Advanced Copper-Catalyzed Goldberg Reaction for Scalable Phenylamide Manufacturing
Advanced Copper-Catalyzed Goldberg Reaction for Scalable Phenylamide Manufacturing
The chemical industry continuously seeks robust methodologies for constructing carbon-nitrogen bonds, particularly for the synthesis of phenylamide compounds which serve as pivotal scaffolds in pharmaceutical and agrochemical portfolios. A significant technological advancement in this domain is detailed in patent CN102432488A, which discloses a novel method for preparing phenylamide compounds utilizing a specialized copper-ligand catalytic system. This innovation addresses long-standing inefficiencies in the traditional Goldberg reaction by employing 1,1'-bis(diphenylphosphino)ferrocene (Dppf) as a bidentate phosphine ligand. By optimizing the coordination environment around the copper center, this process enables the coupling of halogenated aromatic hydrocarbons with amides under remarkably mild conditions, typically ranging from 50°C to 150°C. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this ligand-accelerated catalysis is crucial for evaluating process viability and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylamides via the Goldberg reaction has been plagued by severe operational constraints that hinder scalability and economic efficiency. Traditional protocols often rely on stoichiometric amounts of copper powder or simple copper salts, necessitating extreme reaction temperatures between 180°C and 250°C to overcome the high activation energy barrier. Such harsh thermal conditions not only demand significant energy input but also frequently lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles. Furthermore, older methods often require prolonged reaction times extending into tens of hours, resulting in low throughput and substantial reactor occupancy. While palladium-catalyzed cross-coupling emerged as a milder alternative, its widespread adoption in bulk chemical manufacturing is restricted by the prohibitive cost of noble metal catalysts and the stringent environmental regulations surrounding heavy metal residue removal. These factors collectively create a bottleneck for the commercial scale-up of complex pharmaceutical intermediates, driving the need for more sustainable catalytic solutions.
The Novel Approach
The methodology described in the patent data introduces a paradigm shift by integrating the ferrocene-based diphosphine ligand Dppf into the copper catalytic cycle. This approach effectively lowers the reaction temperature to a manageable range of 50°C to 150°C while reducing reaction times to between 2 and 24 hours. The introduction of this specific ligand system transforms the heterogeneous or poorly soluble copper species into a highly active homogeneous catalyst, facilitating smoother interaction between the aryl halide and the amide nucleophile. As illustrated in the representative reaction scheme below, the coupling of iodobenzene with acetanilide proceeds efficiently to yield N,N-diphenylacetamide, demonstrating the practical applicability of this system for generating high-value amide bonds.
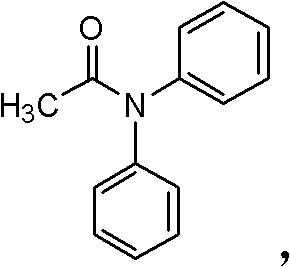
This refined process not only mitigates the thermal stress on equipment but also significantly simplifies the downstream purification workflow. By avoiding the extreme conditions of the past, manufacturers can achieve higher selectivity and reduced byproduct formation, directly translating to improved overall yields and reduced waste generation. For procurement managers focused on cost reduction in fine chemical manufacturing, this transition from high-energy, stoichiometric processes to low-energy, catalytic ones represents a tangible opportunity to optimize production economics without compromising on product quality or purity specifications.
Mechanistic Insights into Dppf-Promoted Copper Catalysis
The efficacy of this synthetic route lies in the unique electronic and steric properties of the 1,1'-bis(diphenylphosphino)ferrocene ligand. Unlike simple monodentate ligands, Dppf possesses a rigid backbone and strong chelating ability that allows it to form stable complexes with cuprous halides. As depicted in the mechanistic diagram, the ligand first coordinates with the copper salt to form a soluble complex (I), which serves as the active catalytic precursor. This solubilization is critical, as it ensures that the catalyst is uniformly distributed throughout the organic reaction medium, maximizing the frequency of productive collisions between reactants. The stability of this complex against air and moisture further enhances the operational robustness of the method, allowing for more flexible handling procedures in a production environment.
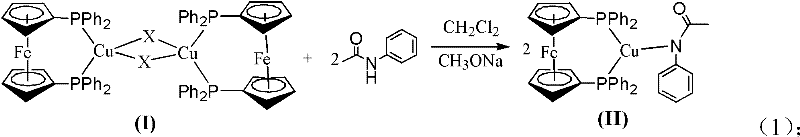
Upon formation of the initial complex, the system readily interacts with the amide substrate to generate a key intermediate (II) containing a copper-nitrogen bond. The formation of this Cu-N species is the rate-determining step that traditionally requires high energy; however, the electron-rich nature of the Dppf ligand stabilizes the copper center and lowers the activation energy for this oxidative addition or coordination step. Consequently, the subsequent coupling with the halogenated aromatic hydrocarbon proceeds rapidly even at moderate temperatures. This mechanistic pathway effectively prevents the catalyst deactivation often observed when nucleophilic amides coordinate too strongly to unprotected copper centers. For technical teams evaluating high-purity pharmaceutical intermediates, this controlled mechanism ensures a cleaner reaction profile with fewer side reactions, thereby reducing the burden on purification units and ensuring consistent batch-to-batch quality.
How to Synthesize N,N-Diphenylacetamide Efficiently
Implementing this advanced catalytic protocol requires precise control over reaction parameters to maximize the benefits of the Dppf-copper system. The general procedure involves charging a reaction vessel with the halogenated aromatic substrate and the amide derivative, typically in a molar ratio ranging from 1:1 to 1:2 to drive the reaction to completion. Following the addition of the copper source and the Dppf ligand, an inorganic base such as potassium carbonate or potassium phosphate is introduced to facilitate the deprotonation of the amide. The reaction is then heated in a polar aprotic solvent like DMF or DMSO, maintaining the temperature within the optimized window of 80°C to 120°C for optimal kinetics. Detailed standardized operating procedures regarding specific reagent grades, mixing rates, and workup protocols are essential for reproducibility.
- Combine halogenated aromatic hydrocarbons and amide derivatives in a molar ratio of 1: 1 to 1:2 within a reaction vessel, ensuring precise stoichiometry for optimal yield.
- Introduce cuprous halide and 1,1'-bis(diphenylphosphino)ferrocene (Dppf) as the catalytic system, followed by the addition of an inorganic base and a polar organic solvent.
- Maintain the reaction mixture at a controlled temperature between 50°C and 150°C for a duration of 2 to 24 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this Dppf-promoted copper catalysis offers compelling advantages that extend beyond mere technical performance. The shift away from palladium-based systems eliminates the volatility associated with noble metal pricing, providing a more predictable cost structure for long-term supply agreements. Furthermore, the use of earth-abundant copper salts significantly reduces the raw material cost per kilogram of product, allowing for more competitive pricing models in the global marketplace. The milder reaction conditions also imply reduced energy consumption for heating and cooling cycles, contributing to a lower carbon footprint and aligning with modern sustainability mandates. These factors collectively enhance the attractiveness of suppliers who can demonstrate mastery over such efficient synthetic routes.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the reduction in energy requirements due to lower operating temperatures result in substantial cost savings. By utilizing catalytic amounts of inexpensive copper salts instead of stoichiometric quantities of reagents, the overall material cost is drastically simplified. This economic efficiency allows for the production of high-quality intermediates at a fraction of the cost associated with traditional noble metal catalysis, providing a distinct competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reagents required for this process, including cuprous halides and Dppf ligands, are commercially available from multiple global sources, mitigating the risk of single-source dependency. The robustness of the reaction against minor variations in moisture and air exposure further reduces the likelihood of batch failures, ensuring consistent delivery schedules. This reliability is critical for downstream manufacturers who depend on uninterrupted flows of high-purity phenylamide derivatives to maintain their own production timelines.
- Scalability and Environmental Compliance: The moderate temperature range and the absence of toxic heavy metals simplify the engineering requirements for scale-up, making the transition from laboratory to pilot and commercial plant straightforward. Additionally, the reduced generation of heavy metal waste simplifies effluent treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This ease of scalability supports the reducing lead time for high-purity intermediates by minimizing the technical hurdles often encountered during process validation and regulatory filing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders assess the feasibility of integrating this technology into their existing manufacturing frameworks.
Q: What are the primary advantages of using Dppf ligands in copper-catalyzed Goldberg reactions?
A: The use of 1,1'-bis(diphenylphosphino)ferrocene (Dppf) significantly enhances the solubility of copper salts in organic solvents and stabilizes the catalytic species. This ligand prevents the deactivation of the catalyst by nucleophilic amides, allowing the reaction to proceed at much lower temperatures (50-150°C) compared to traditional methods requiring over 180°C.
Q: How does this method compare to palladium-catalyzed alternatives in terms of cost and environmental impact?
A: Unlike palladium-catalyzed couplings which involve expensive noble metals and generate toxic heavy metal waste, this copper-based methodology utilizes abundant and inexpensive copper salts. This substitution drastically reduces raw material costs and simplifies waste treatment protocols, making it more suitable for large-scale industrial applications.
Q: What types of substrates are compatible with this synthetic protocol?
A: The protocol demonstrates broad substrate scope, accommodating various halogenated aromatics including iodobenzene and bromobenzene derivatives. It is effective for synthesizing diverse phenylamide structures, including N,N-diphenylacetamides and trifluoroacetamides, which are critical scaffolds in pharmaceutical and agrochemical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Dppf-copper catalytic system are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenylamide compound meets the exacting standards required by the global pharmaceutical industry. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us help you optimize your supply chain with reliable, cost-effective, and high-quality chemical solutions.
