Advanced Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
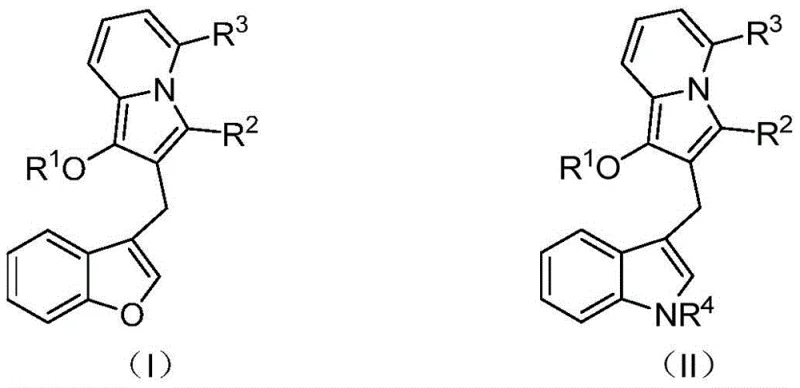
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN114163436A discloses a groundbreaking advancement in the synthesis of diarylmethane derivatives containing an indolizine core, a structural motif renowned for its potent biological profile. This innovation addresses the longstanding challenge of efficiently constructing fused heterocyclic systems that combine indolizine with benzofuran or indole moieties. The disclosed technology enables the production of high-value intermediates that possess dual antibacterial and anticancer activities, representing a significant leap forward from previous generations of compounds that were often limited to single-target inhibition. By leveraging a palladium-catalyzed tandem cyclization strategy, this method offers a streamlined pathway to diverse chemical spaces, making it an attractive candidate for commercial scale-up and drug discovery programs focused on oncology and infectious diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diarylmethane structures incorporating indolizine rings has been plagued by inefficient multi-step sequences that suffer from poor atom economy and low overall yields. Traditional routes often require the pre-functionalization of multiple fragments followed by harsh coupling conditions that can degrade sensitive heterocyclic cores. Furthermore, existing compounds, such as the prior art Compound A mentioned in the background, frequently exhibit a narrow spectrum of biological activity, primarily functioning as secreted phospholipase sPLA2 inhibitors without broader therapeutic utility. This limitation necessitates extensive medicinal chemistry campaigns to modify the scaffold, which is hindered by the rigidity and complexity of conventional synthetic pathways. The reliance on stoichiometric amounts of reagents and the generation of substantial chemical waste further exacerbate the environmental and economic burdens associated with these legacy manufacturing processes.
The Novel Approach
The methodology described in CN114163436A revolutionizes this landscape by introducing a convergent one-pot synthesis that directly assembles the target diarylmethane framework from readily available precursors. By utilizing 2-iodophenyl allene compounds and propargyl pyridine derivatives as starting materials, the process bypasses the need for isolated intermediate steps, thereby drastically reducing the operational complexity and time-to-market for these valuable intermediates. The reaction proceeds through a sophisticated palladium-catalyzed cascade that simultaneously forms multiple bonds, ensuring high structural fidelity and minimizing the formation of byproducts. This approach not only enhances the chemical diversity accessible through simple variation of the R groups but also aligns with green chemistry principles by improving step efficiency. The ability to access both benzofuran-fused and indole-fused variants through minor substrate modifications underscores the versatility of this platform for generating focused libraries for biological screening.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The core of this technological breakthrough lies in the intricate palladium-catalyzed mechanism that drives the tandem cyclization. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the 2-iodophenyl allene substrate, generating a reactive aryl-palladium species. This intermediate subsequently undergoes coordination with the alkyne moiety of the propargyl pyridine, followed by a migratory insertion that constructs the new carbon-carbon bond essential for the diarylmethane backbone. The presence of the allene functionality allows for a unique insertion pathway that facilitates the subsequent intramolecular nucleophilic attack or cyclization event, closing the indolizine ring system. The careful selection of ligands, such as triphenylphosphine in tetrakis(triphenylphosphine)palladium, stabilizes the catalytic cycle and prevents the formation of inactive palladium black, ensuring sustained turnover numbers throughout the reaction duration.
Impurity control is meticulously managed through the optimization of reaction parameters, particularly the choice of base and solvent system. The use of mild inorganic bases like potassium carbonate or cesium carbonate in polar aprotic solvents such as N,N-dimethylformamide (DMF) creates an environment that favors the desired cyclization over competing side reactions like homocoupling or polymerization. The moderate temperature range of 60-90°C provides sufficient thermal energy to overcome activation barriers without inducing thermal decomposition of the sensitive heterocyclic products. Post-reaction workup involving sequential extraction and column chromatography effectively removes residual palladium species and unreacted starting materials, resulting in a final product with high purity profiles suitable for downstream pharmaceutical applications. This mechanistic understanding allows for precise tuning of the process to accommodate various substituents, ensuring consistent quality across different derivative batches.
How to Synthesize Indolizine Diarylmethane Derivatives Efficiently
The practical implementation of this synthesis route is designed for reproducibility and scalability, making it ideal for process chemistry teams aiming to translate laboratory success to pilot plant operations. The protocol involves a straightforward mixing of the 2-iodophenyl allene and propargyl pyridine substrates with a catalytic amount of palladium source and base in an inert atmosphere. Detailed standardized operating procedures regarding stoichiometry, addition rates, and quenching methods are critical to maintaining safety and yield consistency. For a comprehensive guide on the specific molar ratios, solvent volumes, and purification techniques validated in the patent examples, please refer to the structured synthesis protocol below.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium salt catalyst, alkali, and organic solvent in a reaction vessel under inert atmosphere.
- Stir and react the mixture at a temperature between 60-90°C for a duration of 4-12 hours to facilitate the cyclization.
- Perform post-treatment including extraction with ethyl acetate, drying, filtration, solvent removal, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility often encountered in the procurement of complex heterocyclic intermediates. The reliance on commercially available and relatively inexpensive starting materials, such as substituted pyridines and iodobenzenes, mitigates the risk of raw material shortages that can plague supply chains dependent on exotic reagents. Furthermore, the consolidation of multiple synthetic steps into a single reactor operation significantly reduces the consumption of solvents, energy, and labor hours, translating into a leaner manufacturing cost structure. This efficiency gain is particularly valuable for long-term supply agreements where price stability is a key negotiation factor for procurement managers overseeing large-scale API production budgets.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps inherently lowers the operational expenditure associated with manufacturing these complex molecules. By avoiding the use of stoichiometric organometallic reagents and transitioning to a catalytic regime, the process minimizes the cost of goods sold (COGS) related to reagent consumption. Additionally, the simplified workflow reduces the burden on waste treatment facilities, as fewer solvent exchanges and workup phases are required, leading to substantial savings in environmental compliance costs. This economic efficiency allows suppliers to offer more competitive pricing models without compromising on the quality or purity specifications required by regulatory standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and utilize common laboratory solvents like DMF and ethyl acetate, ensures high process reliability and batch-to-batch consistency. This stability is crucial for supply chain heads who must guarantee uninterrupted material flow to downstream formulation sites. The use of stable palladium catalysts that are widely available from multiple global vendors further de-risks the supply chain against single-source dependencies. Consequently, manufacturers can maintain higher safety stock levels and shorter lead times, providing a buffer against market fluctuations and ensuring continuity of supply for critical drug development programs.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis is inherently scalable, as it avoids the accumulation of hazardous intermediates that often limit batch sizes in traditional multi-step processes. The reaction generates minimal heavy metal waste relative to the product mass due to the catalytic nature of the palladium species, simplifying the effluent treatment process and aiding in meeting stringent environmental regulations. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening regulatory frameworks but also enhances the corporate sustainability profile of the supply chain. The ability to scale from gram to kilogram quantities with minimal process re-engineering supports rapid clinical trial material production and seamless transition to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indolizine-containing diarylmethane derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent literature to ensure accuracy and relevance for industry professionals. Understanding these details is essential for R&D directors evaluating the feasibility of incorporating these scaffolds into their drug discovery pipelines and for procurement specialists assessing the viability of long-term sourcing strategies.
Q: What are the primary biological activities of these indolizine derivatives?
A: According to patent CN114163436A, these compounds exhibit significant antibacterial and anticancer activities, overcoming the single-activity limitation of prior art compounds like sPLA2 inhibitors.
Q: What is the key advantage of this synthesis method over conventional routes?
A: The novel approach utilizes a one-step tandem cyclization reaction catalyzed by palladium, which significantly simplifies the workflow compared to multi-step conventional syntheses, thereby reducing production time and complexity.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds at moderate temperatures of 60-90°C (optimally 80°C) using a palladium catalyst such as tetrakis(triphenylphosphine)palladium and a base like potassium carbonate in solvents like DMF.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the indolizine-containing diarylmethane scaffold in modern medicinal chemistry and are committed to supporting its development from bench to bedside. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards for pharmaceutical use. We understand that time is critical in drug development, and our optimized processes are designed to deliver high-quality materials with reduced lead times.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific therapeutic targets. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our efficient manufacturing capabilities can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary analogs. Let us be your strategic partner in bringing innovative anticancer and antibacterial agents to the global market.
