Advanced One-Step Synthesis of Polysubstituted Indoles for Efficient Indomethacin Production
The pharmaceutical industry continuously seeks more efficient pathways to construct complex heterocyclic scaffolds, particularly the indole core which is ubiquitous in bioactive molecules. Patent CN103012241A introduces a groundbreaking methodology for the synthesis of polysubstituted indoles, specifically targeting the production of non-steroidal anti-inflammatory drugs (NSAIDs) like indomethacin. This technology leverages a sophisticated palladium-catalyzed coupling cyclization strategy that utilizes metallic indium as a reducing agent and sodium iodide as a critical additive. By enabling the direct construction of the indole skeleton from readily available o-iodoaniline and propargyl bromide derivatives in a single operational step, this invention addresses significant bottlenecks in traditional synthetic routes. The method demonstrates exceptional functional group compatibility, allowing for the introduction of diverse substituents at multiple positions on the indole ring without compromising yield or purity. For R&D directors and process chemists, this represents a paradigm shift towards more atom-economical and streamlined manufacturing processes for high-value pharmaceutical intermediates.
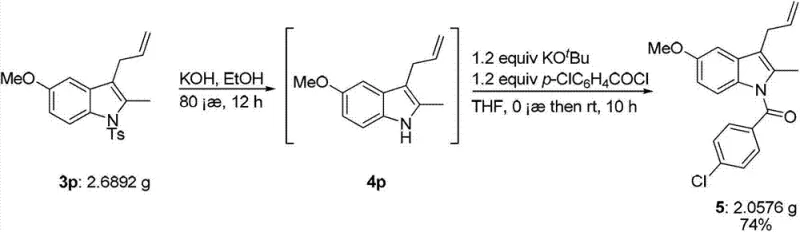
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted indole frameworks has relied heavily on classical methodologies such as the Fisher indole synthesis or various transition-metal catalyzed cyclizations that often suffer from significant drawbacks. Traditional Fisher synthesis typically requires harsh acidic conditions and high temperatures, which can lead to the decomposition of sensitive functional groups and result in complex mixtures of regioisomers that are difficult to separate. Furthermore, many existing transition-metal catalyzed methods necessitate the use of expensive ligands, stoichiometric amounts of organometallic reagents, or multi-step sequences that drastically increase the overall cost of goods sold (COGS). These conventional approaches often lack the robustness required for large-scale manufacturing, where reproducibility and safety are paramount. The accumulation of toxic heavy metal residues and the generation of substantial chemical waste further complicate the regulatory approval process for new drug applications, making these older methods less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient palladium and indium co-catalytic system that achieves the formation of the indole core in a single pot. This method employs o-iodoaniline compounds and propargyl bromides as starting materials, which undergo a tandem coupling and cyclization sequence mediated by palladium acetate and trifurylphosphine (TFP) ligands. The use of metallic indium powder as a reducing agent is particularly innovative, as it effectively regenerates the active palladium species while maintaining mild reaction conditions typically between 50°C and reflux temperatures. This one-step strategy not only simplifies the operational workflow but also significantly enhances the overall yield and selectivity of the transformation. As illustrated in the general reaction scheme below, the versatility of this method allows for the accommodation of various electron-withdrawing and electron-donating groups, making it a universal platform for synthesizing diverse indole libraries.
Mechanistic Insights into Pd/In-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a intricate interplay between palladium catalysis and indium-mediated reduction. Initially, the palladium catalyst undergoes oxidative addition with the o-iodoaniline substrate to form an aryl-palladium intermediate. Subsequently, the propargyl bromide participates in the cycle, likely through a carbopalladation or allylic substitution mechanism, facilitated by the unique electronic properties of the trifurylphosphine ligand which stabilizes the active catalytic species. Metallic indium plays a dual role here; it acts as a terminal reductant to prevent the accumulation of inactive palladium black and may also participate in the transmetallation steps to facilitate the carbon-carbon bond formation. The presence of sodium iodide as an additive is crucial, as it likely assists in the halogen exchange processes, enhancing the reactivity of the organic halides and promoting the smooth progression of the catalytic cycle. This synergistic effect ensures that the reaction proceeds with high turnover numbers, minimizing the required loading of the precious palladium catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or harsh acid-catalyzed pathways. The mild nature of the Pd/In system suppresses common side reactions such as polymerization of the alkyne moiety or over-alkylation of the indole nitrogen. The high specificity of the oxidative addition step ensures that the reaction occurs exclusively at the iodine position, preventing unwanted substitution on other parts of the aromatic ring. Furthermore, the use of indium, which forms water-soluble salts upon workup, simplifies the purification process significantly compared to methods using tin or zinc reagents which often leave stubborn residues. This clean reaction profile translates directly into higher purity of the crude product, reducing the burden on downstream purification units and ensuring that the final pharmaceutical intermediate meets stringent quality standards required by global regulatory bodies.
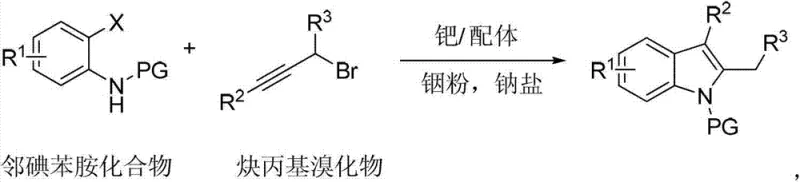
How to Synthesize Polysubstituted Indoles Efficiently
The practical implementation of this synthesis route is designed for ease of operation in standard laboratory and pilot plant settings. The protocol begins with the preparation of a dry reaction vessel under an inert atmosphere to prevent oxidation of the sensitive indium powder and palladium catalyst. Key to the success of this reaction is the precise order of addition and the maintenance of anhydrous conditions during the initial mixing phase. The reaction typically proceeds in polar aprotic solvents such as DMF or NMP, which solubilize both the inorganic salts and organic substrates effectively. Following the reaction period, a straightforward aqueous workup involving extraction with organic solvents like diethyl ether or ethyl acetate allows for the isolation of the target indole derivatives. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for different substrates, are provided in the guide below to ensure reproducible results for your process development teams.
- Prepare the reaction mixture by combining indium powder, sodium iodide, and dry DMF in a reactor under inert atmosphere.
- Add propargyl bromide derivative and stir, followed by the addition of o-iodoaniline, palladium acetate, and trifurylphosphine ligand.
- Heat the mixture to 100°C for 12 hours, then purify the resulting polysubstituted indole via aqueous workup and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the accessibility and cost-effectiveness of the raw materials; o-iodoanilines and propargyl bromides are commodity chemicals available from multiple global suppliers, mitigating the risk of supply chain disruptions associated with exotic or proprietary reagents. By consolidating what was traditionally a multi-step sequence into a single efficient operation, manufacturers can significantly reduce the consumption of solvents, energy, and labor hours, leading to a drastic simplification of the production schedule. This efficiency gain directly correlates to a reduction in the overall manufacturing footprint and a faster time-to-market for new drug candidates relying on these indole scaffolds. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, ensuring consistent output and reliable delivery timelines for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric organometallic reagents and the use of low-loading palladium catalysts driven by the indium regeneration cycle substantially lower the direct material costs. Additionally, the simplified one-pot procedure reduces the need for intermediate isolation and purification steps, which are often the most costly phases in pharmaceutical manufacturing due to solvent usage and yield losses. This streamlined approach allows for a more competitive pricing structure for the final active pharmaceutical ingredient (API) or intermediate, providing a clear margin advantage in a price-sensitive market.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like indium powder and sodium iodide ensures that inventory management is straightforward and less prone to degradation issues compared to sensitive pyrophoric reagents used in alternative methods. The demonstrated scalability of the process, evidenced by successful gram-scale experiments yielding over 2 grams of product with high consistency, suggests a smooth path to kilogram and ton-scale production without the need for extensive re-optimization. This predictability allows supply chain planners to forecast production capacities with greater accuracy and commit to long-term supply agreements with confidence.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste, primarily consisting of indium salts which are easier to treat and dispose of compared to heavy metal sludge from other catalytic systems. The use of DMF, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. The high atom economy of the coupling reaction aligns with green chemistry principles, helping companies meet increasingly strict environmental regulations and sustainability goals. This environmental compliance reduces the risk of regulatory fines and enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polysubstituted indole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's performance and applicability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. We encourage stakeholders to review these insights to fully appreciate the value proposition of this advanced synthetic methodology.
Q: What are the key advantages of this indole synthesis method over traditional Fisher synthesis?
A: This method utilizes a palladium-catalyzed coupling cyclization that operates under milder conditions with superior functional group tolerance, avoiding the harsh acidic environments typical of Fisher synthesis which can degrade sensitive substrates.
Q: Is this process scalable for commercial production of indomethacin intermediates?
A: Yes, the patent explicitly demonstrates gram-scale synthesis (e.g., 2.6g scale in Example 15) with consistent yields around 76%, indicating strong potential for kilogram-to-ton scale-up in industrial settings.
Q: How does the use of metallic indium impact the cost and purity of the final product?
A: Metallic indium acts as an efficient reducing agent that facilitates the catalytic cycle without introducing heavy metal contaminants that are difficult to remove, thereby simplifying downstream purification and ensuring high-purity specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert process chemists has thoroughly analyzed this patented technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex indole intermediates, ensuring that every batch meets the highest international standards. Our commitment to quality and efficiency makes us an ideal partner for companies looking to secure a stable supply of high-purity pharmaceutical intermediates for NSAID production and beyond.
We invite you to collaborate with us to leverage this innovative synthesis method for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data for our indole derivatives and to discuss route feasibility assessments for your target molecules. Let us help you accelerate your development timeline and achieve commercial success with our reliable supply chain solutions.
