Revolutionizing Indole Production: A Green Water-Phase Catalytic Approach for Commercial Scale-Up
The global demand for high-purity indole derivatives continues to surge, driven by their critical role as scaffolds in pharmaceutical agents, agrochemicals, and advanced functional materials. However, traditional synthetic routes often rely heavily on volatile organic compounds (VOCs) and harsh reaction conditions, presenting significant challenges for modern sustainable manufacturing. A groundbreaking solution to these inefficiencies is detailed in Chinese Patent CN101906061B, which discloses a novel method for preparing indole derivatives via a one-pot reaction in a pure aqueous phase. This technology utilizes a specialized water-soluble coordination compound as a catalyst to efficiently couple ortho-haloanilines with terminal alkynes. By shifting the reaction medium from toxic organic solvents to water, this innovation not only aligns with the principles of green chemistry but also offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. The process is characterized by its operational simplicity, high yield, and exceptional selectivity, making it a transformative approach for the fine chemical industry.
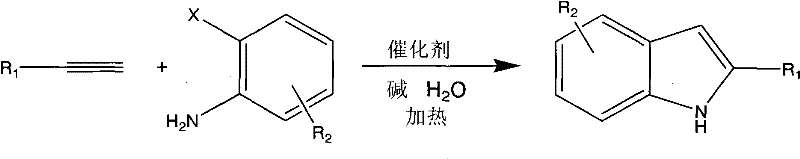
This patent represents a paradigm shift in heterocyclic synthesis, moving away from the legacy reliance on organic media that has plagued the sector for decades. The core innovation lies in the deployment of a water-soluble metal copper complex that remains active and stable in an aqueous environment, facilitating the cyclization process with remarkable efficiency. For R&D directors and process chemists, this offers a compelling alternative to conventional methods that often suffer from low atom economy and difficult waste disposal. The ability to conduct these reactions in water without the need for protecting groups further streamlines the synthetic route, reducing the overall step count and enhancing the economic viability of producing high-purity indole derivatives for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of indoles has been dominated by methods that utilize organic solvents or high-temperature gas-phase reactions, both of which carry substantial drawbacks. Traditional processes, such as those involving aniline and ethylene glycol, often require elevated temperatures and employ catalysts with significant toxicity profiles, posing risks to operator health and environmental safety. Furthermore, the reliance on organic solvents like DMF, DMSO, or toluene introduces severe complications regarding solvent recovery, recycling, and final product purification. These solvents are not only expensive to procure and dispose of but also contribute to a heavy environmental footprint due to their volatility and potential for groundwater contamination. Additionally, many conventional transition-metal catalyzed methods exhibit poor functional group tolerance, limiting the structural diversity of the resulting indole library and necessitating additional synthetic steps to protect sensitive moieties. The cumulative effect of these limitations is a manufacturing process that is costly, hazardous, and increasingly non-compliant with stringent global environmental regulations.
The Novel Approach
In stark contrast, the methodology described in CN101906061B leverages the unique properties of water as a reaction medium to overcome these entrenched barriers. By employing a water-soluble copper complex catalyst, the reaction proceeds smoothly in a pure aqueous phase, eliminating the need for flammable and toxic organic solvents entirely. This shift drastically simplifies the post-reaction workup; since the product is often insoluble in water, it can be easily separated via filtration or simple extraction, bypassing the energy-intensive distillation processes required for organic solvent removal. The method demonstrates exceptional versatility, accommodating a wide range of substituents including electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like methoxy and methyl. This broad substrate scope ensures that the process is applicable to the synthesis of a diverse array of indole analogs without compromising yield or purity. Moreover, the reaction conditions are mild, typically operating between 60°C and 130°C, which reduces energy consumption and enhances the safety profile of the operation, making it an ideal candidate for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Water-Soluble Copper Complex Catalysis
The efficacy of this aqueous synthesis hinges on the specific design of the catalyst, a water-soluble metallic copper complex denoted as structure (I) in the patent documentation. Unlike traditional heterogeneous catalysts that may suffer from leaching or deactivation in water, this homogeneous copper complex maintains high catalytic activity throughout the reaction cycle. The mechanism likely involves the initial coordination of the terminal alkyne to the copper center, followed by oxidative addition of the ortho-haloaniline. The aqueous environment plays a crucial role here, potentially stabilizing charged intermediates through hydrogen bonding and hydrophobic effects, which accelerate the rate-determining steps of the coupling and subsequent intramolecular cyclization. The presence of a base, such as sodium hydroxide or potassium hydroxide, is essential for deprotonating the alkyne and facilitating the reductive elimination step that releases the final indole product. This mechanistic pathway ensures high turnover numbers and minimizes the formation of side products, such as homocoupled alkynes or dehalogenated anilines, which are common impurities in non-optimized systems.
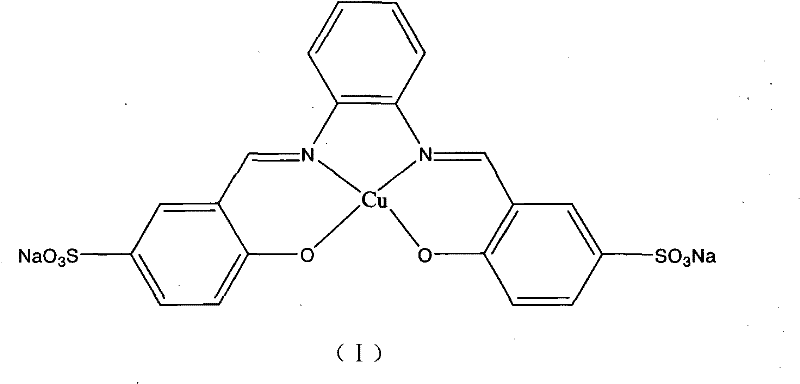
From an impurity control perspective, the use of this specific copper complex in water offers distinct advantages over organic-phase counterparts. The high selectivity of the catalyst prevents the degradation of sensitive functional groups that might otherwise react under harsher acidic or basic conditions found in traditional indole syntheses like the Fischer indole synthesis. The patent data indicates that even with substrates containing nitro groups, which are prone to reduction, the reaction proceeds cleanly to the desired indole without significant byproduct formation. This level of chemoselectivity is critical for pharmaceutical applications where impurity profiles are strictly regulated. Furthermore, the water-soluble nature of the catalyst allows for potential recycling strategies; after the organic product is extracted, the aqueous phase containing the catalyst could theoretically be reused, further driving down the cost of goods and minimizing heavy metal waste. This mechanistic robustness provides R&D teams with the confidence to scale the process, knowing that the reaction kinetics and selectivity are well-understood and controllable.
How to Synthesize Indole Derivatives Efficiently
To implement this technology effectively, process engineers must adhere to specific parameters regarding stoichiometry and reaction conditions to maximize yield and purity. The patent outlines a straightforward protocol where ortho-haloaniline and terminal alkyne are mixed in water with the copper catalyst and a base. The molar ratios are flexible, with the alkyne typically used in slight excess to drive the reaction to completion. The choice of base is also critical, with inorganic bases like NaOH or KOH showing superior performance compared to organic amines. Temperature control is another key variable; while the reaction can proceed at lower temperatures, heating to the optimal range of 100°C to 130°C significantly reduces reaction time and improves conversion rates. For detailed operational procedures and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining o-haloaniline, terminal alkyne, and a water-soluble copper complex catalyst in pure water.
- Add an inorganic base such as sodium hydroxide or potassium hydroxide to facilitate the coupling and cyclization process.
- Heat the reaction mixture to temperatures between 60°C and 130°C for 4 to 30 hours to achieve high conversion yields.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous catalytic technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials and utilities. By replacing expensive, specialty-grade organic solvents with ubiquitous and inexpensive water, the direct material costs associated with the reaction medium are virtually eliminated. This substitution also removes the logistical burden of storing and handling large volumes of flammable liquids, thereby reducing insurance premiums and compliance costs related to hazardous material storage. Furthermore, the simplified workup procedure—often requiring only filtration or a single extraction—reduces the demand for energy-intensive distillation equipment and shortens the batch cycle time. These factors collectively contribute to a leaner, more cost-effective manufacturing process that enhances the overall competitiveness of the final product in the global market.
- Cost Reduction in Manufacturing: The elimination of organic solvents represents a massive opportunity for cost optimization. Organic solvents not only carry a high purchase price but also incur significant costs for recovery, recycling, or disposal as hazardous waste. By switching to water, the process avoids these expenses entirely. Additionally, the high yields reported in the patent examples, often exceeding 90%, mean that less raw material is wasted, improving the overall atom economy. The use of a robust copper catalyst, which is generally cheaper than precious metals like palladium or platinum, further lowers the catalyst cost per kilogram of product. This combination of reduced solvent costs, higher yields, and cheaper catalysts creates a compelling economic case for adopting this technology in large-scale production facilities.
- Enhanced Supply Chain Reliability: Relying on water as the primary solvent inherently de-risks the supply chain. Organic solvents are subject to market volatility, petrochemical feedstock fluctuations, and strict transportation regulations that can lead to delays. Water, conversely, is universally available and immune to such supply shocks. Moreover, the starting materials—ortho-haloanilines and terminal alkynes—are commodity chemicals with well-established global supply networks. The tolerance of the process for various substituents means that procurement teams have flexibility in sourcing different grades or suppliers of raw materials without needing to re-validate the entire process. This flexibility ensures continuity of supply even when specific raw material markets experience temporary disruptions, safeguarding production schedules against external volatility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor is often hindered by heat transfer and safety issues associated with organic solvents. Water has a high heat capacity, which aids in temperature control during exothermic reactions, making the scale-up process safer and more predictable. The absence of flammable vapors significantly reduces the risk of fire and explosion, allowing for simpler reactor designs and lower safety infrastructure costs. From an environmental standpoint, this process generates significantly less hazardous waste, aligning with increasingly strict global environmental regulations such as REACH and TSCA. This compliance advantage future-proofs the manufacturing site against regulatory tightening and enhances the corporate sustainability profile, which is becoming a key differentiator in B2B negotiations with major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this water-phase indole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What are the primary advantages of using a water-phase system for indole synthesis?
A: The water-phase system eliminates the need for toxic and expensive organic solvents, significantly reducing environmental pollution and operational hazards while simplifying product separation and purification.
Q: Does this catalytic method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substituents such as nitro, chloro, bromo, methoxy, and methyl groups on both the aniline and alkyne substrates.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The use of water as a solvent enhances safety by removing flammability risks associated with organic solvents, and the simple workup procedure supports efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
The technological breakthroughs detailed in CN101906061B underscore the immense potential of green chemistry to redefine the production of critical pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative laboratory methods into commercial reality requires deep expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this aqueous catalytic method are fully realized at an industrial level. Our facilities are equipped with rigorous QC labs and advanced processing units capable of maintaining stringent purity specifications, guaranteeing that every batch of indole derivative meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking partners to collaborate with us to leverage this efficient and sustainable synthesis route. By integrating this technology into your supply chain, you can achieve significant operational efficiencies while adhering to the highest environmental standards. We encourage you to contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments, helping you make informed decisions that drive value and innovation in your drug development pipeline.
