Advanced Decarbonylative Coupling for High-Purity Aryl Alkyne Commercial Manufacturing
Advanced Decarbonylative Coupling for High-Purity Aryl Alkyne Commercial Manufacturing
The landscape of organic synthesis is undergoing a transformative shift towards greener, more sustainable methodologies, particularly in the production of high-value building blocks for the pharmaceutical and materials sectors. A pivotal development in this arena is documented in Chinese Patent CN115557824A, which discloses a novel class of aryl alkyne compounds and their preparation methods. This technology represents a significant departure from traditional cross-coupling protocols by utilizing aryl carboxylic acids as the electrophilic coupling partners instead of toxic aryl halides. For R&D directors and procurement specialists seeking a reliable aryl alkyne supplier, this patent offers a compelling pathway to access complex molecular architectures with enhanced environmental profiles. The method employs a sophisticated palladium-catalyzed decarbonylative coupling strategy that not only simplifies the synthetic route but also drastically reduces the generation of hazardous halide waste streams. By leveraging abundant and stable carboxylic acid feedstocks, this innovation addresses critical supply chain vulnerabilities associated with halogenated starting materials while maintaining high reaction efficiency and product purity standards required for downstream drug discovery applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the carbon-carbon triple bond in aryl alkyne scaffolds has been dominated by the Sonogashira cross-coupling reaction, which couples aryl halides with terminal alkynes. While robust, this conventional approach suffers from inherent drawbacks that pose significant challenges for modern green chemistry initiatives and cost reduction in pharmaceutical intermediates manufacturing. The primary limitation lies in the reliance on aryl iodides or bromides, which are often expensive, light-sensitive, and generate stoichiometric amounts of toxic halide salts as byproducts. Furthermore, the purification of final products from residual heavy metals and halide impurities requires rigorous and costly downstream processing steps, including extensive washing and chromatography. From a supply chain perspective, the volatility and potential instability of certain poly-halogenated precursors can lead to logistical complications and increased storage costs. Additionally, the atom economy of traditional Sonogashira reactions is suboptimal due to the loss of the halogen atom, contributing to a higher E-factor and greater environmental burden. These factors collectively drive up the total cost of ownership for manufacturers relying on legacy halide-based technologies, creating a strong imperative for alternative synthetic strategies.
The Novel Approach
In stark contrast to halide-dependent methodologies, the technology described in Patent CN115557824A introduces a paradigm-shifting route that utilizes aryl carboxylic acids and terminal alkynes as the primary raw materials. This decarbonylative coupling protocol effectively bypasses the need for pre-functionalized halides, tapping into a vast reservoir of commercially available and inexpensive benzoic acid derivatives. The reaction proceeds under the synergistic action of a palladium catalyst, specific phosphine ligands, and acid anhydride additives in organic solvents, facilitating the extrusion of carbon monoxide and the formation of the desired C-C triple bond. 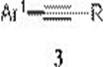 As illustrated in the reaction scheme, the transformation converts readily accessible carboxylic acids directly into valuable aryl alkyne motifs with high atom efficiency regarding the carbon skeleton retention. This approach not only eliminates the toxicity concerns associated with halogenated reagents but also streamlines the workflow by removing the need for harsh activation steps typically required for carboxylic acids in other contexts. The operational simplicity, combined with the use of stable solid reagents, makes this method exceptionally attractive for scaling up complex organic intermediates in an industrial setting, offering a clear competitive advantage in terms of safety and regulatory compliance.
As illustrated in the reaction scheme, the transformation converts readily accessible carboxylic acids directly into valuable aryl alkyne motifs with high atom efficiency regarding the carbon skeleton retention. This approach not only eliminates the toxicity concerns associated with halogenated reagents but also streamlines the workflow by removing the need for harsh activation steps typically required for carboxylic acids in other contexts. The operational simplicity, combined with the use of stable solid reagents, makes this method exceptionally attractive for scaling up complex organic intermediates in an industrial setting, offering a clear competitive advantage in terms of safety and regulatory compliance.
Mechanistic Insights into Palladium-Catalyzed Decarbonylative Coupling
The success of this synthetic methodology hinges on the intricate interplay between the palladium center and the carefully selected ligand system, which orchestrates the challenging decarbonylation process. Mechanistically, the reaction is believed to initiate with the oxidative addition of the aryl carboxylic acid derivative, activated in situ by the acid anhydride, to the low-valent palladium(0) species. This step generates an acyl-palladium intermediate, which subsequently undergoes a crucial decarbonylation event to release carbon monoxide and form an aryl-palladium species. The presence of bulky, electron-rich phosphine ligands, such as 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos), is critical for stabilizing the active catalytic species and promoting the reductive elimination step that forms the final alkyne product.  The structural diversity of the substrates, as shown in the reactant diagrams, indicates that the catalytic system tolerates a wide range of electronic and steric environments on both the aryl ring and the alkyne terminus. This robustness is essential for R&D teams aiming to synthesize libraries of analogs for structure-activity relationship studies without encountering frequent reaction failures. Furthermore, the mechanism inherently minimizes the formation of homocoupling byproducts, a common issue in alkyne chemistry, thereby ensuring a cleaner impurity profile. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as temperature and catalyst loading, to maximize throughput while maintaining stringent quality control standards.
The structural diversity of the substrates, as shown in the reactant diagrams, indicates that the catalytic system tolerates a wide range of electronic and steric environments on both the aryl ring and the alkyne terminus. This robustness is essential for R&D teams aiming to synthesize libraries of analogs for structure-activity relationship studies without encountering frequent reaction failures. Furthermore, the mechanism inherently minimizes the formation of homocoupling byproducts, a common issue in alkyne chemistry, thereby ensuring a cleaner impurity profile. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as temperature and catalyst loading, to maximize throughput while maintaining stringent quality control standards.
Impurity control is another vital aspect where this mechanism offers distinct advantages over traditional routes. In conventional halide couplings, trace amounts of unreacted aryl halides or dehalogenated byproducts can persist, posing genotoxicity risks that require extensive mitigation strategies. In the decarbonylative pathway, the primary byproducts are carbon monoxide and carboxylic acid derivatives, which are easier to manage and remove during workup. The use of acid anhydrides as activators ensures that the carboxylic acid is efficiently converted into a reactive mixed anhydride species in situ, preventing the accumulation of unreactive starting materials. Moreover, the specific choice of solvents like ethylene glycol dimethyl ether or 1,4-dioxane provides a optimal polarity balance that supports the solubility of polar intermediates while facilitating the precipitation or extraction of non-polar impurities. This inherent cleanliness of the reaction profile translates directly into reduced purification burdens, allowing for higher overall yields and shorter cycle times in a GMP manufacturing environment. For quality assurance teams, this means a more predictable and controllable process capable of consistently delivering high-purity aryl alkyne intermediates that meet the rigorous specifications of the global pharmaceutical market.
How to Synthesize Aryl Alkyne Compounds Efficiently
Implementing this advanced synthesis route requires a precise understanding of the reaction conditions and reagent stoichiometry to ensure reproducibility and optimal yield. The patent outlines a generalized procedure where aryl carboxylic acids and terminal alkynes are combined in a reaction vessel with a palladium catalyst source, such as tris(dibenzylideneacetone)dipalladium, and a bidentate phosphine ligand. The mixture is supplemented with an acid anhydride, which serves as a dehydrating and activating agent, and a base to neutralize acidic byproducts generated during the cycle. The reaction is typically conducted at elevated temperatures ranging from 120 °C to 150 °C for a duration of 12 to 16 hours, depending on the specific substrate reactivity. Following the completion of the reaction, standard aqueous workup and column chromatography techniques are employed to isolate the target aryl alkyne in high purity.
- Combine aryl carboxylic acid and terminal alkyne in an organic solvent such as ethylene glycol dimethyl ether or 1,4-dioxane.
- Add palladium catalyst, phosphine ligand, acid anhydride, and base to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 120-150 °C for 12-16 hours, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this decarbonylative coupling technology presents a multitude of strategic benefits that extend beyond mere technical feasibility. The most immediate impact is observed in the realm of raw material sourcing, where the substitution of expensive and regulated aryl halides with commodity-grade aryl carboxylic acids leads to substantial cost savings. Carboxylic acids are produced on a massive industrial scale for various applications, ensuring a stable and competitive supply market that is less susceptible to the geopolitical fluctuations often affecting halogenated chemical supplies. This shift significantly enhances supply chain reliability, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the elimination of toxic halides simplifies the regulatory compliance landscape, reducing the administrative burden and costs associated with handling hazardous substances and disposing of halide-containing waste streams. The operational simplicity of the process, which does not require specialized high-pressure reactors or cryogenic conditions, also lowers the barrier to entry for contract manufacturing organizations looking to expand their service offerings.
- Cost Reduction in Manufacturing: The economic argument for this technology is robust, driven primarily by the lower unit cost of carboxylic acid starting materials compared to their halide counterparts. By removing the need for expensive halogenation steps or the purchase of premium aryl iodides, manufacturers can achieve a leaner cost structure. Additionally, the high yields reported in the patent examples, often exceeding 80% for a broad range of substrates, mean that less raw material is wasted per kilogram of finished product. The reduction in purification complexity further contributes to cost efficiency by lowering solvent consumption and reducing the load on waste treatment facilities. These cumulative effects result in a more competitive pricing model for the final aryl alkyne intermediates, allowing companies to improve their margins or pass savings on to customers in a highly price-sensitive market.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the pharmaceutical industry, and this synthesis method fortifies the supply of critical intermediates. Aryl carboxylic acids are generally solids with excellent shelf-life stability, unlike many liquid aryl halides that may degrade or require special storage conditions. This stability simplifies logistics and inventory management, allowing for larger batch purchases and longer storage periods without quality degradation. The widespread availability of diverse carboxylic acid building blocks also enables rapid adaptation to changing project requirements, facilitating the quick sourcing of analogs for drug development programs. By diversifying the raw material base away from halogenated compounds, companies can insulate themselves from supply disruptions caused by environmental regulations on halogen chemistry or production issues at specific halide manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden bottlenecks, but this decarbonylative route is inherently designed for scalability. The reaction conditions are mild enough to be managed in standard glass-lined or stainless steel reactors without the need for exotic materials of construction. The absence of toxic halide waste aligns perfectly with increasingly stringent global environmental regulations, such as REACH in Europe, reducing the risk of future compliance penalties. The green chemistry credentials of this method, characterized by atom economy and reduced toxicity, also enhance the corporate sustainability profile of manufacturers, which is becoming a key differentiator in securing contracts with major pharmaceutical clients who prioritize ESG (Environmental, Social, and Governance) criteria in their supplier selection process.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled a set of frequently asked questions based on the detailed disclosures within the patent literature. These inquiries address common concerns regarding substrate compatibility, catalyst efficiency, and process safety, providing a clear picture of what to expect during technology transfer. The answers are derived directly from the experimental data and embodiments provided in the patent, ensuring accuracy and relevance for process development teams.
Q: What are the primary advantages of using aryl carboxylic acids over aryl halides?
A: Aryl carboxylic acids are significantly less toxic, more stable during storage, and often cheaper than corresponding aryl halides, reducing hazardous waste generation.
Q: Which palladium catalysts are most effective for this decarbonylative coupling?
A: The patent highlights the efficacy of Pd2(dba)3, Pd(dba)2, and PdCl2(PPh3)2 when paired with bulky phosphine ligands like Xantphos or dppp.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method operates at moderate temperatures without requiring specialized high-pressure equipment, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Alkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the decarbonylative coupling technology described in Patent CN115557824A for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to seamlessly integrate this innovative synthetic route into our existing manufacturing capabilities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is smooth and efficient. We are committed to delivering high-purity aryl alkyne compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch released conforms to the highest industry standards, providing you with the confidence needed to advance your drug candidates through clinical trials.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis method for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how switching to this carboxylic acid-based route can optimize your budget. Please contact us today to request specific COA data for our catalog of aryl alkyne intermediates or to discuss route feasibility assessments for your custom synthesis projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in your chemical manufacturing operations.
