Advanced Palladium-Catalyzed Carbonylation for Scalable Arylacetamide Manufacturing
Advanced Palladium-Catalyzed Carbonylation for Scalable Arylacetamide Manufacturing
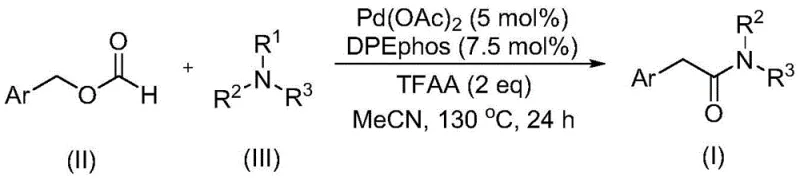
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries, particularly regarding safety, efficiency, and scalability. A significant breakthrough in this domain is documented in patent CN111978194A, which details a robust preparation method for arylacetamide compounds. This technology leverages a transition metal-catalyzed carbonylation strategy that fundamentally shifts the paradigm from hazardous gas handling to safer liquid reagents. By utilizing benzyl formate as both a carbon monoxide source and a reactant, coupled with tertiary amines as the nitrogen source, this method offers a streamlined pathway to valuable amide scaffolds. The process operates under relatively mild thermal conditions compared to traditional high-pressure carbonylations, achieving high conversion rates with excellent functional group tolerance. For R&D directors and process chemists, this represents a critical advancement in accessing complex molecular architectures without the logistical burdens associated with gaseous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of arylacetamide compounds has relied heavily on the direct amidation of phenylacetic acid and its derivatives. While conceptually straightforward, this classical approach often suffers from significant drawbacks, including the requirement for harsh activating agents, elevated temperatures, and the generation of substantial stoichiometric waste. Furthermore, alternative transition metal-catalyzed carbonylation strategies have historically depended on the use of carbon monoxide gas. Handling CO gas presents severe safety challenges due to its toxicity and the potential formation of explosive mixtures with oxygen, necessitating specialized high-pressure reactors and rigorous safety protocols that inflate capital expenditure. Additionally, the activation of tertiary amines via C-N bond cleavage has remained a formidable challenge in organic synthesis, often requiring excessive amounts of strong oxidants that complicate downstream purification and increase the environmental footprint of the manufacturing process.
The Novel Approach
The methodology outlined in the patent introduces a transformative solution by replacing gaseous carbon monoxide with benzyl formate, a stable and easily handled liquid. This substitution not only mitigates safety risks but also simplifies the reactor setup, allowing for standard pressure vessels to be used effectively. The reaction system employs a palladium catalyst, specifically palladium acetate, in conjunction with the bidentate ligand DPEphos, to facilitate the carbonylation process efficiently. Crucially, the protocol achieves the cleavage of the C-N bond in tertiary amines without the addition of external oxidants, a feat that significantly streamlines the reaction profile. This novel approach ensures that the starting materials, which are commercially available and cost-effective, are converted into high-value arylacetamides with remarkable efficiency, as evidenced by yields reaching up to 87% in optimized examples.
Mechanistic Insights into Pd-Catalyzed Carbonylative C-N Bond Activation
At the heart of this synthetic innovation lies a sophisticated palladium-catalyzed cycle that orchestrates the insertion of a carbonyl group between the aryl methyl fragment and the amine nitrogen. The mechanism initiates with the oxidative addition or activation of the benzyl formate by the palladium species, effectively generating a palladium-hydride or palladium-acyl intermediate while releasing the carbonyl unit in situ. This avoids the need for external CO tanks. The presence of trifluoroacetic anhydride (TFAA) plays a pivotal role as an activator, likely assisting in the generation of the reactive acyl-palladium species and promoting the subsequent nucleophilic attack by the amine. The use of DPEphos as a ligand is strategic; its wide bite angle stabilizes the palladium center and facilitates the reductive elimination step, which is often the rate-determining step in such cross-coupling reactions. This careful tuning of the electronic and steric environment around the metal center allows for the activation of the typically inert C-N bond in tertiary amines.
From an impurity control perspective, the mechanism offers distinct advantages. Because the reaction does not rely on harsh oxidants to drive the C-N bond cleavage, the formation of over-oxidized byproducts, such as N-oxides or degraded amine fragments, is minimized. The specificity of the palladium catalyst towards the benzyl formate ensures that the carbonyl insertion occurs selectively at the desired position, preserving sensitive functional groups on the aromatic ring, such as halides, ethers, or trifluoromethyl groups. This high level of chemoselectivity is paramount for pharmaceutical applications where impurity profiles must be tightly controlled. The reaction conditions, typically maintained at 130°C in acetonitrile, provide sufficient energy to overcome activation barriers without inducing thermal decomposition of the product, resulting in a clean reaction mixture that simplifies the final isolation and purification steps.
How to Synthesize Arylacetamide Efficiently
Implementing this synthesis route requires precise attention to reagent ratios and reaction parameters to maximize yield and purity. The protocol is designed to be operationally simple, making it highly attractive for scale-up operations. The standard procedure involves charging a sealed tube or reactor with the palladium catalyst, ligand, activator, benzyl formate, and the specific tertiary amine substrate in an aprotic solvent like acetonitrile. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated by experimental data, are provided in the guide below.
- Charge a reaction vessel with palladium acetate catalyst, DPEphos ligand, trifluoroacetic anhydride, benzyl formate, and tertiary amine in acetonitrile solvent.
- Heat the reaction mixture to 130°C and maintain stirring for 24 hours to ensure complete conversion of starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target arylacetamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational improvements and risk mitigation. The shift away from gaseous carbon monoxide eliminates the need for specialized gas supply contracts and the associated logistics of handling hazardous cylinders, thereby reducing the overall complexity of the supply chain. Furthermore, the raw materials utilized in this process, such as benzyl formate and common tertiary amines, are commodity chemicals that are widely available from multiple global suppliers, ensuring a robust and resilient supply base that is not subject to the bottlenecks often seen with specialized reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and hazardous reagents. By avoiding the use of high-pressure CO gas infrastructure and external oxidants, the capital and operational expenditures associated with safety compliance and waste treatment are drastically reduced. The high atom economy of using benzyl formate as a dual-purpose reagent further contributes to cost efficiency, as fewer equivalents of reagents are wasted. Additionally, the simplified workup procedure, which typically involves filtration and standard chromatography, reduces the labor and solvent costs associated with downstream processing, leading to a more favorable cost of goods sold (COGS) profile for the final arylacetamide products.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of stable, shelf-stable liquid reagents that do not require cryogenic storage or immediate consumption upon delivery. Benzyl formate and tertiary amines can be stocked in bulk without significant degradation, allowing manufacturers to maintain healthy inventory levels and buffer against market fluctuations. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that the same process platform can be adapted for various derivatives without extensive re-optimization, ensuring consistent production schedules and faster time-to-market for new product variants.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the absence of gas-liquid mass transfer limitations that typically plague carbonylation reactions. The homogeneous nature of the reaction mixture in acetonitrile allows for efficient heat and mass transfer in larger reactors. From an environmental standpoint, the process generates less hazardous waste compared to traditional methods involving stoichiometric oxidants. The ability to achieve high yields with minimal byproduct formation aligns with green chemistry principles, simplifying regulatory compliance and reducing the burden on wastewater treatment facilities, which is a critical factor for sustainable long-term manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for large-scale production.
Q: What is the primary advantage of using benzyl formate over carbon monoxide gas?
A: Benzyl formate serves as a safe, liquid carbon monoxide surrogate, eliminating the severe safety hazards and specialized equipment required for handling explosive CO/O2 mixtures or high-pressure CO gas cylinders.
Q: Does this method require additional oxidants for tertiary amine activation?
A: No, a key innovation of this protocol is that the cleavage of the C-N bond in tertiary amines proceeds efficiently without the need for extra oxidants, simplifying the reaction system and reducing waste.
Q: What represents the typical yield range for this synthesis?
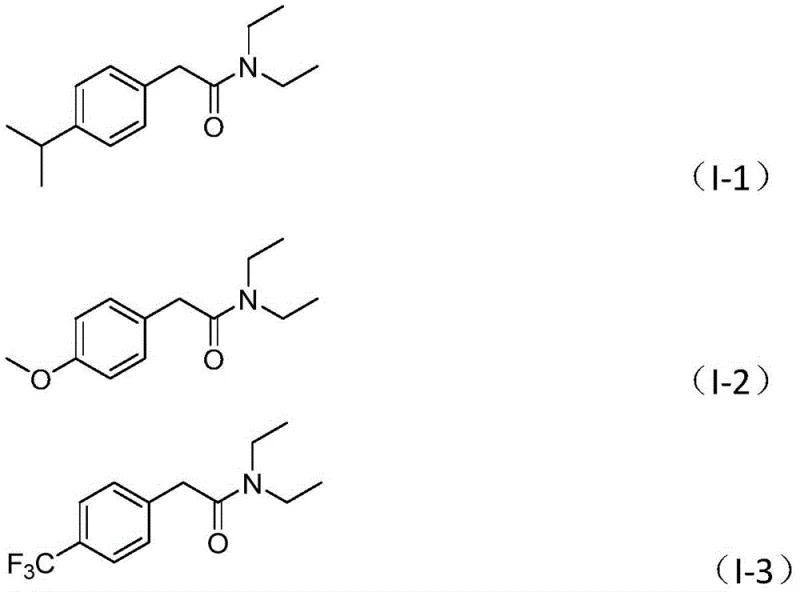
A: Experimental data indicates high efficiency, with isolated yields frequently exceeding 80% for various substrates, including those with electron-donating or electron-withdrawing groups on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylacetamide Supplier
As the demand for high-quality pharmaceutical intermediates continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of modern chemical synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the manufacturing floor. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch, guaranteeing that the arylacetamide compounds we deliver meet the highest industry standards.
We invite you to leverage our technical expertise to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced palladium-catalyzed processes can enhance your product portfolio and drive your business forward.
