Advanced Palladium-Catalyzed Carbonylation for Scalable Quinolinone Intermediate Production
Introduction to Next-Generation Quinolinone Synthesis
The development of efficient synthetic routes for heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, particularly for privileged structures like quinolin-2(1H)ones which are prevalent in bioactive molecules ranging from antibiotics to antitumor agents. A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a robust preparation method for 3-arylquinolin-2(1H)one derivatives. This technology leverages a sophisticated palladium-catalyzed aminocarbonylation strategy that fundamentally alters the traditional approach to constructing these valuable cores. By utilizing benzisoxazole as a dual-purpose reagent serving as both the nitrogen and formyl source, the process achieves remarkable simplicity and efficiency. The versatility of this method is underscored by its ability to generate a wide array of derivatives, including those with potent biological activities such as MAP Kinase inhibition and HBV inhibition, as illustrated in the structural diversity of known bioactive quinolinones.
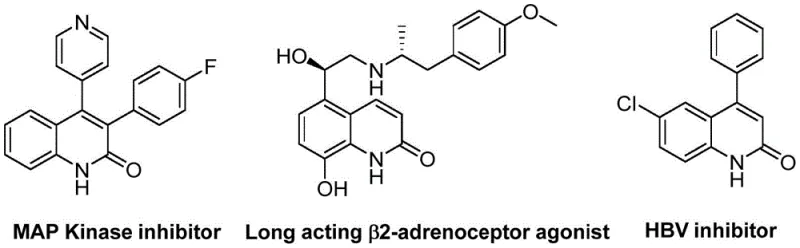
For pharmaceutical manufacturers and R&D teams, the implications of this patent are profound, offering a pathway to access complex heterocycles with reduced step counts and improved operational safety. The method addresses critical pain points in intermediate manufacturing, specifically the reliance on hazardous reagents and multi-step sequences that plague conventional syntheses. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative methodologies is essential for maintaining competitiveness in the global supply chain. The following analysis delves into the technical nuances, mechanistic insights, and commercial advantages of this novel synthetic route, providing a comprehensive overview for decision-makers focused on cost reduction in API manufacturing and the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods are well-established, they often suffer from significant drawbacks that hinder their applicability in modern, large-scale production environments. Traditional routes frequently require harsh reaction conditions, including the use of strong acids, high temperatures, and toxic reagents like phosphorus oxychloride (POCl3), which pose severe safety and environmental challenges. Furthermore, these conventional pathways often lack the necessary regioselectivity and functional group tolerance required for the synthesis of advanced drug candidates, leading to complex impurity profiles that are difficult and costly to purify. The reliance on gaseous carbon monoxide in some transition-metal catalyzed variations introduces additional logistical burdens and safety risks, necessitating specialized high-pressure equipment and rigorous safety protocols that increase capital expenditure and operational complexity.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a palladium-catalyzed system that operates under relatively mild conditions, typically at 100°C in ethylene glycol dimethyl ether (DME). This method ingeniously employs benzisoxazole and benzyl chloride compounds as starting materials, bypassing the need for pre-functionalized anilines or dangerous acylating agents. The reaction proceeds via an aminocarbonylation mechanism where the benzisoxazole ring acts as an internal source of both nitrogen and the carbonyl group, effectively streamlining the atom economy. This one-pot transformation not only simplifies the operational workflow but also significantly enhances the safety profile of the synthesis by eliminating the need for external CO gas. The broad substrate scope allows for the introduction of diverse substituents, enabling the rapid generation of libraries for structure-activity relationship (SAR) studies while maintaining high reaction efficiency and yield.
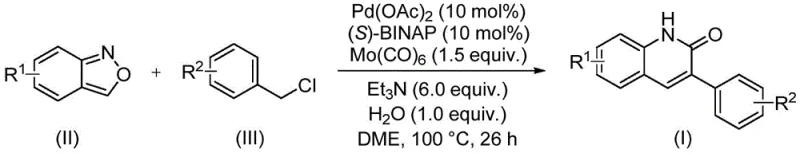
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this innovative synthesis lies in the intricate catalytic cycle driven by the palladium complex formed in situ from palladium acetate and the chiral ligand (S)-1,1'-binaphthyl-2,2'-bis-diphenylphosphine ((S)-BINAP). The mechanism initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species, generating a benzyl-palladium(II) intermediate. Concurrently, the benzisoxazole undergoes coordination and subsequent N-O bond cleavage, a critical step that releases the nitrogen atom required for the heterocycle formation and generates a reactive imine or nitrile oxide species depending on the specific pathway. The presence of molybdenum hexacarbonyl (Mo(CO)6) is pivotal, as it serves as a solid surrogate for carbon monoxide, releasing CO in situ under the reaction conditions to facilitate the carbonylation step. This insertion of the carbonyl group into the palladium-carbon bond is followed by migratory insertion and reductive elimination, ultimately closing the quinolinone ring and regenerating the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic thermal cyclizations. The specificity of the palladium catalyst ensures high regioselectivity, minimizing the formation of isomeric byproducts that often complicate downstream purification. The use of triethylamine as a base and water as a co-additive plays a crucial role in proton management and facilitating the hydrolysis or tautomerization steps necessary to arrive at the final 2(1H)-one tautomer. The tolerance of the catalytic system towards various electronic environments on the aromatic rings—whether electron-withdrawing groups like cyano and trifluorethyl or electron-donating groups like methoxy—suggests a robust catalytic cycle that is not easily poisoned by substrate variations. This mechanistic resilience translates directly to process reliability, ensuring consistent quality and yield across different batches and substrate classes, which is a key metric for high-purity pharmaceutical intermediate production.
How to Synthesize 3-Arylquinolin-2(1H)one Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful weighing and charging of the catalytic system, specifically palladium acetate and the chiral phosphine ligand, along with the solid CO source Mo(CO)6. The stoichiometry is critical, with the patent recommending a molar ratio of benzisoxazole to benzyl chloride of approximately 1:2.5 to ensure complete consumption of the limiting reagent. The reaction is conducted in DME solvent at a concentration that allows for effective mixing, typically around 0.5 mmol of substrate per 1-3 mL of solvent in lab scale, which scales linearly to larger volumes. The addition of water (1.0 equivalent) and triethylamine (6.0 equivalents) is essential for driving the reaction to completion and neutralizing the HCl byproduct generated from the benzyl chloride. Detailed standardized synthesis steps see the guide below.
- Charge a sealed reactor with palladium acetate, (S)-BINAP ligand, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride substrate.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture to ensure homogeneous suspension of all catalytic components and reactants.
- Heat the reaction mixture to 100°C and maintain this temperature for approximately 26 hours to allow complete conversion before proceeding to filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the raw material supply chain. Benzisoxazoles and substituted benzyl chlorides are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency and ensuring supply continuity. Unlike traditional methods that might require custom-synthesized anilines or unstable acid chlorides, this route utilizes stable, shelf-stable starting materials that can be stocked in bulk without special storage conditions. This stability translates directly into inventory management efficiencies and reduces the working capital tied up in specialized precursors. Furthermore, the elimination of gaseous carbon monoxide removes the need for complex gas handling infrastructure, lowering the barrier to entry for contract manufacturing organizations (CMOs) and enabling more flexible production scheduling.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the use of inexpensive, commercially available reagents. By avoiding the use of expensive coupling reagents or toxic halogenating agents like POCl3, the direct material costs are substantially lowered. Additionally, the simplified workup procedure, which involves basic filtration and standard column chromatography, reduces the consumption of solvents and silica gel compared to multi-step purification sequences required by older methods. The high yields reported (often exceeding 90% for optimized substrates) mean less waste generation and higher throughput per batch, effectively lowering the cost per kilogram of the final API intermediate. The use of a solid CO source also mitigates the costs associated with high-pressure reactor maintenance and safety compliance audits.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable and reliable supply chain. The tolerance for a wide range of functional groups means that the same core process can be adapted for multiple derivatives without extensive re-optimization, allowing for agile responses to changing market demands. Since the starting materials are common industrial chemicals, lead times for raw material procurement are minimized, preventing production bottlenecks. The scalability of the reaction from milligram to kilogram scale has been demonstrated with consistent results, assuring supply chain heads that the process can meet commercial volume requirements without the teething problems often associated with scaling novel chemistries. This reliability is crucial for maintaining just-in-time delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a greener alternative to traditional quinolinone synthesis. The avoidance of highly toxic gases and corrosive liquids reduces the facility's environmental footprint and simplifies waste disposal protocols. The reaction produces fewer hazardous byproducts, aligning with increasingly stringent global regulations on chemical manufacturing emissions. The ability to run the reaction at atmospheric pressure (in a sealed tube or standard reactor) rather than high pressure reduces energy consumption and equipment stress. These factors collectively enhance the sustainability profile of the manufacturing process, which is becoming a key differentiator in vendor selection for major pharmaceutical companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing production workflows. The responses cover aspects of safety, substrate compatibility, and purification strategies to ensure a comprehensive understanding of the technology's value proposition.
Q: What are the safety advantages of using Mo(CO)6 over gaseous CO in this synthesis?
A: Using molybdenum hexacarbonyl (Mo(CO)6) as a solid carbon monoxide source eliminates the need for high-pressure CO gas cylinders, significantly reducing safety risks associated with toxic gas handling and allowing for simpler reactor setups in standard laboratory or pilot plant environments.
Q: How does the benzisoxazole substrate function in this reaction mechanism?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (carbonyl) source. The cleavage of the N-O bond under palladium catalysis releases the necessary atoms to construct the quinolinone core, streamlining the atom economy of the process.
Q: Is this method compatible with sensitive functional groups on the aromatic rings?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substituents such as cyano, trifluoromethyl, methoxy, and halogens without significant degradation or side reactions, making it suitable for diverse medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and optimizing production costs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. Our commitment to quality assurance means that every batch of 3-arylquinolin-2(1H)one derivatives produced adheres to the highest international standards, providing our partners with the confidence needed to advance their clinical programs. We leverage our deep expertise in palladium catalysis and heterocyclic chemistry to troubleshoot and optimize processes, guaranteeing consistent supply and superior product quality.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this method for your specific target molecules. We encourage you to contact us to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments. Our goal is to act as a strategic extension of your R&D and supply chain operations, delivering not just chemicals, but solutions that drive your business forward in the competitive landscape of pharmaceutical manufacturing.
