Advanced Manufacturing of High-Purity Fasudil Hydrochloride for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Fasudil Hydrochloride for Global Pharmaceutical Supply Chains
The global demand for high-quality cardiovascular therapeutics continues to drive innovation in the synthesis of key active pharmaceutical ingredients (APIs) and their intermediates. Among these, Fasudil Hydrochloride stands out as a critical Rho-kinase inhibitor used extensively for the treatment of cerebral vasospasm and ischemia. However, traditional manufacturing routes have long struggled with significant challenges, including low yields, hazardous reaction conditions, and the persistent formation of difficult-to-remove dimer impurities. A groundbreaking technical solution, detailed in patent CN103724326A, offers a transformative approach to overcoming these bottlenecks. This novel preparation method leverages optimized stoichiometry and advanced crystallization techniques to achieve chromatographic purity exceeding 99.9% while virtually eliminating dimer contaminants. For international procurement teams and R&D directors, this represents a pivotal shift towards more sustainable, safe, and cost-effective production capabilities.
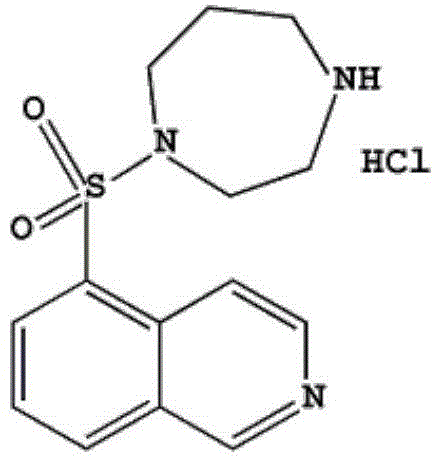
At the core of this technological advancement is a fundamental re-engineering of the sulfonylation reaction between isoquinoline-5-sulfonyl chloride and homopiperazine. Unlike conventional processes that rely on excessive reagents and complex purification steps like column chromatography, this method introduces a precise control mechanism over the reaction environment. By utilizing anhydrous sodium sulfate and solid sodium bicarbonate within the dichloromethane phase, the process effectively neutralizes hydrochloric acid byproducts in situ while preventing the hydrolysis of the sensitive sulfonyl chloride intermediate. This meticulous attention to moisture control and pH regulation ensures that the reaction proceeds with high selectivity, minimizing side reactions that typically degrade product quality. The result is a robust synthetic pathway that aligns perfectly with the stringent quality requirements of modern regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Fasudil Hydrochloride has been plagued by inherent inefficiencies that compromise both economic viability and operational safety. Traditional protocols, such as those referenced in earlier patents like US4678783, typically employ a massive excess of homopiperazine—often in a molar ratio of up to 4:1 relative to the sulfonyl chloride—to suppress the formation of dimer impurities. While this brute-force approach attempts to drive the reaction to completion, it creates a cascade of downstream problems. Firstly, the unreacted homopiperazine is difficult to separate completely, often requiring multiple acid-base extraction cycles that generate substantial volumes of organic waste. Secondly, the use of dichloromethane to dissolve solid homopiperazine presents a severe safety hazard; the dissolution process can be violently exothermic, leading to potential thermal runaways or even explosion risks in large-scale reactors. Furthermore, the reliance on column chromatography for final purification, as seen in some prior art, is economically unsustainable for commercial scale-up due to high solvent consumption and low throughput.
The Novel Approach
The methodology disclosed in patent CN103724326A fundamentally disrupts these legacy constraints by introducing a smarter, chemistry-driven solution rather than a procedural workaround. The most striking innovation is the drastic reduction in the homopiperazine feed ratio to a mere 1:1.5 to 1:2. This optimization is made possible by the introduction of a unique workup procedure where the reaction mixture is adjusted to a specific pH range of 5.0 to 6.5. At this precise acidity, unreacted homopiperazine precipitates out of the solution as a salt, allowing for its physical removal via filtration and subsequent recovery. This not only purifies the product stream but also allows for the recycling of valuable starting materials, creating a closed-loop efficiency that traditional methods lack. Additionally, the switch to using absolute ethanol for dissolving homopiperazine eliminates the explosion risks associated with dichloromethane, significantly enhancing the safety profile of the manufacturing process. The elimination of column chromatography in favor of repeated vacuum concentration and crystallization further streamlines the operation, making it highly amenable to multi-ton production.
Mechanistic Insights into Sulfonylation and Impurity Control
To fully appreciate the technical superiority of this synthesis, one must examine the mechanistic nuances of the sulfonylation reaction and the subsequent purification logic. The reaction involves the nucleophilic attack of the secondary amine in homopiperazine on the sulfur atom of the isoquinoline-5-sulfonyl chloride. In traditional settings, the presence of excess base or improper solvent conditions can facilitate a second nucleophilic attack by another molecule of homopiperazine or the product itself, leading to the formation of sulfonamide dimers. These dimers are structurally similar to the target molecule, making them notoriously difficult to separate via standard recrystallization. The patented method mitigates this by strictly controlling the reaction temperature at ≤10°C and utilizing a biphasic system where anhydrous sodium sulfate acts as a desiccant. This ensures that the highly reactive sulfonyl chloride does not hydrolyze into the corresponding sulfonic acid, a common side reaction that reduces yield and complicates purification. The presence of solid sodium bicarbonate serves to scavenge the HCl generated during the reaction without introducing water, maintaining an anhydrous environment crucial for high conversion rates.
Furthermore, the impurity control mechanism extends beyond the reaction vessel into the workup phase. The adjustment of the pH to 5.0–6.5 is a critical step that exploits the pKa differences between the product, the starting material, and the impurities. Under these mildly acidic conditions, the free base form of homopiperazine is protonated and precipitates, while the Fasudil Hydrochloride remains in solution or is managed in a way that prevents co-precipitation of the dimer. This selective precipitation is a masterstroke of process chemistry, effectively acting as a "chemical filter" that removes the primary source of contamination before the final crystallization. The subsequent refining step involves the use of activated carbon (0.1–2g/ml) in anhydrous ethanol, which adsorbs trace colored impurities and residual organics. This combination of pH-controlled precipitation and adsorption purification ensures that the final product achieves a dimer impurity content of 0% and a total purity of over 99.9%, surpassing the capabilities of older chromatographic methods.
How to Synthesize Fasudil Hydrochloride Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and solvent ratios to ensure reproducibility and safety. The process begins with the preparation of the sulfonyl chloride solution under anhydrous conditions, followed by the controlled addition to the homopiperazine ethanol solution. The key to success lies in the precise management of the exotherm during the mixing phase and the accurate pH adjustment during the filtration step. Operators must ensure that the reaction temperature never exceeds 10°C to prevent thermal degradation and dimerization. Following the reaction, the filtrate undergoes a two-stage concentration process under reduced pressure to isolate the crude product, which is then subjected to the final decolorization and crystallization steps. For a detailed, step-by-step breakdown of the exact quantities, stirring times, and drying temperatures required for GMP-compliant production, please refer to the standardized protocol below.
- Prepare isoquinoline-5-sulfonyl chloride solution in dichloromethane with anhydrous sodium sulfate and solid sodium bicarbonate to prevent hydrolysis.
- Dissolve homopiperazine in absolute ethanol and cool to ≤10°C, then slowly add the sulfonyl chloride solution while maintaining low temperature.
- Adjust pH to 5.0-6.5 to precipitate unreacted homopiperazine for recovery, followed by concentration, crystallization, and activated carbon decolorization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates into tangible strategic advantages that extend far beyond simple chemical yield. The most immediate impact is seen in the drastic reduction of raw material costs. By lowering the molar ratio of homopiperazine from the traditional excess of 4 equivalents down to 1.5 equivalents, manufacturers can achieve substantial savings on one of the most expensive reagents in the process. This efficiency is compounded by the ability to recover and recycle the precipitated homopiperazine, further driving down the effective cost per kilogram of the final API. Moreover, the elimination of column chromatography removes a significant bottleneck in production throughput. Chromatography is not only solvent-intensive and slow but also difficult to scale; replacing it with crystallization and filtration allows for continuous or large-batch processing, significantly increasing plant capacity without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the optimization of reagent stoichiometry and the simplification of unit operations. Traditional methods incur high costs due to the wasteful use of homopiperazine and the extensive solvent consumption required for chromatographic purification. By contrast, this new route minimizes reagent usage and replaces complex separation techniques with efficient crystallization. The recovery of unreacted homopiperazine adds an additional layer of cost efficiency, ensuring that raw material utilization is maximized. Consequently, the overall production cost is significantly lowered, providing a competitive pricing advantage in the global market for cardiovascular intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on process safety and scalability. The replacement of dichloromethane with absolute ethanol for dissolving homopiperazine mitigates the risk of thermal runaway incidents, ensuring uninterrupted production schedules. Safety incidents are a major cause of supply disruption in the fine chemical industry; by adopting a safer solvent system, manufacturers can maintain consistent output levels. Additionally, the simplified workflow reduces the dependency on specialized chromatography columns and resins, which can sometimes face supply shortages. The robustness of the crystallization-based purification ensures that the process can be reliably scaled from pilot plants to multi-ton commercial reactors without losing efficiency.
- Scalability and Environmental Compliance: Environmental regulations are becoming increasingly stringent, particularly regarding the discharge of organic solvents and chemical waste. This synthesis method offers a greener alternative by significantly reducing the volume of organic liquid waste generated. The removal of the chromatography step alone eliminates a major source of solvent effluent. Furthermore, the ability to recycle homopiperazine reduces the chemical load entering the waste stream. The process is designed to be environmentally compliant, utilizing standard filtration and distillation equipment that meets modern EHS (Environment, Health, and Safety) standards. This makes the technology highly attractive for manufacturers looking to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy processes. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or for procurement specialists assessing the quality consistency of potential suppliers.
Q: How does the new method eliminate dimer impurities in Fasudil Hydrochloride?
A: The method optimizes the molar ratio of reactants to 1:1.5~2 and utilizes precise pH control (5.0-6.5) during workup. This prevents the excessive presence of free homopiperazine that typically drives dimerization, resulting in 0% detectable dimer impurity.
Q: Why is absolute ethanol used instead of dichloromethane for dissolving homopiperazine?
A: Using absolute ethanol significantly enhances process safety. Dissolving solid homopiperazine directly in dichloromethane can lead to violent exothermic reactions and potential explosion hazards, whereas ethanol provides a safer, more controlled solvent environment.
Q: What represents the major cost advantage of this synthesis route?
A: The primary cost driver is the reduction in homopiperazine usage. Traditional methods require a large excess (up to 4 equivalents) to suppress impurities, whereas this patented method achieves high purity with only 1.5 to 2 equivalents, drastically lowering raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fasudil Hydrochloride Supplier
As the pharmaceutical industry continues to demand higher purity standards and more sustainable manufacturing practices, partnering with a technically proficient CDMO is essential. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in patent CN103724326A to deliver superior intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate under stringent purity specifications and utilize rigorous QC labs to verify that every batch of Fasudil Hydrochloride meets the >99.9% purity benchmark with zero detectable dimer impurities, providing you with a reliable foundation for your final drug product.
We invite you to explore how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how our process improvements translate to bottom-line value. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring high-quality, cost-effective cardiovascular therapies to the market faster and more efficiently.
