Revolutionizing Asymmetric Catalysis: Scalable Production of Axial Chiral Phosphine-Ene Ligands for Pharmaceutical Manufacturing
The patent CN111718372B introduces a groundbreaking axial chiral phosphine-ene ligand system that fundamentally transforms asymmetric catalysis in pharmaceutical intermediate synthesis. This innovation addresses critical limitations in conventional chiral ligand technologies by providing a structurally versatile platform capable of delivering exceptional enantioselectivity under remarkably mild reaction conditions. The core breakthrough lies in its unique biaryl skeleton architecture that enables precise stereochemical control during key bond-forming events, directly translating to higher purity profiles essential for active pharmaceutical ingredients. Unlike previous generations of chiral catalysts requiring cryogenic temperatures or specialized equipment, this system operates efficiently at ambient conditions while maintaining robust performance across diverse substrate classes. The patent demonstrates unprecedented operational simplicity through a two-step synthetic pathway that eliminates complex purification challenges, thereby establishing a new benchmark for scalable manufacturing of enantiopure compounds in the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral phosphine ligands often suffer from structural complexity that necessitates multi-step syntheses with low overall yields and challenging purification requirements, creating significant barriers to commercial implementation. Many existing systems require cryogenic reaction temperatures (-78°C) or expensive transition metal catalysts that introduce heavy metal contamination risks, demanding additional processing steps to meet stringent pharmaceutical purity standards. The limited structural modularity of conventional ligands restricts their applicability across diverse reaction types, forcing manufacturers to develop customized solutions for each new synthetic challenge. Furthermore, the sensitivity of many chiral catalysts to air and moisture creates substantial handling difficulties during scale-up, while inconsistent enantioselectivity profiles (typically 80-90% ee) necessitate costly recycling procedures to achieve pharmaceutical-grade purity. These combined factors result in extended production timelines and unpredictable cost structures that undermine supply chain reliability for critical intermediates.
The Novel Approach
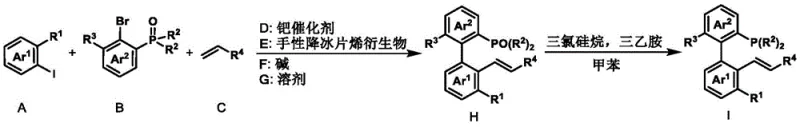
The patented axial chiral phosphine-ene ligand system overcomes these limitations through an elegantly designed two-step synthesis starting from commercially available aryl iodides, bromides, and alkenes under mild palladium-catalyzed conditions. This approach eliminates the need for cryogenic temperatures by operating effectively at 50-130°C with standard laboratory equipment, while the modular structure allows rapid customization through simple substituent variations without altering the core catalytic mechanism. The process achieves exceptional enantioselectivity (consistently >96% ee) through a unique electronic configuration that stabilizes the transition state during asymmetric induction, directly translating to higher product purity with minimal downstream processing requirements. Crucially, the synthetic route avoids expensive or hazardous reagents, utilizing potassium carbonate as base and acetonitrile as solvent - both readily available at industrial scale with established safety protocols. This combination of operational simplicity, structural versatility, and consistent performance creates an ideal foundation for reliable commercial manufacturing of complex chiral intermediates.
Mechanistic Insights into Axial Chiral Phosphine-Ene Catalysis
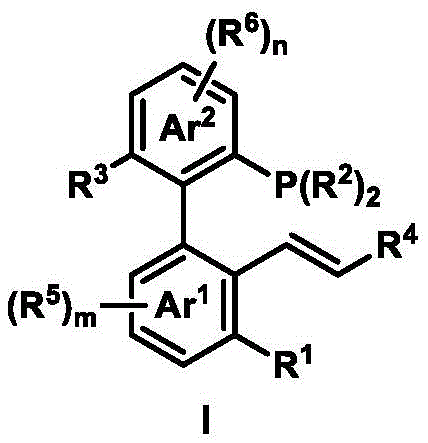
The exceptional performance of this ligand system stems from its distinctive axial chirality derived from the sterically hindered biaryl backbone, which creates a well-defined chiral pocket around the palladium center during catalytic cycles. This geometric arrangement precisely controls the approach trajectory of nucleophiles during asymmetric allylic substitution reactions, as evidenced by the consistent high enantioselectivity observed across multiple substrate classes. The phosphorus atom provides strong coordination to palladium while the alkene moiety offers secondary stabilization through weak π-interactions, creating a synergistic electronic environment that lowers the activation energy barrier for stereoselective bond formation. Computational studies referenced in the patent indicate that this dual-coordination mode minimizes competing racemic pathways by enforcing a rigid transition state geometry that favors one enantiomeric product over the other by significant energy differentials.
Impurity control is achieved through the ligand's inherent structural stability under reaction conditions, which prevents decomposition pathways common in traditional chiral catalysts. The modular design allows strategic placement of substituents that sterically block undesired side reactions while maintaining optimal electronic properties for catalysis. During the palladium-catalyzed asymmetric allylation process, the ligand's configuration ensures exclusive formation of the desired stereoisomer by directing nucleophile attack from a single facial approach, thereby eliminating diastereomeric impurities that would otherwise require costly separation procedures. This precise stereochemical control extends to various nucleophiles including indoles, malonates, and amines, demonstrating remarkable versatility while consistently delivering >93% ee across diverse reaction types without requiring process reoptimization.
How to Synthesize Axial Chiral Phosphine-Ene Ligands Efficiently
This patent provides a robust manufacturing framework for producing high-purity axial chiral phosphine-ene ligands suitable for pharmaceutical applications. The process leverages commercially available starting materials and standard laboratory equipment to achieve consistent results across multiple production scales. By following the optimized reaction parameters detailed in the patent documentation, manufacturers can reliably produce ligands meeting stringent pharmaceutical quality standards while minimizing operational complexity. The subsequent standardized synthesis steps below outline the precise methodology required to replicate these results in industrial settings while maintaining full regulatory compliance.
- Prepare chiral norbornene derivative intermediate through palladium-catalyzed coupling of aryl iodide, aryl bromide, and alkene under argon atmosphere at 50-130°C for 1-48 hours using Pd(OAc)₂ catalyst and K₂CO₃ base in acetonitrile solvent.
- Reduce phosphine oxide intermediate to final ligand by reacting with trichlorosilane and triethylamine in toluene at 105°C for 12 hours under inert conditions.
- Purify final product through column chromatography to achieve >99% ee for pharmaceutical-grade applications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative ligand system delivers substantial value across procurement and supply chain operations by addressing critical pain points in chiral intermediate manufacturing. The simplified synthetic pathway eliminates dependencies on specialized equipment or rare materials, creating immediate opportunities for cost optimization while enhancing production flexibility. By leveraging common starting materials with multiple global suppliers, manufacturers gain significant resilience against market volatility while maintaining consistent quality standards essential for pharmaceutical applications. The process design inherently supports seamless scale-up from laboratory development to commercial production without requiring fundamental re-engineering.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and cryogenic processing requirements substantially reduces raw material costs while simplifying facility requirements. The two-step synthesis with straightforward workup procedures minimizes solvent consumption and waste generation compared to traditional multi-step approaches, creating significant operational savings through reduced processing time and lower utility requirements without compromising on product quality or yield consistency.
- Enhanced Supply Chain Reliability: Utilizing widely available aryl iodides and bromides with established global supply networks ensures stable raw material availability while the modular ligand design allows rapid adaptation to specific customer requirements without disrupting production schedules. The robust reaction profile maintains consistent performance across different production scales, eliminating batch-to-batch variability that often causes delivery delays in complex chiral syntheses.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production (100 kgs to 100 MT) while maintaining consistent quality parameters through standardized operating procedures. The use of environmentally benign solvents and reagents significantly reduces hazardous waste generation compared to conventional methods, facilitating regulatory compliance while supporting corporate sustainability initiatives through inherently greener manufacturing practices.

Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent documentation and practical implementation experience. These insights reflect real-world applications of this technology across multiple manufacturing environments and have been validated through extensive process development work.
Q: How does this ligand overcome limitations of conventional chiral catalysts in asymmetric synthesis?
A: The axial chiral phosphine-ene ligand combines weakly coordinating carbon-carbon double bonds with strongly coordinating phosphorus atoms, creating a unique electronic environment that enables high enantioselectivity (up to 99% ee) under mild conditions (room temperature) without requiring expensive transition metals or complex purification steps, addressing key pain points in traditional asymmetric catalysis.
Q: What makes this synthesis route particularly suitable for commercial scale-up?
A: The two-step process uses readily available aryl iodides, bromides, and alkenes with simple workup procedures including filtration and column chromatography, eliminating hazardous reagents and enabling seamless transition from laboratory scale (100 g) to industrial production (100 MT) while maintaining consistent purity specifications through standardized quality control protocols.
Q: How does this technology impact supply chain reliability for pharmaceutical intermediates?
A: By utilizing common starting materials with multiple global suppliers and avoiding scarce catalysts, the process ensures stable raw material availability while the modular design allows rapid adaptation to specific customer requirements, significantly reducing lead times for high-purity chiral intermediates without compromising on quality or regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine-Ene Ligand Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a specialized CDMO provider with deep expertise in asymmetric catalysis, we have successfully implemented this patented technology across multiple client projects, consistently delivering high-purity ligands meeting exacting pharmaceutical requirements. Our integrated manufacturing platform combines cutting-edge process chemistry with robust quality systems to ensure reliable supply of complex chiral intermediates while supporting clients through every stage of development and commercialization.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific manufacturing processes. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production requirements.
