Revolutionizing 4,4'-Diaminodiphenyl Ether Production: A One-Step Room Temperature Catalytic Strategy
Introduction to Advanced Monomer Synthesis
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by the need for greener, more efficient synthetic routes. Patent CN114085158A introduces a groundbreaking methodology for the production of 4,4'-diaminodiphenyl ether, a critical monomer for polyimides and advanced engineering plastics. This invention addresses long-standing industrial challenges by replacing multi-step, harsh chemical processes with a streamlined one-step catalytic reaction. By utilizing diphenyl ether, hydrogen peroxide, and ammonia water in the presence of a specialized copper-ligand system, the process achieves exceptional selectivity and yield under ambient conditions. For R&D directors and procurement strategists, this represents a pivotal shift towards sustainable chemistry that does not compromise on product quality or economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
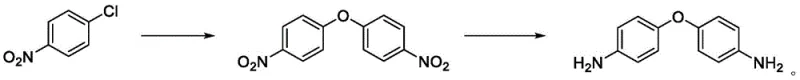
Historically, the industrial synthesis of 4,4'-diaminodiphenyl ether has been dominated by two primary routes, both of which suffer from significant technical and economic drawbacks. The first conventional pathway involves the nitration of diphenyl ether followed by hydrogenation reduction. As illustrated in the reaction scheme below, this method relies heavily on nitric acid, leading to severe environmental pollution and the generation of hazardous waste streams. Furthermore, the electronic effects during nitration often result in the formation of substantial amounts of the 2,2'-dinitrodiphenyl ether isomer. This poor selectivity creates a complex mixture that is difficult and costly to separate, ultimately reducing the overall yield to a range of merely 78-88% and compromising the purity required for high-end polymer applications.

The second traditional route utilizes p-nitrochlorobenzene as a starting material, involving hydrolysis followed by high-pressure hydrogenation. While this method offers better selectivity, it imposes severe constraints on manufacturing infrastructure. The requirement for temperatures exceeding 140°C and high-pressure hydrogenation reactors necessitates substantial capital investment in specialized equipment. Moreover, the reliance on palladium-carbon catalysts introduces significant raw material costs and supply chain vulnerabilities associated with precious metals. The safety risks inherent in high-temperature and high-pressure operations further limit the scalability of this process, making it less attractive for large-scale commercial production where operational expenditure and safety compliance are paramount concerns.
The Novel Approach
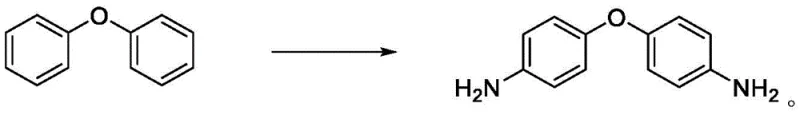
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated copper-catalyzed oxidative amination strategy. This one-step process transforms diphenyl ether directly into 4,4'-diaminodiphenyl ether using hydrogen peroxide as a green oxidant and ammonia water as the nitrogen source. The reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or pressurized vessels. The core innovation lies in the synergistic interaction between the copper salt catalyst and a specifically designed Mannich base ligand. This system facilitates an oxidative bromination of the diphenyl ether to form a 4,4'-dibromo intermediate in situ, which subsequently undergoes C-N coupling with ammonia. The result is a highly selective transformation that bypasses the formation of ortho-isomers entirely, delivering product purities exceeding 99% with yields reaching up to 96%.
Mechanistic Insights into Copper-Catalyzed Oxidative Amination
The success of this synthetic route hinges on the precise engineering of the catalytic cycle, particularly the role of the Mannich base ligand. Unlike conventional ligands such as 1,10-phenanthroline, which fail to drive this specific transformation, the Mannich base ligand possesses unique electronic and steric properties. Its strong electron-donating capability enhances the nucleophilicity of the copper center, facilitating the activation of the C-H bond in diphenyl ether. Simultaneously, the bulky steric environment created by the ligand shields the active site, effectively preventing the attack at the ortho-position. This steric control is the mechanistic basis for the observed high regioselectivity, ensuring that bromination and subsequent amination occur exclusively at the para-positions. Without this specific ligand architecture, the reaction either fails to initiate or produces negligible amounts of the desired product, as evidenced by comparative experiments where standard ligands yielded no detectable product.
Furthermore, the mechanism involves a delicate balance of oxidative and coupling steps mediated by the bromide salt. The bromide acts as a mediator, reacting with hydrogen peroxide to generate an electrophilic brominating species that selectively targets the electron-rich para-positions of the diphenyl ether. This generates the key 4,4'-dibromodiphenyl ether intermediate, which is then rapidly consumed in the copper-catalyzed amination step. This tandem sequence prevents the accumulation of reactive intermediates that could lead to side reactions or polymerization. The use of hydrogen peroxide as the terminal oxidant ensures that the only byproduct is water, aligning the process with green chemistry principles. This mechanistic elegance translates directly to simplified downstream processing, as the absence of complex byproduct mixtures allows for straightforward purification via simple crystallization.
How to Synthesize 4,4'-Diaminodiphenyl Ether Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to minimize operational complexity while maximizing output. The process begins with the charging of a standard reaction vessel with diphenyl ether, a suitable solvent such as tetrahydrofuran or dichloromethane, and the requisite amounts of ammonia water and bromide salt. The addition of the copper catalyst and the proprietary Mannich base ligand initiates the catalytic cycle upon the introduction of hydrogen peroxide. The reaction is maintained at room temperature with vigorous stirring, typically completing within 6 to 12 hours depending on the specific catalyst loading and solvent choice. Following the reaction, the workup involves a simple phase separation and water wash to remove inorganic salts, followed by solvent recovery and crystallization from methanol to yield the high-purity final product.
- Charge a reaction vessel with diphenyl ether, a solvent such as tetrahydrofuran or dichloromethane, ammonia water, a bromide salt, a copper salt catalyst, and a specific Mannich base ligand.
- Add hydrogen peroxide as the oxidant and maintain the reaction mixture at room temperature with vigorous stirring for 6 to 12 hours to ensure complete conversion.
- Upon completion, perform liquid separation and water washing, recover the solvent from the organic layer, and crystallize the residue using methanol to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond mere technical feasibility. The shift from a multi-step, high-energy process to a one-step, ambient temperature reaction fundamentally alters the cost structure of 4,4'-diaminodiphenyl ether manufacturing. By eliminating the need for high-pressure autoclaves and expensive noble metal catalysts like palladium, the capital expenditure (CAPEX) required for plant setup is drastically reduced. Furthermore, the operational expenditure (OPEX) benefits from the use of commodity chemicals—diphenyl ether, ammonia, and hydrogen peroxide—which are widely available and subject to stable pricing, unlike specialized nitrating agents or precious metals that are prone to market volatility.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the process flow. Traditional methods require distinct units for nitration, separation, and high-pressure hydrogenation, each adding layers of cost and complexity. In contrast, this one-pot synthesis consolidates these steps into a single reactor, significantly reducing utility consumption and labor costs. The elimination of the palladium catalyst removes a major cost driver and the associated expense of metal recovery or disposal. Additionally, the high selectivity of the reaction minimizes raw material waste, ensuring that a greater proportion of the input feedstock is converted into saleable product, thereby enhancing overall process efficiency and margin potential.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on readily available bulk chemicals mitigates the risk of raw material shortages. Conventional routes often depend on specific nitrated intermediates or chlorinated precursors that may face supply constraints or regulatory scrutiny. By utilizing diphenyl ether and ammonia, manufacturers can leverage established, robust supply chains that are less susceptible to disruption. The mild reaction conditions also reduce the logistical burden of transporting hazardous high-pressure gases or corrosive acids, simplifying compliance with transportation regulations and lowering insurance premiums. This stability ensures a consistent supply of high-purity monomers to downstream polymer producers, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The scalability of this process is inherently superior due to the absence of thermal and pressure hazards. Scaling up exothermic nitration reactions or high-pressure hydrogenations requires rigorous safety engineering and often faces regulatory hurdles. Conversely, running a reaction at room temperature allows for larger batch sizes with minimal additional safety infrastructure. Environmentally, the process generates significantly less hazardous waste. The replacement of nitric acid with hydrogen peroxide means that the primary effluent is water, drastically reducing the load on wastewater treatment facilities. This alignment with increasingly stringent environmental regulations future-proofs the manufacturing asset, avoiding potential fines and shutdowns associated with non-compliant legacy processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits and operational parameters of the new method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement specialists assessing the long-term value proposition of suppliers adopting this technology.
Q: Why is the Mannich base ligand critical for this synthesis?
A: The specific Mannich base ligand provides strong electron-donating capability and significant steric hindrance. This coordination with the copper salt drastically improves catalytic activity and selectivity, preventing the formation of unwanted isomers that plague conventional nitration routes.
Q: How does this method compare to traditional nitration-hydrogenation routes in terms of safety?
A: Unlike traditional methods that require high temperatures, high pressures, and hazardous nitric acid, this novel process operates at room temperature and atmospheric pressure. This eliminates the need for expensive high-pressure autoclaves and significantly reduces safety risks associated with exothermic nitration reactions.
Q: What represents the primary cost advantage of this new catalytic system?
A: The primary cost advantage stems from the elimination of noble metal catalysts like palladium and the removal of energy-intensive high-pressure hydrogenation steps. Additionally, the use of commodity chemicals like hydrogen peroxide and ammonia water further drives down raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Diaminodiphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN114085158A are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific requirements of this copper-catalyzed process, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity 4,4'-diaminodiphenyl ether that meets the exacting standards of the polyimide and advanced materials industries.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective supply of this critical monomer for your next-generation polymer projects.
