Advanced Synthesis of Red-Emitting Perylene Bisimide Derivatives for High-Performance OLED Displays
Advanced Synthesis of Red-Emitting Perylene Bisimide Derivatives for High-Performance OLED Displays
The rapid evolution of the organic optoelectronics sector demands materials that not only exhibit superior photophysical properties but also possess structural stability and synthetic accessibility. Patent CN113956254A introduces a groundbreaking methodology for constructing organic photoelectric materials based on perylene bisimide (PDI) derivatives. This technology addresses a critical gap in the market where red-light emitting electroluminescent materials are significantly less prevalent than their blue-light counterparts. By utilizing perylene bisimide as a robust construction unit, the inventors have developed a versatile platform capable of fine-tuning energy band structures through strategic side-chain modifications. The resulting compounds demonstrate exceptional fluorescence quantum yields and emission wavelengths predominantly exceeding 600nm, making them ideal candidates for next-generation display technologies and organic light-emitting diodes (OLEDs). 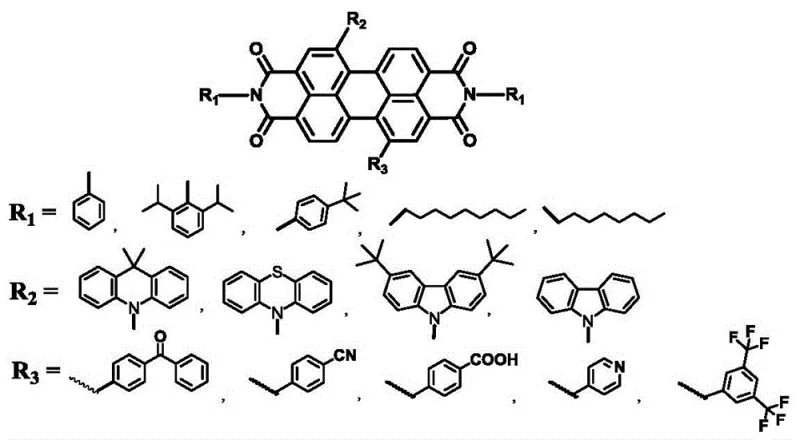
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance organic luminescent materials has been plagued by complex multi-step routes that often require harsh reaction conditions and expensive transition metal catalysts. Traditional methods for modifying the perylene core frequently involve aggressive halogenation or oxidation steps that can degrade the conjugated system, leading to lower yields and inconsistent batch-to-batch quality. Furthermore, many existing protocols rely on solvents and reagents that are difficult to remove completely, resulting in residual impurities that quench fluorescence and reduce device efficiency. The lack of modularity in older synthetic strategies also limits the ability to rapidly screen different push-pull electronic structures, slowing down the R&D cycle for new emissive materials. These factors collectively increase the cost of goods sold and create significant bottlenecks for supply chain managers aiming for consistent commercial production.
The Novel Approach
The patented methodology revolutionizes this landscape by employing a streamlined three-step sequence comprising amidation, bromine substitution, and coupling reactions under remarkably mild conditions. Instead of relying on destructive functionalization, this approach builds complexity onto a stable 1,7-dibromo perylene diimide scaffold, preserving the integrity of the pi-conjugated system. The use of common organic acids like propionic acid as solvents for the initial amidation step eliminates the need for specialized high-pressure equipment, while the subsequent nucleophilic substitution and palladium-catalyzed coupling proceed efficiently in polar aprotic solvents like DMF and DMSO. This modular design allows for the easy introduction of diverse functional groups at the bay positions, enabling precise tuning of the HOMO-LUMO gap without compromising the thermal stability of the core. Consequently, this route offers a direct path to high-purity intermediates with minimal byproduct formation, significantly enhancing the overall process economics.
Mechanistic Insights into the Three-Step Cascade Synthesis
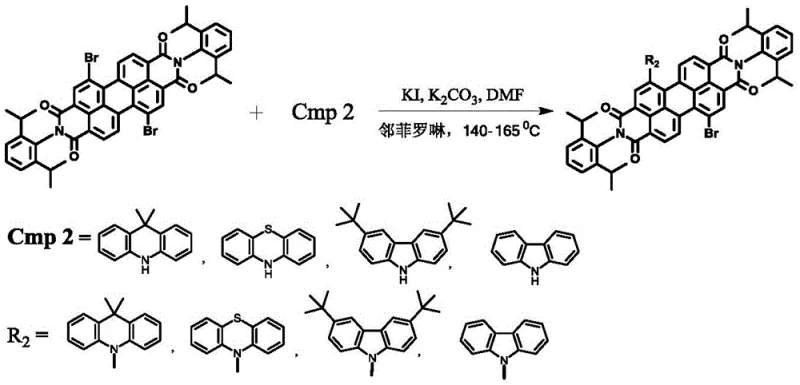
The core of this innovation lies in the sequential functionalization of the perylene backbone, which is meticulously designed to maximize yield and minimize steric hindrance. The first stage involves an amidation reaction where 1,7-dibromo perylene dianhydride reacts with primary amines, such as 2,6-diisopropylaniline, in refluxing propionic acid. This step forms the imide rings that define the PDI structure, locking the core into a planar configuration essential for charge transport. Following this, the second stage employs a nucleophilic aromatic substitution mechanism where electron-rich heterocycles like carbazole derivatives displace the bromine atoms at the bay positions. This reaction is facilitated by potassium carbonate and potassium iodide in DMF at elevated temperatures (140-165°C), effectively extending the conjugation length and introducing hole-transporting characteristics to the molecule. 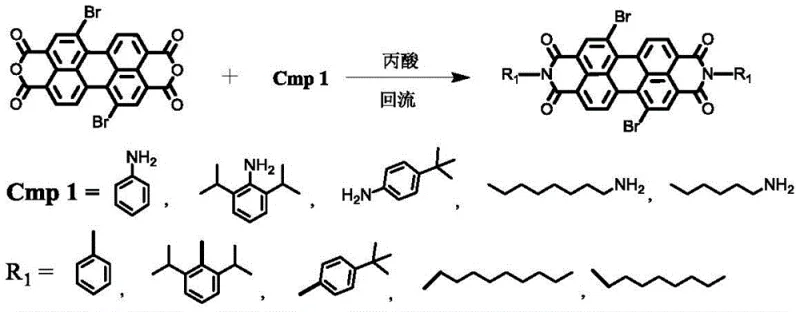
The final transformation utilizes a Suzuki-Miyaura cross-coupling reaction to attach aryl boronate esters to the remaining reactive sites or to further functionalize the structure. Catalyzed by tetrakis(triphenylphosphine)palladium(0) in a DMSO/water mixture, this step allows for the attachment of diverse electron-withdrawing or electron-donating groups, such as benzoyl or pyridine moieties. This late-stage functionalization is crucial for adjusting the emission color and improving the solubility of the final material for solution processing. Throughout these transformations, the reaction conditions are optimized to prevent over-reaction or decomposition, ensuring that the delicate balance of the push-pull electronic system is maintained. The purification protocols, involving recrystallization and column chromatography, are specifically tailored to remove palladium residues and unreacted starting materials, guaranteeing the high purity required for electronic applications. 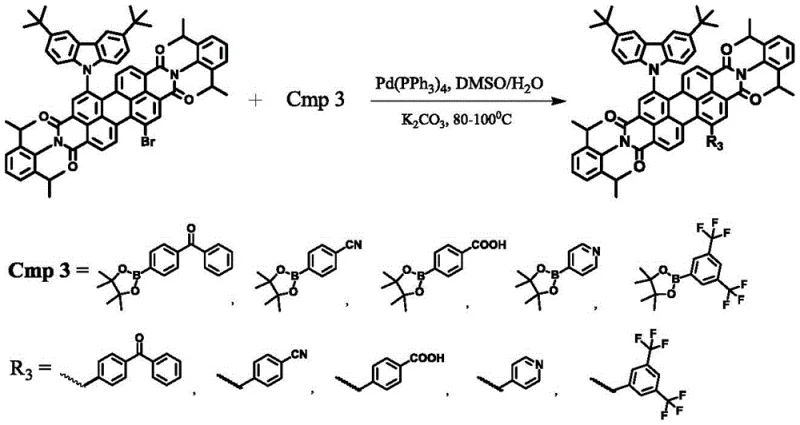
How to Synthesize Perylene Bisimide Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced materials with high reproducibility. The process begins with the careful selection of aniline derivatives to control the solubility and packing of the PDI core, followed by precise temperature control during the substitution steps to avoid side reactions. Operators must adhere to strict nitrogen protection protocols to prevent oxidation of the sensitive intermediates, particularly during the high-temperature DMF reflux. The workup procedures are straightforward, utilizing standard aqueous quenching and filtration techniques that are easily adaptable to large-scale reactors. For detailed operational parameters, stoichiometry, and specific purification gradients, please refer to the standardized guide below.
- Perform amidation reaction between 1,7-dibromo perylene diimide and aniline derivatives in propionic acid at 120-125°C to form PDI-Br.
- Conduct nucleophilic substitution using PDI-Br, carbazole derivatives, and potassium carbonate in DMF at 140-165°C to obtain PDI-KZ.
- Execute Suzuki coupling reaction between PDI-KZ and boronate esters using Pd(PPh3)4 catalyst in DMSO at 80-100°C to finalize the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by leveraging commodity chemicals and minimizing the reliance on exotic reagents. The starting material, 1,7-dibromo perylene dianhydride, is a well-established intermediate with a stable supply chain, reducing the risk of raw material shortages. The solvents used, including propionic acid, DMF, and DMSO, are bulk industrial chemicals that are cost-effective and readily available in most global markets. This accessibility translates directly into lower input costs and greater flexibility in sourcing, allowing procurement managers to negotiate better terms with multiple vendors. Furthermore, the high yields reported in the examples, such as 92% for the initial amidation and 82% for the substitution step, indicate a highly efficient atom economy that reduces waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of simple solvent systems significantly lower the energy consumption and equipment maintenance costs associated with production. By avoiding the need for cryogenic temperatures or ultra-high vacuum systems, the capital expenditure for setting up production lines is drastically reduced. The high conversion rates minimize the loss of expensive perylene precursors, ensuring that the cost per kilogram of the final active material remains competitive. Additionally, the simplified purification process reduces the consumption of silica gel and eluents, further driving down the operational expenses.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, leading to more consistent batch outcomes. This reliability is critical for maintaining continuous supply to downstream OLED manufacturers who cannot afford interruptions in their production schedules. The modular nature of the synthesis also allows for the rapid substitution of alternative amine or boronic acid building blocks if specific supply chains are disrupted, providing a strategic buffer against market volatility. Shorter reaction times compared to traditional methods also mean faster turnaround times from order to delivery.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard oil bath heating and atmospheric pressure reactions which translate easily from laboratory flasks to industrial reactors. The waste streams generated are primarily organic solvents that can be recovered and recycled using standard distillation units, aligning with modern green chemistry principles. The absence of heavy metal contaminants in the final product, due to effective purification, simplifies the regulatory compliance process for electronic materials. This environmental friendliness enhances the corporate social responsibility profile of the supply chain, appealing to eco-conscious end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these perylene bisimide derivatives. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details is essential for R&D teams evaluating the material for specific device architectures and for procurement officers assessing the feasibility of long-term contracts.
Q: What are the key advantages of this perylene bisimide synthesis route?
A: The patented method offers mild reaction conditions, simple operational procedures, and low equipment requirements. It utilizes readily available raw materials and achieves high yields, making it highly suitable for industrial scale-up compared to traditional harsh methods.
Q: What is the emission characteristic of the synthesized material?
A: Unlike many reported materials that focus on blue light emission, this invention specifically targets red light emission with wavelengths mostly larger than 600nm. The materials exhibit high fluorescence quantum yields and excellent light stability.
Q: How is purity ensured in the final product?
A: The process incorporates rigorous purification steps including vacuum filtration, recrystallization with ethanol, and column chromatography using specific mobile phases like dichloromethane/petroleum ether or dichloromethane/methanol to remove impurities effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perylene Bisimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these red-emitting perylene bisimide derivatives in the field of organic electronics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass market adoption is seamless. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature profiles required for this synthesis, while our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards of the semiconductor industry. We are committed to delivering high-purity OLED material that performs consistently in your devices.
We invite you to collaborate with us to optimize this synthetic route for your specific volume requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project, identifying opportunities to further streamline the process. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly growing optoelectronics market.
