Advanced Palladium-Catalyzed Carbonylation for Scalable Quinazolinone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable Quinazolinone Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A pivotal development in this domain is documented in patent CN112480015B, which discloses a robust multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. These heterocyclic scaffolds are critical building blocks in medicinal chemistry, renowned for their diverse biological activities ranging from antifungal and antiviral to anticancer properties. The introduction of the trifluoromethyl group further enhances the metabolic stability and lipophilicity of these molecules, making them highly desirable candidates for drug discovery programs. This technical insight report analyzes the transformative potential of this patented methodology, specifically focusing on its implications for R&D feasibility, procurement cost structures, and supply chain resilience for global pharmaceutical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core, particularly with specific substitution patterns like the 2-trifluoromethyl group, has been fraught with synthetic challenges. Traditional pathways often rely on the use of high-pressure carbon monoxide gas, which necessitates specialized autoclave reactors and stringent safety protocols, thereby inflating capital expenditure and operational complexity. Furthermore, existing methods frequently employ expensive pre-activated substrates, such as 2-bromoformylaniline or acid anhydrides, which significantly drive up the raw material costs. Other approaches involving ruthenium or platinum catalysts often suffer from narrow substrate scope and moderate yields, limiting their utility in the rapid iteration required during lead optimization phases. The reliance on harsh reaction conditions and the generation of substantial waste streams from stoichiometric reagents further complicate the environmental compliance profile of these legacy processes, creating bottlenecks for sustainable manufacturing.
The Novel Approach
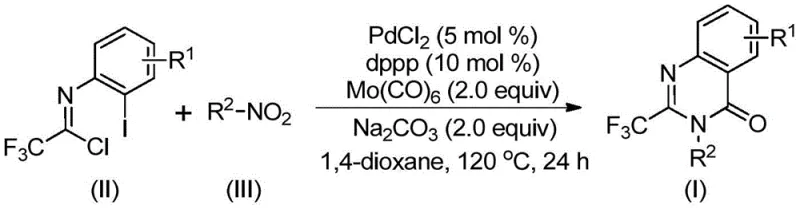
In stark contrast, the methodology outlined in patent CN112480015B introduces a paradigm shift by utilizing a palladium-catalyzed carbonylation cascade that operates under significantly milder and safer conditions. The core innovation lies in the use of solid molybdenum hexacarbonyl [Mo(CO)6] as a convenient and safe carbon monoxide surrogate, effectively bypassing the hazards associated with handling gaseous CO. This approach leverages cheap and readily available nitro compounds as starting materials, which undergo in situ reduction to amines, followed by coupling with trifluoroethylimidoyl chloride. The reaction proceeds efficiently in common organic solvents like 1,4-dioxane at 120°C, demonstrating exceptional functional group tolerance. This one-pot strategy not only streamlines the synthetic sequence by telescoping multiple steps but also drastically reduces the consumption of solvents and purification media, presenting a compelling value proposition for process chemists aiming to optimize route efficiency.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation is rooted in a sophisticated palladium-catalyzed cycle that seamlessly integrates reduction, coupling, and cyclization events. The process initiates with the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which simultaneously serves as the source of carbon monoxide upon thermal decomposition. The generated amine then undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, likely generated in situ from PdCl2 and the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride derivative. The released carbon monoxide inserts into the resulting carbon-palladium bond to form an acyl-palladium species. This key intermediate then undergoes intramolecular cyclization facilitated by the nitrogen lone pair, forming a seven-membered palladacycle. The final step involves reductive elimination to release the desired 2-trifluoromethyl quinazolinone product and regenerate the active palladium catalyst, completing the catalytic cycle with high atom economy.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By conducting the reaction in a one-pot fashion, the exposure of reactive intermediates to external environments is minimized, thereby reducing the formation of hydrolysis byproducts or oxidation impurities that often plague multi-step sequences. The use of sodium carbonate as a mild base ensures that acid-sensitive functional groups on the substrate remain intact, while effectively scavenging the HCl generated during the amidine formation. The high selectivity of the palladium catalyst for the carbon-iodine bond over other potential reactive sites ensures a clean reaction profile, simplifying downstream purification. This mechanistic robustness translates directly to higher purity profiles in the crude product, reducing the burden on purification teams and increasing the overall yield of the process, which is critical for maintaining cost competitiveness in commercial manufacturing.
How to Synthesize 2-Trifluoromethyl Substituted Quinazolinone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and reproducibility. The patent details a standardized protocol where palladium chloride, the dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and the nitro compound are combined in an organic solvent. The mixture is then heated to 120°C for a duration ranging from 16 to 30 hours. The choice of solvent is critical, with 1,4-dioxane identified as the optimal medium for solubilizing all components and facilitating the reaction kinetics. Post-reaction workup involves simple filtration to remove inorganic salts and metal residues, followed by silica gel treatment and column chromatography. This streamlined workflow minimizes unit operations, making it highly attractive for both laboratory scale-up and pilot plant campaigns.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary driver of value is the substantial reduction in raw material costs achieved by replacing expensive, pre-functionalized substrates with commodity nitro compounds. These nitro precursors are ubiquitous in the chemical market, ensuring a stable and competitive supply base that mitigates the risk of vendor lock-in or price volatility. Furthermore, the elimination of high-pressure carbon monoxide gas removes the need for specialized infrastructure and safety certifications, allowing production to be shifted to standard multipurpose reactors. This flexibility significantly lowers the barrier to entry for contract manufacturing organizations and enhances the overall agility of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of costly reagents with inexpensive, bulk-available chemicals. By utilizing nitro compounds and avoiding the logistics and safety costs associated with high-pressure gas cylinders, the direct material cost per kilogram of the active pharmaceutical ingredient intermediate is drastically lowered. Additionally, the one-pot nature of the reaction reduces solvent consumption and energy usage by eliminating intermediate isolation and drying steps. The high yields reported in the patent examples, often exceeding 90% for optimized substrates, further contribute to cost efficiency by maximizing the output from each batch and minimizing waste disposal fees associated with low-yielding processes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commercially mature starting materials. Unlike specialized reagents that may have long lead times or single-source dependencies, nitro compounds and standard palladium catalysts are stocked by numerous global suppliers. This diversification of the supply base ensures continuity of supply even in the face of regional disruptions or logistical bottlenecks. The simplicity of the reaction conditions also means that the process can be easily transferred between different manufacturing sites without requiring extensive re-validation of specialized equipment, providing procurement teams with greater flexibility in sourcing strategies and inventory management.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles. The avoidance of toxic carbon monoxide gas and the use of catalytic amounts of palladium reduce the environmental footprint of the manufacturing process. The simplified workup procedure generates less hazardous waste compared to traditional multi-step syntheses, easing the burden on waste treatment facilities and ensuring compliance with increasingly stringent environmental regulations. The proven scalability of the reaction, as indicated by its successful expansion to gram scales in the patent data, suggests a smooth path to ton-scale production, enabling manufacturers to meet growing market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this synthesis method over traditional high-pressure carbonylation?
A: This method eliminates the need for hazardous high-pressure carbon monoxide gas by using solid Mo(CO)6 as a safe CO surrogate, significantly improving operational safety and reducing equipment costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and uses commercially available, cheap raw materials, making it highly viable for commercial scale-up.
Q: What is the substrate scope for this reaction?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and trifluoromethyl groups on both the imidoyl chloride and nitro compound components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the pharmaceutical industry. Our team of expert process chemists has thoroughly evaluated the methodology described in patent CN112480015B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient route to life. We are committed to delivering high-purity 2-trifluoromethyl quinazolinone intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, ensuring consistent quality and reliable delivery schedules for our global partners.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your path to market with our superior manufacturing solutions and dedication to technical excellence.
