Advanced Palladium-Catalyzed Synthesis of Arylacetamides for Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing amide bonds, a ubiquitous motif in bioactive molecules and functional materials. Patent CN111978194A introduces a transformative approach to synthesizing arylacetamide compounds, addressing long-standing challenges in carbonylation chemistry. This innovation leverages a palladium-catalyzed system that utilizes benzyl formate as a safe and effective carbon monoxide source, coupled with readily available tertiary amines. By circumventing the need for toxic gaseous CO and avoiding harsh oxidative conditions typically required for C-N bond activation, this technology offers a streamlined pathway for producing high-value intermediates. For R&D directors and procurement specialists, this represents a significant opportunity to enhance synthetic efficiency while mitigating safety risks associated with traditional carbonylation protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing arylacetamides often rely on the direct amidation of phenylacetic acids or their derivatives, which can suffer from poor atom economy and require aggressive activation agents. Alternatively, transition metal-catalyzed carbonylation has emerged as a powerful tool, yet it frequently necessitates the use of primary or secondary amines, leaving tertiary amines largely underutilized due to the inherent stability of their C-N bonds. Furthermore, conventional carbonylation processes often demand the handling of explosive carbon monoxide and oxygen mixtures or require stoichiometric amounts of hazardous oxidants to facilitate bond cleavage. These limitations not only increase operational complexity and safety liabilities but also restrict the structural diversity of the resulting amide libraries, posing significant hurdles for the rapid development of novel drug candidates and agrochemical agents.
The Novel Approach
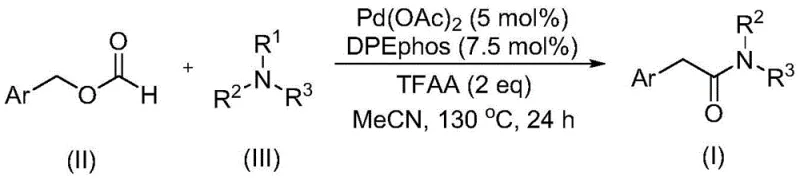
In stark contrast, the methodology disclosed in CN111978194A employs a sophisticated palladium catalytic system that enables the direct carbonylation of tertiary amines using benzyl formate. This approach elegantly bypasses the need for external oxidants, as the catalytic cycle inherently promotes the cleavage of the tertiary amine C-N bond under relatively mild thermal conditions. The use of benzyl formate as a liquid CO surrogate significantly enhances process safety by eliminating the infrastructure requirements for high-pressure gas handling. Moreover, the reaction demonstrates exceptional functional group tolerance, accommodating various substituents on the aryl ring, including electron-donating and electron-withdrawing groups, thereby providing a versatile platform for the late-stage functionalization of complex molecular scaffolds.
Mechanistic Insights into Pd-Catalyzed Carbonylative C-N Cleavage
The core of this technological advancement lies in the synergistic interaction between the palladium catalyst, specifically palladium(II) acetate, and the bidentate phosphine ligand DPEphos. This catalytic ensemble generates a highly active species capable of orchestrating the oxidative addition into the benzylic C-O bond of the formate ester, followed by decarbonylation to release the active carbonyl fragment in situ. The presence of trifluoroacetic anhydride (TFAA) plays a critical role in activating the substrate and stabilizing intermediate species, ensuring high turnover frequencies. Unlike traditional pathways that stall at the tertiary amine stage, this system facilitates a unique migratory insertion and subsequent reductive elimination sequence that effectively breaks the sterically hindered C-N bond, forging the new amide linkage with high precision and minimal byproduct formation.
Impurity control is paramount in the synthesis of pharmaceutical intermediates, and this catalytic protocol offers distinct advantages in managing side reactions. The specificity of the Pd/DPEphos system minimizes homocoupling of the aryl components and prevents over-carbonylation or decomposition of the sensitive amine substrates. The reaction conditions, typically conducted at 130°C in acetonitrile, are optimized to balance reaction kinetics with thermal stability, ensuring that the desired arylacetamide is formed selectively. Post-reaction processing is straightforward, involving simple filtration and silica gel treatment followed by column chromatography, which effectively removes residual palladium species and ligand fragments. This results in a final product profile that meets stringent purity specifications required for downstream applications in drug discovery and process chemistry.
How to Synthesize Arylacetamide Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility. The process begins with the precise weighing of the palladium catalyst and ligand, followed by the addition of the benzyl formate derivative and the chosen tertiary amine in an anhydrous organic solvent. The inclusion of trifluoroacetic anhydride is essential for driving the reaction to completion within a practical timeframe. While the general procedure is robust, scaling this reaction necessitates rigorous control over temperature profiles and mixing efficiency to maintain the integrity of the catalytic cycle. For detailed operational protocols and specific molar ratios tailored to different substrate classes, please refer to the standardized synthesis guide provided below.
- Combine palladium catalyst (Pd(OAc)2), ligand (DPEphos), benzyl formate, tertiary amine, and trifluoroacetic anhydride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C in a sealed vessel and maintain this temperature for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target arylacetamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology addresses several critical pain points associated with the sourcing and manufacturing of arylacetamide intermediates. The shift from hazardous gaseous reagents to stable liquid surrogates like benzyl formate drastically simplifies the supply chain logistics and reduces the capital expenditure required for specialized high-pressure reactor equipment. Furthermore, the ability to utilize inexpensive and widely available tertiary amines as direct building blocks opens up new avenues for cost optimization, allowing manufacturers to leverage bulk commodity chemicals rather than custom-synthesized precursors. This strategic alignment of raw material availability with efficient catalytic转化 creates a resilient supply chain capable of withstanding market fluctuations and raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive oxidants and the use of low-loading palladium catalysts significantly lower the direct material costs associated with production. By avoiding the need for specialized high-pressure CO infrastructure, facilities can reduce both CAPEX and OPEX, leading to substantial overall cost savings. The simplified workup procedure further reduces labor and solvent consumption, enhancing the economic viability of large-scale manufacturing campaigns for these valuable intermediates.
- Enhanced Supply Chain Reliability: Benzyl formate and common tertiary amines are commercially available from multiple global suppliers, ensuring a stable and continuous flow of raw materials. This diversification of the supply base mitigates the risk of single-source dependency and potential disruptions. Additionally, the robustness of the reaction conditions allows for flexible scheduling and batch processing, enabling manufacturers to respond rapidly to changing demand signals from downstream pharmaceutical clients without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions compared to traditional high-pressure carbonylations, making it inherently safer and easier to scale from kilogram to multi-ton quantities. The absence of toxic gas emissions and the reduced generation of hazardous waste streams align with increasingly stringent environmental regulations. This green chemistry profile not only facilitates regulatory approval but also enhances the corporate sustainability metrics of the manufacturing entity, appealing to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this arylacetamide synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent literature, providing a clear framework for evaluating the feasibility of this route for specific project needs. Understanding these nuances is essential for process chemists and project managers aiming to integrate this methodology into their existing development pipelines.
Q: What is the primary advantage of using benzyl formate in this synthesis?
A: Benzyl formate serves a dual role as both a safe carbon monoxide surrogate and a reactant, eliminating the need for handling hazardous high-pressure CO gas while providing the necessary carbonyl group for amide formation.
Q: Can this method utilize tertiary amines effectively?
A: Yes, unlike conventional methods that struggle with tertiary amines, this palladium-catalyzed process facilitates the cleavage of the C-N bond in tertiary amines without requiring additional oxidants, expanding the scope of accessible amide structures.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds in acetonitrile at 130°C for 24 hours using a catalyst system of Pd(OAc)2 and DPEphos, with trifluoroacetic anhydride acting as a promoter to drive the carbonylation efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to maintain a competitive edge in the global marketplace. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the Pd-catalyzed carbonylation described in CN111978194A are seamlessly translated into robust industrial processes. We are committed to delivering high-purity arylacetamide intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch supplied adheres to the highest industry standards, providing our partners with the confidence needed to advance their drug development programs.
We invite you to collaborate with us to explore the full potential of this advanced synthesis route for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how this technology can optimize your budget and timeline. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to accelerate the delivery of life-saving therapies to the market.
