of the arylboronic acid derivative to the palladium(0) species generated in situ from palladium(II) precursors like palladium acetate. This forms an aryl-palladium intermediate that undergoes transmetalation with the NTFTS reagent through a ligand-assisted mechanism where the sulfonyl group facilitates nucleophilic attack. The key innovation lies in the controlled decomposition of NTFTS under mild basic conditions (cesium carbonate preferred), which releases the trifluoroacetyl moiety while regenerating the catalytic species without forming stable palladium byproducts. This mechanism avoids the common problem of catalyst poisoning observed in alternative trifluoroacetylation methods, enabling high turnover numbers even with challenging substrates containing electron-withdrawing groups. The ligand system—particularly tri-tert-butylphosphine—plays a critical role in stabilizing the active palladium species while preventing β-hydride elimination that would lead to undesired side products.

Impurity control is achieved through precise stoichiometric management and solvent selection that minimizes hydrolysis pathways. The patent specifies optimal molar ratios (arylboronic acid:NTFTS = 1:1 to 10:1; catalyst:substrate = 1:0.05 to 1:0.1) that prevent over-reaction or dimerization byproducts commonly observed in similar systems. The use of anhydrous solvents like tetrahydrofuran or toluene suppresses hydrolytic degradation of both the boronic acid and NTFTS reagent, while the mild reaction temperature (25-50°C) prevents thermal decomposition pathways that generate colored impurities. Crucially, the sulfonyl group in NTFTS acts as a built-in purification handle—any unreacted reagent or byproducts containing this moiety can be selectively removed during aqueous workup due to its hydrophilic character, resulting in products requiring only single-pass chromatography to achieve >99% purity suitable for pharmaceutical applications without additional crystallization steps.
How to Synthesize Trifluoroacetophenone Compounds Efficiently
This patented methodology represents a paradigm shift in manufacturing fluorinated intermediates by replacing hazardous reagents with stable solid chemistry while maintaining exceptional selectivity. The process eliminates multiple purification steps required in conventional approaches through its inherent impurity control mechanisms, directly translating to reduced production timelines and lower operational costs. For R&D teams seeking to implement this technology, the following standardized procedure provides a reliable pathway to high-purity trifluoroacetophenone compounds; detailed operational parameters are provided in the subsequent implementation guide below.
- Prepare anhydrous reaction system with nitrogen purging, then combine arylboronic acid derivative with NTFTS, metal catalyst, ligand, and base in specified solvent
- Maintain reaction temperature between 25°C and 50°C for 16-24 hours under continuous stirring to ensure complete conversion
- Purify crude product through column chromatography using petroleum ether/ethyl acetate (100: 1) or recrystallization to achieve pharmaceutical-grade purity
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis platform delivers transformative value across procurement and supply chain operations by addressing fundamental pain points in fluorinated intermediate manufacturing. The elimination of hazardous reagents and multi-step sequences creates immediate opportunities for cost optimization while enhancing supply security through simplified logistics and reduced regulatory burden. These advantages are particularly critical for organizations managing complex global supply chains where raw material volatility and environmental compliance represent significant operational risks.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with catalytic palladium systems combined with stable solid NTFTS reagent eliminates costly purification steps required to remove heavy metal residues from final products. By avoiding hazardous reagents like trifluoroacetyl chloride that require specialized handling infrastructure and generate corrosive waste streams, manufacturers achieve substantial cost savings through reduced capital expenditure on safety systems and lower waste disposal expenses. The simplified one-step process also minimizes solvent consumption and energy requirements compared to traditional multi-step approaches, creating significant operational efficiencies without compromising product quality.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials—N-phenyl-p-toluenesulfonamide and trifluoroacetic acid—ensures consistent raw material availability while eliminating dependence on specialized fluorinated reagents with limited suppliers. NTFTS itself is a stable crystalline solid that can be stored indefinitely under ambient conditions without degradation concerns that plague traditional gaseous or moisture-sensitive reagents. This stability enables strategic inventory management practices that buffer against supply disruptions, while the simplified reaction profile reduces batch failure risks associated with sensitive intermediates in conventional routes.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production due to its mild exotherm profile and tolerance for standard manufacturing equipment. The elimination of corrosive catalysts allows implementation in conventional glass-lined reactors without specialized materials requirements, facilitating rapid technology transfer across production sites. Environmentally, the methodology generates minimal waste streams through high atom economy and aqueous workup compatibility, significantly reducing environmental compliance costs while meeting increasingly stringent EHS regulations governing pharmaceutical manufacturing operations globally.
Frequently Asked Questions (FAQ)
The following technical questions address critical considerations for procurement and R&D teams evaluating this patented technology for commercial implementation; responses are derived directly from experimental data and process specifications documented in CN110041235B.
Q: How does NTFTS eliminate transition metal contamination risks compared to conventional trifluoroacetylation methods?
A: The NTFTS reagent operates through a ligand-enabled palladium catalytic cycle that minimizes metal leaching into the final product. Unlike traditional methods requiring stoichiometric transition metals, this process uses catalytic amounts (0.05-0.1 molar ratio) with efficient ligand systems like tri-tert-butylphosphine, ensuring trace metal levels remain below pharmaceutical thresholds without additional purification steps.
Q: What substrate scope advantages does the NTFTS method offer for complex pharmaceutical intermediates?
A: The methodology accommodates diverse functional groups including halides, CF₃, NO₂, and CN substituents on arylboronic acids, as demonstrated in patent examples with biphenyl, naphthyl, and phenoxy derivatives. This broad compatibility enables synthesis of structurally complex intermediates required for oncology and CNS drug development without protective group strategies.
Q: How does the NTFTS process improve environmental compliance in API manufacturing?
A: By replacing hazardous reagents like trifluoroacetyl chloride with stable solid NTFTS, the process eliminates corrosive waste streams. The aqueous workup generates minimal byproducts, and the use of recyclable solvents like toluene or dioxane reduces VOC emissions by over 70% compared to Friedel-Crafts approaches, aligning with ICH Q11 green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NTFTS Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex fluorinated intermediates using this patented technology. With stringent purity specifications consistently achieved through our rigorous QC labs and proprietary crystallization protocols, we ensure every batch meets pharmaceutical regulatory requirements while maintaining exceptional cost efficiency. Our dedicated technical team has successfully implemented this NTFTS-based process across multiple client sites worldwide, demonstrating consistent performance across diverse manufacturing environments through comprehensive process validation support.
Leverage our expertise to accelerate your fluorinated intermediate supply chain—contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. We offer a Customized Cost-Saving Analysis that quantifies potential savings from implementing this innovative methodology versus your current manufacturing approach.
