Revolutionizing Deuterated Aromatic Synthesis for High-Performance OLED and Pharmaceutical Manufacturing
The rapid advancement of organic light-emitting diode (OLED) technology and the increasing demand for metabolically stable pharmaceutical agents have placed deuterated aromatic compounds at the forefront of modern chemical synthesis. Patent CN114853557A introduces a groundbreaking preparation method that addresses the critical bottlenecks of cost and purity in producing these high-value intermediates. By leveraging polyfluoric anhydrides as potent catalysts in conjunction with accessible deuterium sources like heavy water, this technology enables the efficient conversion of standard aromatic hydrocarbons into their fully deuterated counterparts. This innovation is particularly significant for the manufacturing of long-life OLED materials, where the replacement of carbon-hydrogen bonds with stronger carbon-deuterium bonds drastically improves device longevity and luminous efficiency. For global supply chains, this represents a shift towards more sustainable and economically viable production routes for complex deuterated aromatics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated aromatic compounds has been plagued by prohibitive costs and operational complexities that hinder large-scale adoption. Traditional routes often rely on multi-step syntheses starting from expensive perdeuterated benzene, requiring intricate functionalization strategies that result in poor atom economy and significant waste generation. Alternatively, transition metal-catalyzed hydrogen-deuterium exchange reactions, while effective, frequently utilize precious metals such as palladium, platinum, or iridium, which not only inflate raw material costs but also introduce the risk of toxic metal residues that are unacceptable in high-purity electronic applications. Furthermore, conventional acid-catalyzed methods often suffer from the inherent acidity of the catalyst itself, which introduces protic hydrogen into the reaction system, thereby diluting the isotopic abundance of the final product and necessitating costly purification steps to achieve the required deuterium levels.
The Novel Approach
The methodology disclosed in the patent data offers a transformative solution by employing polyfluoric anhydrides, such as trifluoromethanesulfonic anhydride, as superacidic catalysts that activate deuterium sources without introducing competing protons. This approach allows for the direct use of heavy water, a significantly cheaper and more abundant deuterium source compared to deuterated organic solvents or gases. The reaction proceeds under inert atmosphere protection in common organic solvents, facilitating a one-pot process that eliminates the need for intermediate solvent exchanges or complex workup procedures. By operating at elevated temperatures between 100-250°C, the system ensures high conversion rates and exceptional deuterium incorporation, often exceeding 99% after optimization, while maintaining a green profile through the avoidance of heavy metal contaminants and the use of recyclable solvent systems.
Mechanistic Insights into Polyfluoric Anhydride-Catalyzed Deuteration
The core of this technological breakthrough lies in the unique ability of polyfluoric anhydrides to generate superacidic conditions that facilitate electrophilic aromatic substitution with deuterium ions. In this mechanism, the polyfluoric anhydride reacts with the deuterium source, typically heavy water or deuterated alcohols, to generate a highly reactive deuterating species in situ. This species acts as a potent electrophile, attacking the electron-rich aromatic ring of substrates such as anthracene, carbazole, or naphthalene. The absence of exchangeable protons in the fluorinated anhydride structure is crucial, as it prevents the back-exchange of deuterium with hydrogen, a common failure mode in traditional acid catalysis. This ensures that the equilibrium of the reaction strongly favors the formation of the carbon-deuterium bond, leading to the high isotopic purity observed in the experimental data.
Furthermore, the choice of solvent plays a pivotal role in the success of this reaction, particularly for substrates prone to sublimation or poor solubility at high temperatures. The patent specifies the use of C5-C10 alkanes or substituted aromatic hydrocarbons, which provide an optimal balance of solvating power and thermal stability. These solvents effectively dissolve the aromatic substrates and the catalyst system while preventing the loss of volatile components during the extended heating periods required for complete deuteration. The robustness of this mechanistic pathway allows for the deuteration of a wide range of polycyclic aromatic hydrocarbons, including those with heteroatoms like nitrogen or oxygen, without compromising the integrity of sensitive functional groups, thereby offering a versatile platform for synthesizing diverse deuterated building blocks.
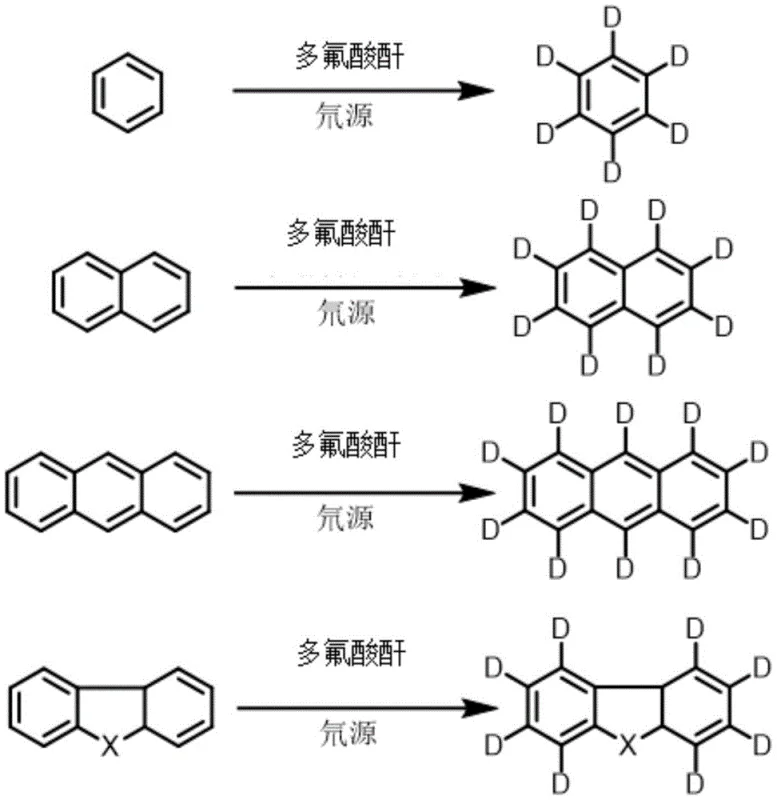
How to Synthesize Deuterated Aromatic Compounds Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing chemical manufacturing infrastructure, requiring only standard reactor setups capable of handling elevated temperatures and inert atmospheres. The process begins with the precise charging of the aromatic substrate, the selected polyfluoric anhydride catalyst, and the deuterium source into a reactor containing the appropriate organic solvent. Following the reaction period, which typically spans 15 to 24 hours depending on the substrate reactivity, the mixture undergoes a straightforward separation process to isolate the crude deuterated product. Detailed standardized synthetic steps, including specific molar ratios and temperature profiles for various substrates, are outlined in the technical guide below to ensure reproducibility and optimal yield.
- Charge the reactor with the aromatic substrate, a C5-C10 alkane or substituted aromatic solvent, polyfluoric anhydride catalyst, and heavy water under inert gas protection.
- Heat the reaction mixture to temperatures between 100-250°C and maintain stirring for 15-24 hours to ensure complete hydrogen-deuterium exchange.
- Upon completion, separate the organic phase, wash to remove acid residues, and purify via crystallization or chromatography to obtain the final deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this polyfluoric anhydride-catalyzed method presents a compelling value proposition centered on cost efficiency and supply security. By shifting away from noble metal catalysts and exotic deuterated reagents, manufacturers can significantly reduce the raw material expenditure associated with producing high-purity deuterated intermediates. The simplification of the workflow into a one-pot reaction reduces labor hours and energy consumption, while the elimination of heavy metal removal steps streamlines the downstream purification process, leading to faster batch turnover times and improved overall equipment effectiveness.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with commercially available polyfluoric anhydrides results in substantial savings on catalyst procurement costs. Additionally, the ability to utilize heavy water as the primary deuterium source, rather than costly deuterated organic solvents or gases, drastically lowers the variable cost per kilogram of the final product. The high yields and purity achieved minimize the need for reprocessing or recycling off-spec material, further enhancing the economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as trifluoroacetic anhydride and heavy water ensures a stable and resilient supply chain, free from the geopolitical risks often associated with precious metal sourcing. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in feedstock quality compared to sensitive metal-catalyzed systems. This reliability translates to consistent delivery performance for downstream customers in the OLED and pharmaceutical sectors who depend on uninterrupted supplies of critical deuterated building blocks.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies waste treatment protocols, reducing the environmental footprint and regulatory burden associated with hazardous waste disposal. The process is inherently scalable, as the exothermic nature of the reaction can be managed effectively in larger reactors through controlled addition and cooling strategies. This scalability supports the transition from pilot-scale development to multi-ton annual production capacities, meeting the growing global demand for deuterated materials without the need for specialized high-pressure equipment or complex containment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterated aromatic compounds using this novel methodology. These insights are derived directly from the patent specifications and experimental data to provide clarity on process capabilities and product quality standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating these materials into their own formulation or synthesis pipelines.
Q: What are the advantages of using polyfluoric anhydride over transition metal catalysts for deuteration?
A: Polyfluoric anhydrides avoid the use of expensive noble metals like Palladium or Platinum, significantly reducing raw material costs and eliminating the risk of heavy metal contamination in the final electronic or pharmaceutical grade product.
Q: Can this method achieve high deuterium incorporation rates?
A: Yes, the method utilizes heavy water directly with superacidic activation, achieving deuterium abundances up to 99% after iterative processing, which is critical for enhancing the stability of OLED materials.
Q: Is this process scalable for industrial production of deuterated intermediates?
A: The process is designed as a simple one-pot reaction without the need for intermediate solvent exchanges, making it highly suitable for large-scale commercial manufacturing with simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Aromatic Compounds Supplier
As the global demand for high-performance electronic materials and stable pharmaceutical intermediates continues to surge, securing a partner with deep technical expertise in isotope chemistry is paramount. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver deuterated aromatic compounds that meet the most stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify deuterium incorporation rates and impurity profiles, ensuring that every batch delivered supports the high reliability requirements of OLED display manufacturing and drug development programs.
We invite industry leaders to collaborate with us to optimize their supply chains and reduce time-to-market for next-generation products. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can enhance your product performance while driving down overall manufacturing costs.
