Advanced Green Synthesis of Fasudil Hydrochloride for Commercial Scale Production
Advanced Green Synthesis of Fasudil Hydrochloride for Commercial Scale Production
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical vasoactive agents. A significant breakthrough in this domain is detailed in patent CN111303120A, which discloses a novel preparation method for Fasudil Hydrochloride, a potent Rho kinase inhibitor used extensively in the treatment of cerebral vasospasm and ischemia. Unlike conventional methodologies that rely on hazardous chlorinating agents and unstable intermediates, this innovative process utilizes an active ester strategy to achieve exceptional purity levels exceeding 99.5%. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediates supplier partnerships, this technology represents a paradigm shift towards greener chemistry. By fundamentally altering the activation step of the sulfonic acid moiety, the process eliminates the generation of corrosive waste gases and problematic pigment impurities, thereby offering a compelling value proposition for cost reduction in API manufacturing while ensuring consistent supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Fasudil Hydrochloride has been plagued by significant technical and environmental hurdles associated with the use of sulfonyl chloride intermediates. Traditional routes, such as those described in US4678783 and various Chinese patents, typically involve the sulfonation of isoquinoline followed by chlorination using reagents like thionyl chloride, oxalyl chloride, or chlorosulfonic acid. These chlorinating agents are notoriously aggressive, causing severe corrosion to reaction equipment and generating substantial quantities of acidic wastewater and sulfur dioxide gas, which imposes heavy burdens on environmental compliance and waste treatment infrastructure. Furthermore, the resulting isoquinoline-5-sulfonyl chloride hydrochloride is chemically unstable and prone to hydrolysis back to the sulfonic acid, a side reaction that introduces difficult-to-remove yellow-green pigment impurities into the reaction matrix. To mitigate these color issues, manufacturers are often forced to employ costly and labor-intensive purification steps involving activated carbon, silica gel, or resin adsorption, which drastically reduces overall yield and increases the cost of goods sold.
The Novel Approach
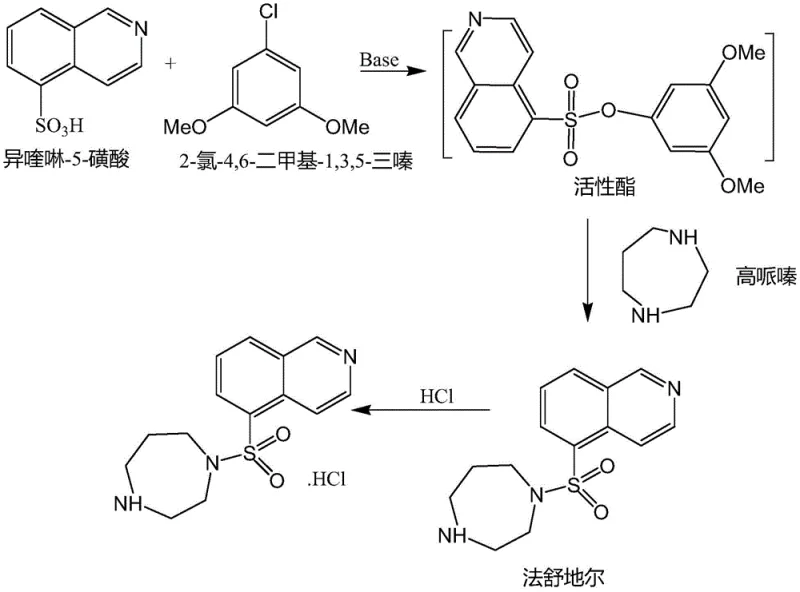
In stark contrast to the legacy chlorination pathways, the method disclosed in CN111303120A introduces a sophisticated active ester coupling strategy that circumvents the formation of the unstable acyl chloride entirely. By reacting isoquinoline-5-sulfonic acid directly with 2-chloro-4,6-dimethoxy-1,3,5-triazine in the presence of an organic base, the process generates a highly reactive yet stable sulfonic acid active ester in situ. This intermediate then undergoes nucleophilic substitution with homopiperazine under mild conditions to form the Fasudil backbone. This strategic pivot not only eliminates the safety risks associated with handling large volumes of thionyl chloride but also inherently prevents the hydrolysis-induced pigment formation that plagues traditional methods. Consequently, the crude product obtained is of significantly higher quality, requiring only simple washing and recrystallization to achieve pharmaceutical-grade purity, thus streamlining the entire production workflow and enhancing the economic viability of commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Active Ester-Mediated Sulfonamide Formation
The core chemical innovation of this process lies in the activation of the sulfonic acid group using a triazine derivative, which serves as a superior leaving group compared to the chloride ion in traditional sulfonyl chlorides. In the initial step, isoquinoline-5-sulfonic acid reacts with 2-chloro-4,6-dimethoxy-1,3,5-triazine in a solvent such as dichloromethane or chloroform, facilitated by an organic base like triethylamine or N-methylmorpholine. The base deprotonates the sulfonic acid, generating a sulfonate anion that attacks the electron-deficient carbon of the triazine ring, displacing a chloride ion and forming the sulfonic acid active ester. This activation occurs efficiently at temperatures between 10-30°C, preventing thermal degradation of sensitive functional groups. The resulting active ester is sufficiently electrophilic to react with the secondary amine of homopiperazine but lacks the extreme reactivity of a sulfonyl chloride, which minimizes non-specific side reactions and hydrolysis during the subsequent coupling phase.
Following the formation of the active ester, homopiperazine is introduced to the reaction mixture to effect the sulfonamide bond formation. The nucleophilic nitrogen of the homopiperazine attacks the sulfur center of the activated ester, displacing the triazine byproduct and yielding the free base of Fasudil. A critical advantage of this mechanism is the suppression of dimer impurities, which are a common byproduct in acid chloride reactions where excess amine is typically required to scavenge HCl. In this active ester system, the stoichiometry can be tightly controlled (molar ratio of 1:1 to 1:1.5), and the absence of strong acid byproducts prevents the protonation of the amine nucleophile, ensuring high conversion rates. As illustrated in the reaction scheme below, the pathway proceeds cleanly to the final product, which is subsequently converted to the hydrochloride salt through pH adjustment, yielding a white crystalline solid with single impurity content below 0.1%.
How to Synthesize Fasudil Hydrochloride Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity, particularly during the activation and coupling stages. The process begins with the dispersion of isoquinoline-5-sulfonic acid and the triazine activator in a halogenated solvent, followed by the controlled addition of base to manage exotherms. Once the active ester is formed, the homopiperazine solution is added dropwise to maintain reaction homogeneity and prevent local concentration spikes that could lead to byproducts. The subsequent workup involves a sequential washing protocol to remove organic bases and triazine byproducts, followed by salting out the product with hydrochloric acid. For a detailed breakdown of the specific operational parameters, solvent ratios, and temperature profiles required to replicate this high-efficiency process, please refer to the standardized synthesis guide provided below.
- Activate isoquinoline-5-sulfonic acid with 2-chloro-4,6-dimethoxy-1,3,5-triazine and organic base in dichloromethane at 10-30°C to form the active ester.
- React the active ester solution with homopiperazine at 10-30°C, followed by heating to reflux to complete the coupling reaction.
- Perform aqueous workup including water wash, alkali wash, and acidification with HCl to precipitate the crude product, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this active ester-based synthesis offers profound advantages that extend beyond mere chemical elegance, directly impacting the bottom line and supply chain resilience. Traditional methods relying on sulfonyl chlorides introduce significant volatility into the supply chain due to the instability of the intermediate and the regulatory scrutiny surrounding chlorinating reagents. By transitioning to this greener alternative, manufacturers can secure a more reliable supply of high-purity Fasudil Hydrochloride while mitigating the risks associated with hazardous material transport and storage. The elimination of corrosive gases like SO2 and HCl reduces the maintenance frequency of reactor vessels and scrubbing systems, leading to lower capital expenditure on equipment and reduced downtime for repairs. Furthermore, the simplified purification process, which avoids column chromatography, significantly shortens the batch cycle time, allowing for increased throughput and faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and hazardous chlorinating reagents such as thionyl chloride and oxalyl chloride, which require specialized handling and disposal protocols. By replacing these with 2-chloro-4,6-dimethoxy-1,3,5-triazine, the process reduces the cost of raw materials and significantly lowers the expense associated with waste acid neutralization and treatment. Additionally, the high selectivity of the reaction minimizes the loss of valuable homopiperazine, a relatively costly starting material, as there is no need to use a large excess to drive the reaction or scavenge acid. The avoidance of pigment impurities also removes the need for expensive adsorbents like silica gel or specialized resins, further driving down the variable costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the regulatory restrictions placed on the transport and storage of corrosive chlorinating agents. This novel method utilizes stable solid reagents and common organic solvents, simplifying logistics and reducing the risk of shipment delays due to hazardous material compliance issues. The stability of the active ester intermediate allows for greater flexibility in batch scheduling, as the reaction is less sensitive to minor variations in moisture or temperature compared to the hydrolysis-prone sulfonyl chloride route. This robustness ensures consistent batch-to-batch quality, reducing the rate of rejected lots and ensuring that downstream formulation teams receive material that consistently meets stringent specifications without unexpected delays.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, but this route is inherently designed for green manufacturing. The absence of sulfur dioxide emissions and acidic wastewater aligns perfectly with increasingly strict global environmental regulations, future-proofing the production facility against tightening compliance standards. The workup procedure relies on simple liquid-liquid extraction and crystallization, unit operations that are easily scaled from pilot plants to multi-ton reactors without the need for complex preparative HPLC or chromatography columns. This scalability ensures that the process remains economically viable at large volumes, supporting the growing global demand for Fasudil in cardiovascular and cerebrovascular therapies while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of how this method outperforms legacy processes. Understanding these distinctions is crucial for technical teams evaluating process transfer opportunities and for procurement officers assessing the long-term viability of their supply sources.
Q: Why does this new method avoid the pigment issues found in traditional Fasudil synthesis?
A: Traditional methods utilize unstable isoquinoline-5-sulfonyl chloride, which easily hydrolyzes and generates yellow-green pigment impurities. This novel route bypasses the acyl chloride intermediate entirely by using a stable active ester, completely eliminating pigment formation and simplifying purification.
Q: How does the active ester approach improve impurity profiles compared to sulfonyl chloride routes?
A: The reaction of sulfonyl chloride with homopiperazine often leads to difficult-to-remove dimer impurities. The active ester mechanism described in CN111303120A selectively favors the mono-substituted product, effectively suppressing dimer formation and achieving single impurity levels below 0.1% without column chromatography.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It operates under mild conditions (10-60°C), uses common solvents like dichloromethane, and avoids hazardous chlorinating reagents like thionyl chloride. The workup involves simple phase separation and crystallization, making it ideal for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fasudil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the active ester method are fully realized in a GMP-compliant environment. Our facilities are equipped with state-of-the-art corrosion-resistant reactors and rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of Fasudil Hydrochloride meets stringent purity specifications required by global regulatory agencies. We are committed to delivering not just a chemical product, but a comprehensive supply solution that integrates process optimization with reliable logistics.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their development pipelines. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green synthesis route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data from our recent batches and to discuss route feasibility assessments for your upcoming projects, ensuring a seamless integration of high-quality intermediates into your manufacturing operations.
