Revolutionizing Edoxaban Intermediate Production Through Advanced Asymmetric Organocatalysis and Scalable Process Design
Revolutionizing Edoxaban Intermediate Production Through Advanced Asymmetric Organocatalysis and Scalable Process Design
The global demand for direct oral anticoagulants continues to surge as the population ages and cardiovascular diseases become more prevalent, placing immense pressure on supply chains to deliver high-quality active pharmaceutical ingredients efficiently. Patent CN111763157B represents a significant technological leap in this domain by disclosing a novel preparation method for chiral amino compounds that serve as critical precursors for edoxaban, a potent Factor Xa inhibitor. This intellectual property outlines a streamlined synthetic strategy that bypasses the traditional reliance on expensive chiral pool starting materials and hazardous reagents, offering a robust alternative for the production of high-purity pharmaceutical intermediates. The core innovation lies in the ability to construct the chiral cyclohexane diamine scaffold through a highly efficient, one-step asymmetric catalytic reaction that delivers exceptional optical purity without the need for downstream resolution processes. 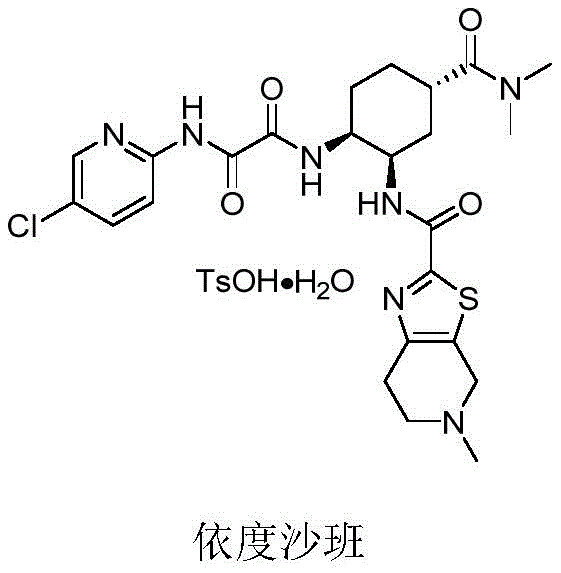 For R&D directors and process chemists, this patent provides a blueprint for overcoming the longstanding bottlenecks associated with the stereochemical control of edoxaban intermediates. By leveraging advanced organocatalysis, the disclosed method ensures that the configuration of the product is strictly determined by the chirality of the catalyst, thereby guaranteeing consistent quality and reducing the variability often seen in biological resolution methods. This level of control is paramount for meeting the stringent regulatory requirements of major health authorities, ensuring that the final drug substance maintains its efficacy and safety profile throughout its shelf life.
For R&D directors and process chemists, this patent provides a blueprint for overcoming the longstanding bottlenecks associated with the stereochemical control of edoxaban intermediates. By leveraging advanced organocatalysis, the disclosed method ensures that the configuration of the product is strictly determined by the chirality of the catalyst, thereby guaranteeing consistent quality and reducing the variability often seen in biological resolution methods. This level of control is paramount for meeting the stringent regulatory requirements of major health authorities, ensuring that the final drug substance maintains its efficacy and safety profile throughout its shelf life.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of edoxaban has been plagued by inefficiencies inherent in classical resolution techniques and the use of dangerous chemical reagents. Traditional routes typically commence with racemic 3-cyclohexene-1-carboxylic acid, which must undergo a tedious chiral resolution process using expensive chiral amines to isolate the desired (S)-enantiomer, inherently limiting the maximum theoretical yield to 50% and generating substantial waste. Furthermore, subsequent steps in these legacy pathways often necessitate the introduction of amino groups via nucleophilic substitution using sodium azide, a reagent known for its high toxicity and explosive potential, which imposes severe safety constraints on manufacturing facilities and complicates waste treatment protocols. 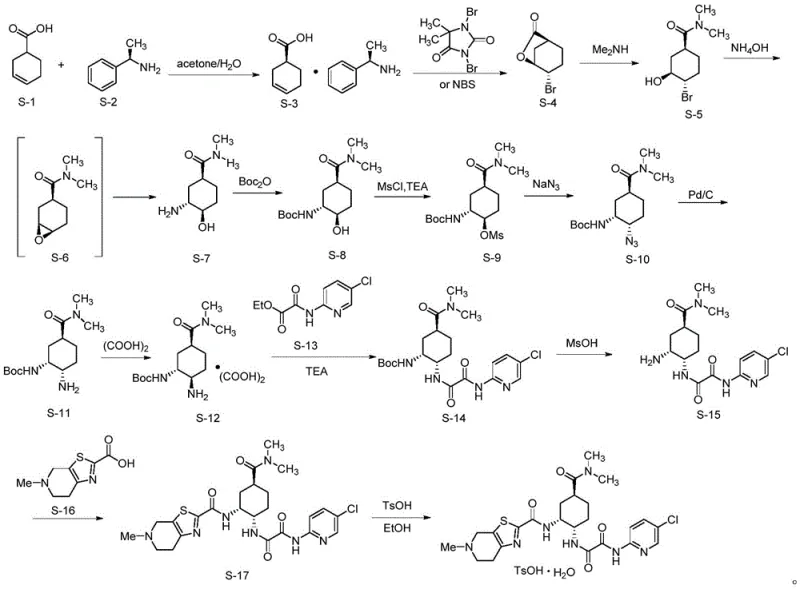 The reliance on these archaic methodologies results in a prolonged production timeline, increased operational costs due to the need for specialized safety equipment, and a larger environmental footprint due to poor atom economy. Additionally, the multi-step nature of the conventional synthesis, involving repeated protection and deprotection of functional groups, increases the likelihood of impurity formation and reduces the overall process robustness, making it difficult to maintain consistent batch-to-batch quality at a commercial scale. These factors collectively hinder the ability of manufacturers to respond agilely to market demands and keep production costs competitive in a price-sensitive generic pharmaceutical landscape.
The reliance on these archaic methodologies results in a prolonged production timeline, increased operational costs due to the need for specialized safety equipment, and a larger environmental footprint due to poor atom economy. Additionally, the multi-step nature of the conventional synthesis, involving repeated protection and deprotection of functional groups, increases the likelihood of impurity formation and reduces the overall process robustness, making it difficult to maintain consistent batch-to-batch quality at a commercial scale. These factors collectively hinder the ability of manufacturers to respond agilely to market demands and keep production costs competitive in a price-sensitive generic pharmaceutical landscape.
The Novel Approach
In stark contrast, the methodology described in Patent CN111763157B introduces a paradigm shift by utilizing a direct asymmetric construction of the chiral center from simple, achiral starting materials. The novel approach employs a catalytic reaction between a nitroalkene derivative and acetaldehyde, mediated by a chiral organocatalyst, to generate the key chiral amino compound in a single step with high enantiomeric excess. This strategy effectively circumvents the need for chiral resolution, theoretically doubling the yield potential compared to resolution-based methods and significantly reducing the consumption of raw materials.  Moreover, the new route successfully eliminates the use of sodium azide, replacing it with safer and more manageable reagents, which drastically lowers the process risk profile and simplifies the regulatory compliance burden for manufacturing sites. The brevity of the synthetic sequence, combined with mild reaction conditions and the use of commercially available catalysts, enhances the overall feasibility of the process for large-scale industrial application. This modernization of the synthetic pathway not only improves the economic viability of edoxaban production but also aligns with the principles of green chemistry by minimizing waste generation and energy consumption.
Moreover, the new route successfully eliminates the use of sodium azide, replacing it with safer and more manageable reagents, which drastically lowers the process risk profile and simplifies the regulatory compliance burden for manufacturing sites. The brevity of the synthetic sequence, combined with mild reaction conditions and the use of commercially available catalysts, enhances the overall feasibility of the process for large-scale industrial application. This modernization of the synthetic pathway not only improves the economic viability of edoxaban production but also aligns with the principles of green chemistry by minimizing waste generation and energy consumption.
Mechanistic Insights into Organocatalytic Asymmetric Michael Addition
The heart of this technological advancement lies in the sophisticated mechanism of the organocatalytic reaction, which leverages the power of chiral amines to induce stereochemistry during carbon-carbon bond formation. The reaction proceeds via the formation of a chiral enamine or iminium ion intermediate between the catalyst and the aldehyde substrate, which then undergoes a stereoselective attack on the nitroalkene electrophile. The specific spatial arrangement of the catalyst, often derived from proline or its derivatives with bulky aryl substituents, creates a well-defined chiral environment that shields one face of the reacting species, ensuring that the nucleophilic attack occurs exclusively from the desired trajectory.  This precise control over the transition state geometry is what allows the process to achieve optical purity levels exceeding 90% ee without the need for further purification. The choice of additives, such as organic acids, plays a crucial role in modulating the acidity of the reaction medium and stabilizing the transition state, further enhancing both the reaction rate and the stereoselectivity. Understanding these mechanistic nuances is vital for process optimization, as slight variations in catalyst loading, solvent polarity, or temperature can have profound effects on the diastereomeric ratio and overall yield. The robustness of this catalytic system suggests that it can be fine-tuned to accommodate various substrates, offering a versatile platform for the synthesis of related chiral building blocks beyond just edoxaban intermediates.
This precise control over the transition state geometry is what allows the process to achieve optical purity levels exceeding 90% ee without the need for further purification. The choice of additives, such as organic acids, plays a crucial role in modulating the acidity of the reaction medium and stabilizing the transition state, further enhancing both the reaction rate and the stereoselectivity. Understanding these mechanistic nuances is vital for process optimization, as slight variations in catalyst loading, solvent polarity, or temperature can have profound effects on the diastereomeric ratio and overall yield. The robustness of this catalytic system suggests that it can be fine-tuned to accommodate various substrates, offering a versatile platform for the synthesis of related chiral building blocks beyond just edoxaban intermediates.
From an impurity control perspective, the direct nature of this synthesis minimizes the formation of side products that are typically associated with multi-step sequences. In conventional routes, each additional step introduces opportunities for side reactions, such as over-alkylation, elimination, or hydrolysis, which can lead to complex impurity profiles that are difficult to separate. By condensing the synthesis into fewer steps, the novel method inherently reduces the cumulative impurity load, simplifying the downstream purification process. The high diastereoselectivity observed in the subsequent cyclization step, where the chiral amino compound reacts with a phosphonate ester, further ensures that the final intermediate possesses the correct relative stereochemistry required for biological activity. This level of purity is essential for preventing the formation of toxic metabolites and ensuring the therapeutic efficacy of the final drug product.
How to Synthesize Chiral Amino Compound Efficiently
The practical implementation of this patented technology involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the reaction mixture under controlled atmospheric conditions to prevent moisture interference, followed by the sequential addition of the nitroalkene substrate, the chiral catalyst, and the aldehyde source. Reaction monitoring is typically conducted using thin-layer chromatography (TLC) or HPLC to determine the endpoint, ensuring that the starting material is fully consumed before proceeding to workup.
- React nitroalkene derivatives with acetaldehyde in the presence of a chiral organocatalyst (e.g., proline derivative) and an organic acid additive in a suitable solvent like 1,4-dioxane.
- Monitor the reaction progress via TLC until the nitroalkene starting material is fully consumed, typically requiring 1 to 5 days at temperatures between -20°C and 30°C.
- Purify the resulting chiral amino aldehyde via column chromatography to achieve high optical purity (ee ≥ 90%) ready for subsequent cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers compelling strategic advantages that extend far beyond simple technical metrics. The primary benefit lies in the drastic simplification of the supply chain, as the new method relies on commodity chemicals like acetaldehyde and readily available nitroalkenes rather than scarce and expensive chiral acids. This shift in raw material sourcing mitigates the risk of supply disruptions and price volatility often associated with specialty chiral pool materials, ensuring a more stable and predictable cost structure for long-term production contracts. Furthermore, the elimination of hazardous reagents like sodium azide reduces the need for specialized storage facilities and expensive safety protocols, leading to significant operational cost savings and lower insurance premiums for manufacturing sites.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the removal of the chiral resolution step which traditionally caps yield at 50% and requires the purchase of stoichiometric amounts of expensive resolving agents. By achieving high optical purity directly through catalysis, the process maximizes atom economy and reduces the volume of waste solvents and reagents that must be treated or disposed of. Additionally, the shorter synthetic sequence translates to reduced labor hours, lower energy consumption for heating and cooling, and decreased equipment occupancy time, all of which contribute to a substantially lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals with established global supply networks enhances the resilience of the supply chain against geopolitical or logistical shocks. Unlike complex chiral intermediates that may be sourced from a limited number of suppliers, the starting materials for this new route are produced by multiple vendors worldwide, providing procurement teams with greater flexibility and negotiating power. The robustness of the catalytic process also means that production can be scaled up rapidly to meet sudden spikes in demand without the lengthy lead times associated with sourcing specialized chiral starting materials, ensuring continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology represents a significant improvement over legacy methods. The avoidance of explosive azides and the reduction in waste generation align perfectly with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals and audits. The mild reaction conditions, often operating at or near room temperature, reduce the energy intensity of the process, contributing to a lower carbon footprint. These sustainability credentials are becoming increasingly important for pharmaceutical companies aiming to meet their corporate social responsibility goals and can serve as a key differentiator when bidding for contracts with environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial viability of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making.
Q: How does this new method improve safety compared to traditional edoxaban synthesis?
A: The novel process completely eliminates the use of sodium azide, a hazardous and potentially explosive reagent required in conventional routes, thereby significantly reducing process risk and simplifying safety compliance.
Q: What is the optical purity achievable with this asymmetric catalytic route?
A: The method utilizes specific chiral organocatalysts to directly induce asymmetry, achieving high optical purity with ee values greater than or equal to 90% without the need for chiral resolution.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the route uses cheap and readily available raw materials like acetaldehyde and avoids complex resolution steps, making it highly suitable for large-scale industrial production with stable yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic strategies outlined in Patent CN111763157B and are uniquely positioned to bring this technology to commercial reality. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing our state-of-the-art rigorous QC labs to verify every batch against the highest international standards. Our infrastructure is designed to handle complex asymmetric syntheses with precision, guaranteeing the consistent optical purity required for potent anticoagulant drugs.
We invite potential partners to engage with our technical procurement team to discuss how we can tailor this innovative process to your specific volume and quality requirements. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient synthetic route. We encourage you to request specific COA data and route feasibility assessments to validate the superiority of our manufacturing capabilities. Let us be your trusted partner in navigating the complexities of modern pharmaceutical supply chains and securing a competitive edge in the global market for cardiovascular therapeutics.
