Advanced Synthesis of Axial Chiral Indole-Naphthalene Compounds for Catalytic Applications
Advanced Synthesis of Axial Chiral Indole-Naphthalene Compounds for Catalytic Applications
The landscape of asymmetric catalysis is continually evolving, driven by the demand for more efficient and sustainable methods to construct complex chiral scaffolds. A significant breakthrough in this domain is detailed in patent CN110452150B, which discloses a novel preparation method for axial chiral indole-naphthalene compounds. These compounds are not merely structural curiosities; they serve as potent organic small-molecule catalysts capable of driving sophisticated transformations such as asymmetric [3+2] cyclizations and allylation reactions. The core innovation lies in the ability to construct the axially chiral indole-naphthalene structure in a single step from racemic starting materials, utilizing a highly effective chiral phosphoric acid catalyst. This approach represents a paradigm shift from traditional resolution methods, offering a streamlined pathway to high-value intermediates that are critical for the development of next-generation pharmaceutical and agrochemical agents.
For procurement managers and supply chain directors seeking a reliable pharmaceutical intermediate supplier, the implications of this technology are profound. The method described eliminates the reliance on scarce transition metals and harsh reaction conditions, replacing them with an organocatalytic system that operates under mild thermal parameters. This transition not only enhances the environmental profile of the manufacturing process but also drastically simplifies the purification workflow. By achieving high optical purity directly from the reaction vessel, the need for extensive downstream processing is minimized, leading to substantial reductions in production time and operational expenditure. As we delve deeper into the technical specifics, it becomes clear that this patent offers a robust solution for the commercial scale-up of complex chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral biaryl compounds, particularly those featuring an indole-naphthalene skeleton, has been fraught with challenges that hinder industrial adoption. Conventional strategies often rely on the direct coupling of an indole ring with a naphthalene ring, a process that frequently suffers from low regioselectivity and poor stereocontrol. Furthermore, many existing literature methods require the use of expensive chiral auxiliaries or stoichiometric amounts of chiral reagents, which inflates the cost of goods sold (COGS) and generates significant chemical waste. Another critical bottleneck is the reliance on transition metal catalysis, which introduces the risk of heavy metal contamination in the final product—a major concern for high-purity API intermediates destined for pharmaceutical use. The removal of these metal residues often necessitates additional purification steps, such as specialized scavenging or repeated crystallizations, which further erodes yield and extends lead times.
The Novel Approach
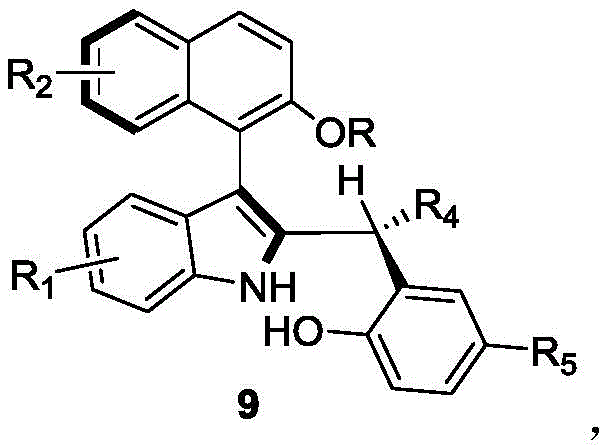
In stark contrast, the methodology outlined in CN110452150B leverages a dynamic kinetic resolution (DKR) strategy mediated by a chiral phosphoric acid catalyst. This innovative approach allows for the conversion of racemic raw materials directly into the desired axially chiral product with exceptional efficiency. The reaction proceeds via an asymmetric addition mechanism, where the chiral catalyst simultaneously activates the electrophile and nucleophile within a well-defined chiral pocket, ensuring high stereoselectivity. As illustrated in the reaction scheme below, the process combines a compound of formula 7 with a compound of formula 8 in a mixed solvent system, yielding the target structure (formula 9) in a single operational step.
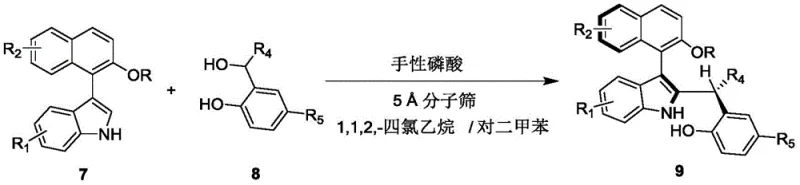
This novel route offers distinct advantages for cost reduction in pharmaceutical intermediate manufacturing. By operating at ambient temperatures (20-30°C) and utilizing readily available organic solvents like 1,1,2,2-tetrachloroethane and p-xylene, the process avoids the energy-intensive requirements of cryogenic cooling or high-pressure reactors. Moreover, the use of 5 Å molecular sieves as an additive helps drive the equilibrium forward by sequestering water, thereby enhancing yield without the need for complex azeotropic distillation setups. The result is a streamlined protocol that is not only chemically elegant but also economically superior, addressing the key pain points of scalability and cost-efficiency that plague traditional synthetic routes.
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
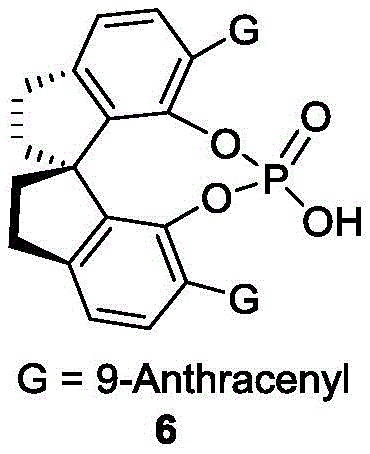
The success of this synthesis hinges on the precise design of the chiral phosphoric acid catalyst, specifically the spiro-binaphthyl skeleton derivatives exemplified by compound of formula 6. These catalysts function through a dual-activation mode, where the acidic proton of the phosphate group hydrogen-bonds with the hydroxyl group of the naphthol-derived substrate, increasing its electrophilicity. Simultaneously, the bulky three-dimensional architecture of the catalyst, often adorned with large substituents like 9-anthracenyl groups, creates a sterically confined environment that dictates the facial selectivity of the nucleophilic attack by the indole moiety. This rigorous spatial control is what enables the formation of the axial chirality with such high fidelity, effectively distinguishing between the enantiomers of the racemic starting material and funneling them into a single chiral product.
From an impurity control perspective, this mechanistic pathway is exceptionally clean. The high diastereomeric ratios (dr >95:5) and enantiomeric ratios (er up to 99.8:0.2 after recrystallization) reported in the patent data indicate that side reactions and non-selective background reactions are effectively suppressed. The catalyst's ability to differentiate between the pro-chiral faces of the substrate minimizes the formation of unwanted atropisomers, which are notoriously difficult to separate due to their similar physical properties. For R&D directors, this level of control translates to a simplified analytical profile and a more robust quality assurance process. The stability of the catalyst under the reaction conditions also ensures consistent performance batch-to-batch, a critical factor when transitioning from laboratory discovery to pilot plant operations.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring standard laboratory equipment and straightforward workup procedures. The process begins with the preparation of a reaction mixture containing the indole-naphthalene precursor (formula 7) and the benzylic alcohol derivative (formula 8) in a specific solvent ratio. The addition of the chiral catalyst and molecular sieves initiates the transformation, which can be monitored via thin-layer chromatography (TLC). Upon completion, the heterogeneous mixture is simply filtered to remove the solid sieves, and the filtrate is concentrated. Final purification is achieved through standard silica gel column chromatography, yielding the target compound as a white solid with high optical purity. This operational simplicity underscores the method's suitability for rapid iteration in process development.
- Prepare the reaction mixture by combining compound of formula 7 and compound of formula 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene.
- Add 5 Å molecular sieves and a chiral phosphoric acid catalyst (such as compound of formula 6) to the mixture.
- Stir the reaction at 20-30°C until completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this organocatalytic route offers compelling strategic benefits. The shift away from transition metal catalysis removes a significant variable from the supply chain equation, eliminating the volatility associated with precious metal pricing and availability. Furthermore, the mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with modern sustainability goals and potentially lowering utility costs. The robustness of the reaction across a wide range of substrates, as evidenced by the diverse R-groups tolerated in the patent examples, ensures that this platform technology can be adapted for the synthesis of various analogues without requiring a complete process redevelopment.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated heavy metal removal steps leads to a direct decrease in raw material and processing costs. Additionally, the high yields (up to 97%) and excellent selectivity minimize the loss of valuable starting materials, maximizing the overall material throughput and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The starting materials (compounds of formula 7 and 8) are synthesized via known, scalable routes or are commercially available, reducing the risk of supply bottlenecks. The use of common organic solvents and ambient temperature conditions further de-risks the supply chain by removing dependencies on specialized cryogenic infrastructure or hazardous high-pressure equipment, ensuring consistent production capabilities.
- Scalability and Environmental Compliance: The process generates minimal waste due to its high atom economy and the use of recyclable molecular sieves. The absence of toxic heavy metals simplifies waste treatment and disposal, facilitating compliance with stringent environmental regulations. This green chemistry profile makes the process highly attractive for large-scale commercial production, where regulatory scrutiny and environmental impact are paramount concerns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these axial chiral compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees.
Q: What is the primary advantage of this synthesis method over traditional coupling reactions?
A: This method utilizes dynamic kinetic resolution of racemic raw materials to construct the axial chiral skeleton in a single step, avoiding the need for complex multi-step resolutions and offering superior atom economy.
Q: What level of optical purity can be achieved with this process?
A: The process achieves high enantioselectivity, with enantiomeric ratios (er) reaching up to 99.8:0.2 after recrystallization, making it suitable for high-value catalytic applications.
Q: Are the reaction conditions suitable for large-scale production?
A: Yes, the reaction operates under mild conditions (20-30°C) without the need for cryogenic cooling or expensive transition metal catalysts, significantly simplifying scale-up and safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN110452150B for the production of advanced chiral catalysts and intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of axial chiral indole-naphthalene meets the exacting standards required for asymmetric catalysis applications. Our infrastructure is designed to handle complex organic syntheses with precision, maintaining the integrity of the chiral axis throughout the manufacturing process.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
