Revolutionizing Chloramphenicol Production: A High-Efficiency Nitromethane-Based Synthetic Route
Revolutionizing Chloramphenicol Production: A High-Efficiency Nitromethane-Based Synthetic Route
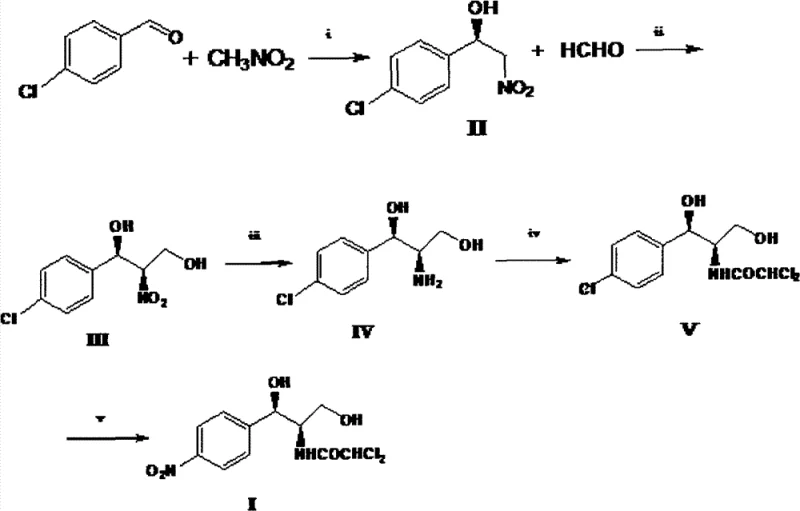
The pharmaceutical industry constantly seeks more efficient pathways for producing broad-spectrum antibiotics, and Patent CN102399165B presents a transformative methodology for synthesizing Chloramphenicol, also known as Paraxin. This intellectual property details a novel five-step synthetic route that fundamentally shifts the starting materials from traditional ethylbenzene to a more atom-economical combination of p-chlorobenzaldehyde and nitromethane. By leveraging advanced asymmetric catalysis, this method circumvents the inherent inefficiencies of classical resolution techniques, offering a pathway that is not only chemically elegant but also commercially superior. For R&D directors and procurement strategists, understanding this shift is critical, as it represents a move towards greener chemistry with significantly reduced operational complexity. The patent underscores the viability of using nitromethane as a C1 building block to construct the propanediol backbone with high stereochemical fidelity, setting a new benchmark for antibiotic intermediate manufacturing.
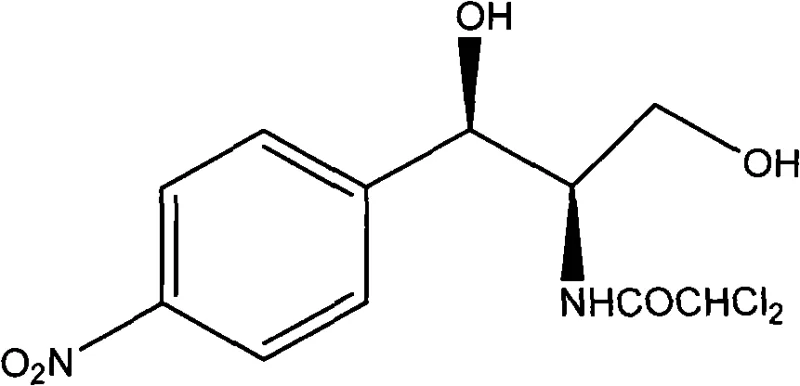
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Chloramphenicol has relied heavily on a multi-step sequence originating from ethylbenzene, a process fraught with inefficiencies and environmental burdens. As illustrated in the traditional workflow, the synthesis requires a cumbersome series of transformations including nitration, oxidation, bromination, ammonification, and acetylation before even reaching the critical aldol condensation stage. The most significant bottleneck in this legacy approach is the reliance on chiral resolution to separate enantiomers, a step that theoretically caps the maximum yield at 50% regardless of optimization efforts. Furthermore, the conventional route necessitates the use of aluminum isopropoxide for the reduction of the ketone to the alcohol, a reagent that generates substantial quantities of aluminum-containing waste that is difficult and costly to treat. These factors combine to create a process with a low overall yield, typically around 30% in domestic production settings, driving up the cost of goods sold and complicating supply chain logistics due to the sheer number of unit operations required.
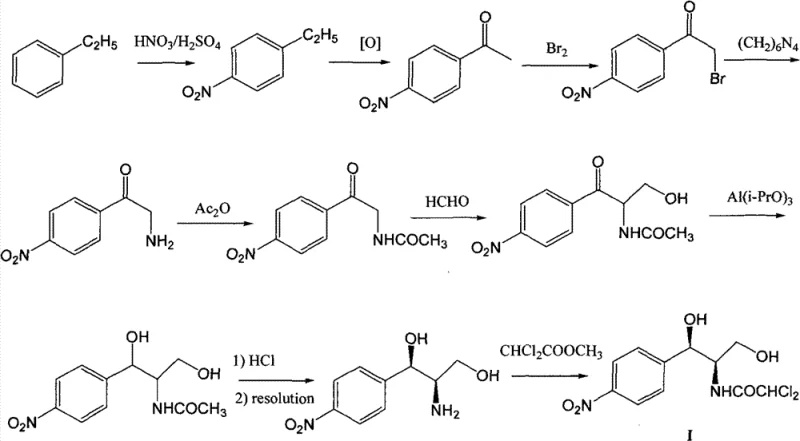
The Novel Approach
In stark contrast to the convoluted ethylbenzene pathway, the methodology disclosed in CN102399165B introduces a streamlined, five-step sequence that begins with the direct condensation of p-chlorobenzaldehyde and nitromethane. This innovative strategy employs an asymmetric Henry reaction to establish the crucial chiral center at the very beginning of the synthesis, thereby completely eliminating the need for the yield-limiting chiral resolution step found in traditional methods. By bypassing the resolution bottleneck, the theoretical yield ceiling is removed, allowing for a much more efficient utilization of raw materials. Additionally, the replacement of the aluminum isopropoxide reduction with catalytic hydrogenation represents a major green chemistry advancement, significantly reducing the generation of hazardous three wastes. The resulting process is not only shorter and cleaner but also utilizes commodity chemicals that are inexpensive and readily available, making it an ideal candidate for cost-effective, large-scale manufacturing of high-purity Chloramphenicol intermediates.
Mechanistic Insights into Asymmetric Henry Reaction and Pd-Catalyzed Substitution
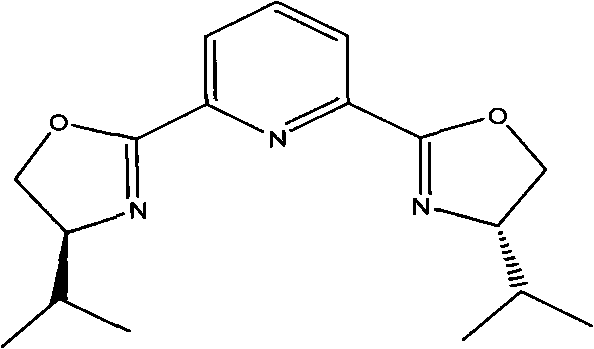
The cornerstone of this novel synthetic route is the initial asymmetric Henry reaction, which dictates the stereochemical outcome of the entire sequence. This transformation utilizes a sophisticated chiral catalyst system comprising a Lewis acid, such as copper triflate, coordinated with a chiral ligand like 2,6-bis[(S)-4-isopropyl-1-phenyl-4,5-dihydro-1H-2-imidazolyl]pyridine. The precise geometry of this ligand, as depicted in the structural diagrams, creates a chiral environment that favors the formation of the (R)-enantiomer of 2-nitro-1-(4-chlorophenyl)ethanol with exceptional enantiomeric excess, reported to be as high as 96.5% in experimental embodiments. This high level of stereocontrol is paramount for pharmaceutical applications, ensuring that the downstream intermediates maintain the requisite optical purity without the need for corrective purification steps. The ability to install chirality with such fidelity in the first step effectively locks in the quality of the final product, reducing the risk of impurity carryover and simplifying the overall quality control protocol for the manufacturing site.
Following the construction of the carbon backbone and the reduction of the nitro group to an amine, the synthesis concludes with a highly specialized palladium-catalyzed nitro substitution to install the final dichloroacetamide moiety. This step utilizes a palladium source, such as tris(dibenzylideneacetone)dipalladium, in conjunction with bulky phosphine ligands like t-BuXPhos to facilitate the coupling reaction under relatively mild conditions. The choice of ligand is critical here, as it modulates the electronic and steric properties of the palladium center, enabling the selective substitution of the hydroxyl group while preserving the sensitive dichloroacetamide functionality. This mechanistic nuance ensures that the final Chloramphenicol product is obtained with high chemical purity, exceeding 98.0% after simple recrystallization. For process chemists, this indicates a robust reaction profile that tolerates the functional groups present in the molecule, minimizing the formation of side products and maximizing the isolated yield of the active pharmaceutical ingredient.
How to Synthesize Chloramphenicol Efficiently
The implementation of this synthetic strategy requires careful attention to reaction conditions, particularly during the asymmetric catalysis and hydrogenation stages, to ensure optimal yield and purity. The patent provides detailed experimental embodiments that serve as a robust foundation for scaling this chemistry from the laboratory to pilot and commercial production scales. Operators must maintain strict control over temperature and stoichiometry, especially during the initial Henry reaction where the ratio of nitromethane to aldehyde and the loading of the chiral catalyst play pivotal roles in determining enantioselectivity. The subsequent steps, including the formaldehyde condensation and catalytic hydrogenation, are designed to be operationally simple, utilizing standard equipment found in most fine chemical facilities. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol outlined below, which details the specific reagents, solvents, and workup procedures necessary to achieve the high performance metrics described in the patent literature.
- Perform an asymmetric Henry reaction between p-chlorobenzaldehyde and nitromethane using a chiral copper catalyst to generate (R)-2-nitro-1-(4-chlorophenyl)ethanol.
- React the nitro-alcohol intermediate with formaldehyde to extend the carbon chain, forming the diol backbone (1R,2R)-2-nitro-1-(4-chlorophenyl)-1,3-propanediol.
- Execute catalytic hydrogenation to reduce the nitro group to an amine, followed by dichloroacetylation and a final palladium-catalyzed nitro substitution to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nitromethane-based route offers profound advantages for procurement managers and supply chain leaders tasked with optimizing the cost structure of antibiotic production. The primary driver of value creation is the drastic simplification of the synthetic sequence, which reduces the number of unit operations and the associated labor, energy, and equipment costs. By eliminating the chiral resolution step, the process avoids the inherent 50% material loss of the traditional method, effectively doubling the theoretical output from the same amount of starting material. This improvement in atom economy translates directly into a lower cost per kilogram of the final API, providing a competitive edge in price-sensitive markets. Furthermore, the use of commodity raw materials like p-chlorobenzaldehyde and nitromethane ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized or volatile precursors that often plague complex pharmaceutical syntheses.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and wasteful reagents, specifically the aluminum isopropoxide used in conventional reductions. Removing this reagent not only lowers the direct material cost but also significantly reduces the expense associated with waste disposal and environmental compliance, as the treatment of aluminum-heavy waste streams is notoriously costly. Additionally, the higher overall yield means that less raw material is required to produce the same quantity of product, further compressing the variable cost of manufacturing. These cumulative savings allow for a more aggressive pricing strategy or improved margin retention, making the operation financially resilient against market fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals rather than specialized intermediates enhances the robustness of the supply chain. p-Chlorobenzaldehyde and nitromethane are produced on a massive global scale, ensuring that supply disruptions are unlikely to impact production schedules. This stability is crucial for maintaining continuous manufacturing operations and meeting the rigorous delivery timelines demanded by downstream pharmaceutical customers. Moreover, the shortened reaction sequence reduces the total cycle time from raw material intake to finished goods, allowing for faster response to changes in market demand and reducing the working capital tied up in work-in-progress inventory.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are amenable to large-scale batch or continuous processing. The avoidance of hazardous reagents and the generation of minimal waste align perfectly with modern environmental, social, and governance (ESG) goals, reducing the regulatory burden on the manufacturing facility. This green chemistry profile facilitates easier permitting and expansion, ensuring long-term operational continuity without the threat of environmental shutdowns. The high purity of the crude product also minimizes the need for extensive purification steps, streamlining the final isolation process and further enhancing the throughput capacity of the production plant.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Chloramphenicol synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of the technology. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or licensing agreements.
Q: How does the nitromethane route improve yield compared to the ethylbenzene method?
A: The traditional ethylbenzene route involves a chiral resolution step which theoretically limits yield to 50%. The new nitromethane route utilizes asymmetric catalysis to establish chirality early, avoiding the 50% loss associated with resolution and significantly boosting total recovery.
Q: What are the environmental benefits of avoiding aluminum isopropoxide?
A: Conventional methods rely on aluminum isopropoxide for reduction, generating substantial difficult-to-treat waste. The new method replaces this with catalytic hydrogenation, drastically reducing hazardous waste generation and simplifying downstream purification.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of cheap, readily available raw materials like p-chlorobenzaldehyde and nitromethane. The shortened five-step sequence and high purity (>98%) make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloramphenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic routes like the one described in CN102399165B to maintain competitiveness in the global antibiotic market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Chloramphenicol or its intermediates meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of asymmetric catalysis and palladium-mediated reactions with precision, delivering consistent quality that pharmaceutical partners can rely on for their critical drug formulations.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this optimized synthesis can drive value for your organization. Let us partner to engineer a more efficient, sustainable, and cost-effective future for your antibiotic production capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
