Revolutionizing Benzoxazole Production: A Transition-Metal-Free One-Step Strategy for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize critical heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN115028597A, which discloses a novel method for the preparation of benzoxazole derivatives via continuous dehydrocyclization mediated by TEMPO (2,2,6,6-tetramethylpiperidine oxide). This technology represents a paradigm shift from traditional multi-step syntheses, offering a streamlined one-pot protocol that constructs the benzoxazole heterocycle directly from readily available cyclohexanones and alkylamines. For R&D directors and procurement managers alike, this innovation addresses long-standing challenges regarding step economy, functional group tolerance, and environmental impact. By leveraging this oxidative dehydrogenation strategy, manufacturers can achieve high-purity intermediates essential for drug development while significantly simplifying the supply chain logistics associated with complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-substituted benzoxazole core has relied on strategies that are often fraught with inefficiencies and operational hazards. The most common traditional approach involves the condensation of o-aminophenols with carboxylic acid derivatives, such as acid halides or esters, typically requiring harsh thermal conditions and strong condensing agents. As illustrated in the prior art reaction schemes, these methods suffer from poor functional group compatibility due to the high temperatures required, and the starting o-aminophenols themselves are prone to oxidation and difficult to store long-term.
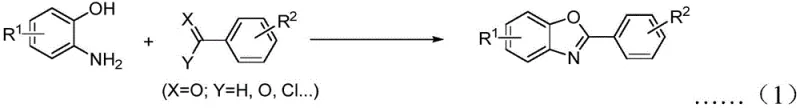
Alternative strategies involving transition metal-catalyzed coupling or intramolecular cyclization of substituted amidobenzenes introduce further complications. These routes often necessitate the use of expensive palladium or copper catalysts, which pose significant challenges for downstream purification, particularly in pharmaceutical applications where residual heavy metal limits are strictly regulated. Furthermore, the requirement for strong bases in these metal-catalyzed processes can lead to the degradation of sensitive functional groups on the substrate, thereby narrowing the scope of accessible chemical space and limiting the versatility of the synthesis for diverse drug candidates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN115028597A introduces a transition-metal-free pathway that utilizes TEMPO as both an oxidant and an oxygen transfer reagent. This innovative approach enables the direct conversion of cyclohexanone compounds and alkylamines into benzoxazole derivatives through a continuous dehydrocyclization process. The reaction proceeds under relatively mild conditions, typically in 1,4-dioxane at 120°C, avoiding the extreme thermal stress associated with traditional condensation reactions. This method not only simplifies the operational workflow by combining multiple transformation steps into a single vessel but also enhances the overall atom economy of the process.

The versatility of this new synthetic route is evidenced by its broad substrate scope, accommodating a wide range of substituents on both the cyclohexanone and amine components without compromising yield or selectivity. By eliminating the need for pre-functionalized starting materials like o-aminophenols or ortho-halo amides, this method drastically reduces the number of synthetic steps required to access the target scaffold. For a reliable pharmaceutical intermediate supplier, this translates to a more robust and flexible manufacturing platform capable of delivering diverse benzoxazole libraries with improved purity profiles and reduced environmental footprint.
Mechanistic Insights into TEMPO-Catalyzed Oxidative Dehydrocyclization
The core of this technological advancement lies in the unique reactivity of the TEMPO radical system, which facilitates a cascade of oxidative transformations. Mechanistically, the reaction initiates with the in situ condensation of the cyclohexanone and the alkylamine to form an imine or enamine intermediate. TEMPO then acts as a hydrogen abstractor, promoting the sequential dehydrogenation of this intermediate. This oxidative process is critical for aromatizing the six-membered ring, effectively converting the saturated cyclohexanone moiety into the aromatic phenol component of the benzoxazole ring system. Simultaneously, TEMPO participates in the oxygen transfer necessary to close the oxazole ring, completing the heterocyclic construction in a single operational sequence.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal-induced side reactions, such as homocoupling or undesired C-H activation at non-target sites, which often complicate the impurity profile of the final product. Furthermore, the high selectivity of the TEMPO-mediated oxidation ensures that sensitive functional groups, such as esters, halides, and protected amines present on the R1 and R2 substituents, remain intact throughout the reaction. This chemoselectivity is paramount for synthesizing complex pharmaceutical intermediates where preserving structural integrity is essential for biological activity.
How to Synthesize Benzoxazole Derivatives Efficiently
The practical implementation of this synthesis is straightforward, making it highly attractive for process chemistry teams aiming to scale up production. The standard protocol involves mixing the cyclohexanone substrate, the alkylamine, and TEMPO in a solvent such as 1,4-dioxane, often with the addition of molecular sieves to drive the equilibrium by removing water. The mixture is stirred at room temperature briefly before being heated to 120°C under an inert atmosphere for approximately 36 hours. Upon completion, the workup is simple, involving filtration to remove the desiccant and catalyst residues, followed by concentration and standard purification techniques like column chromatography. Detailed standardized synthesis steps are provided in the guide below.
- Mix cyclohexanone compound, alkylamine, and TEMPO oxidant in 1,4-dioxane solvent with molecular sieves.
- Stir the mixture at room temperature for 1 hour, then heat to 120°C under inert atmosphere for 36 hours.
- Filter the reaction mixture, concentrate under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TEMPO-mediated synthesis offers compelling economic and logistical benefits. The primary advantage stems from the drastic simplification of the raw material portfolio; instead of sourcing expensive, unstable, or highly specialized precursors like o-aminophenols or halogenated amides, manufacturers can utilize commodity chemicals such as substituted cyclohexanones and benzylamines. These starting materials are widely available from bulk chemical suppliers, ensuring a stable and resilient supply chain that is less susceptible to market fluctuations or geopolitical disruptions affecting niche reagents.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts represents a significant cost saving opportunity, not only in terms of reagent purchase but also in downstream processing. Removing the need for expensive scavengers or specialized filtration steps to meet strict heavy metal specifications (e.g., ICH Q3D guidelines) drastically reduces production costs. Additionally, the one-pot nature of the reaction minimizes solvent usage, labor hours, and waste generation, leading to substantial overall cost reductions in pharmaceutical intermediate manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: By relying on robust, commercially available starting materials and avoiding air-sensitive catalysts, the manufacturing process becomes inherently more reliable. The stability of the reagents allows for easier storage and handling, reducing the risk of batch failures due to reagent degradation. This reliability translates to shorter lead times for high-purity benzoxazole derivatives, enabling faster response to customer demands and more predictable delivery schedules for critical drug development projects.
- Scalability and Environmental Compliance: The transition-metal-free nature of this process aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The absence of heavy metals simplifies waste treatment and disposal, lowering the environmental compliance burden. Furthermore, the reaction conditions are amenable to scale-up from gram to kilogram and ton scales, as demonstrated by the successful synthesis of various derivatives in the patent data, ensuring that the process can meet the growing demands of commercial API production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this novel synthesis method. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the technology's capabilities and limitations for potential partners and stakeholders.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method utilizes TEMPO as an organic oxidant, completely eliminating the need for expensive and toxic transition metal catalysts.
Q: What are the typical reaction conditions for this benzoxazole synthesis?
A: The reaction typically proceeds in 1,4-dioxane at 120°C for approximately 36 hours under an inert nitrogen atmosphere.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the one-pot nature, absence of heavy metals, and use of stable starting materials make it highly suitable for commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this TEMPO-mediated dehydrocyclization technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzoxazole derivative we deliver adheres to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this metal-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with superior chemical solutions.
