Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Advanced Drug Discovery
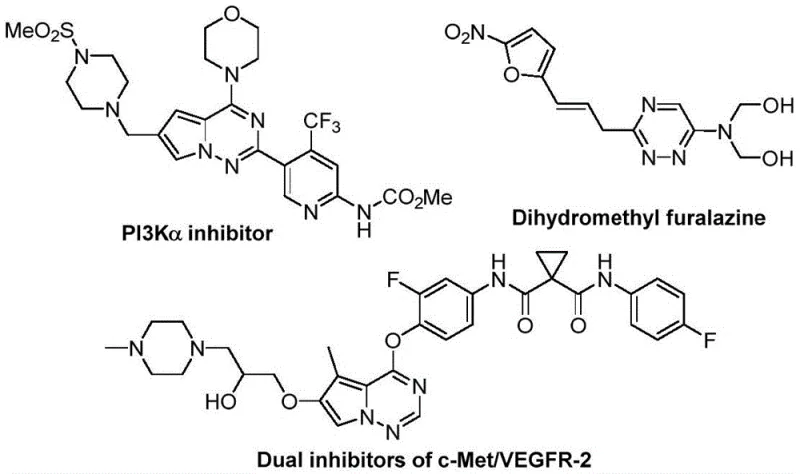
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methodologies to access nitrogen-rich heterocyclic scaffolds that possess enhanced metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN116253692A, which discloses a novel preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, base-promoted cycloaddition strategy. By leveraging cheap and readily available starting materials such as chlorohydrazones and trifluoroacetyl thio ylides, this invention addresses critical pain points in modern process chemistry, specifically regarding cost, safety, and environmental impact. For R&D directors and procurement managers alike, understanding the nuances of this metal-free pathway is essential for optimizing supply chains and reducing the overall cost of goods for complex active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied heavily on condensation reactions between amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical methods are well-documented, they suffer from inherent inefficiencies that hinder their application in modern, high-throughput drug discovery and large-scale manufacturing. Conventional protocols often necessitate harsh reaction conditions, including elevated temperatures and strictly anhydrous environments, which increase energy consumption and operational complexity. Furthermore, many existing routes exhibit poor structural diversity, limiting the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR) around the triazine core. Perhaps most critically for industrial applications, traditional methods frequently rely on stoichiometric amounts of reagents that generate significant waste or require difficult purification steps to remove toxic byproducts, thereby inflating the process mass intensity (PMI) and complicating regulatory compliance.
The Novel Approach
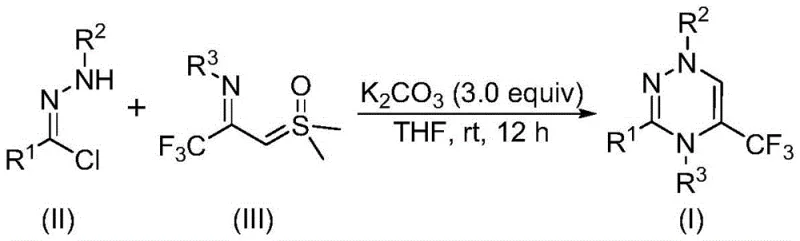
In stark contrast to these legacy techniques, the methodology described in CN116253692A introduces a streamlined [3+3] cycloaddition strategy that operates under remarkably mild conditions. The core innovation lies in the synergistic reaction between a chlorohydrazone and a trifluoroacetyl sulfur ylide, promoted simply by potassium carbonate in an organic solvent. This approach eliminates the need for expensive and potentially toxic transition metal catalysts, such as palladium or copper, which are often difficult to remove to the stringent parts-per-million levels required for pharmaceutical products. The reaction proceeds efficiently at room temperature (20-40°C) and, crucially, tolerates an air atmosphere, removing the need for costly inert gas protection systems. This operational simplicity not only accelerates the reaction timeline but also significantly lowers the barrier to entry for scale-up, making it an attractive option for both laboratory synthesis and commercial production of high-value intermediates.
Mechanistic Insights into Base-Promoted [3+3] Cycloaddition
To fully appreciate the robustness of this synthetic route, one must delve into the mechanistic underpinnings that drive the formation of the trifluoromethyl-substituted 1,2,4-triazine ring. The process initiates with the deprotonation of the chlorohydrazone precursor by the mild inorganic base, potassium carbonate. This deprotonation triggers the elimination of hydrogen chloride, generating a highly reactive nitrile imine intermediate in situ. This 1,3-dipole is then poised to engage in a concerted cycloaddition with the trifluoroacetyl sulfur ylide, which acts as the dipolarophile. The interaction between these two species facilitates the formation of the six-membered triazine ring through a synergistic [3+3] cyclization event. Following the ring closure, the elimination of dimethyl sulfoxide (DMSO) drives the equilibrium forward, yielding the final aromatic heterocycle. This mechanism is distinct from stepwise nucleophilic additions, offering a more direct and atom-economical pathway to the target scaffold.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring high product purity. High-temperature cyclizations often lead to polymerization or decomposition of sensitive functional groups, resulting in complex impurity profiles that are challenging to separate. By maintaining the reaction at ambient temperatures, this method minimizes thermal degradation and side reactions, leading to cleaner crude reaction mixtures. Furthermore, the use of potassium carbonate, a non-nucleophilic weak base, prevents unwanted nucleophilic attacks on other electrophilic sites within the substrate molecules. This selectivity is particularly advantageous when synthesizing libraries of compounds with diverse functional groups, as it ensures that the integrity of sensitive moieties—such as esters, halides, or ethers—is preserved throughout the transformation, thereby simplifying downstream purification and enhancing overall yield.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and mixing efficiency, although the protocol itself is designed for simplicity. The standard procedure involves dissolving the chlorohydrazone and trifluoroacetyl sulfur ylide in a suitable aprotic solvent, with tetrahydrofuran (THF) being the preferred medium due to its ability to solubilize both organic precursors and the inorganic base effectively. The addition of potassium carbonate serves as the driving force for the reaction, and maintaining a slight excess of the base ensures complete conversion of the starting materials. The reaction mixture is then stirred at room temperature for a period of 10 to 14 hours, allowing sufficient time for the cycloaddition and subsequent elimination steps to reach completion. Detailed standardized synthesis steps follow below.
- Combine potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike methods requiring specialized organometallic catalysts that may have long lead times or geopolitical supply risks, the reagents for this process—chlorohydrazones, sulfur ylides, and potassium carbonate—are commodity chemicals available from multiple global suppliers. This redundancy in sourcing mitigates the risk of production stoppages due to raw material shortages. Additionally, the elimination of heavy metals removes the necessity for expensive scavenging resins or complex filtration systems typically required to meet residual metal specifications in APIs. This reduction in downstream processing steps translates directly into lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry mandates.
- Cost Reduction in Manufacturing: The economic implications of adopting this metal-free protocol are profound. By removing the reliance on precious metal catalysts, manufacturers can avoid the volatile pricing associated with commodities like palladium or rhodium. Furthermore, the ability to run the reaction under air at room temperature significantly reduces energy costs associated with heating, cooling, and nitrogen blanketing. The simplified workup procedure, which often involves basic filtration and chromatography rather than complex extractions or distillations, reduces labor hours and solvent consumption. These cumulative efficiencies result in substantial cost savings per kilogram of produced intermediate, enhancing the overall margin profile for the final drug product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance for air and moisture means that the process is less susceptible to failures caused by equipment leaks or imperfect inerting, which are common causes of batch rejection in sensitive organometallic chemistry. The use of stable, shelf-stable starting materials further ensures that inventory can be held for longer periods without degradation, allowing for better demand forecasting and stock management. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently scalable due to its exothermic neutrality and lack of hazardous reagents. The absence of toxic heavy metals simplifies waste stream management, reducing the costs and regulatory burdens associated with hazardous waste disposal. The reaction generates benign byproducts like potassium chloride and DMSO, which are easier to treat or recycle compared to heavy metal sludge. This alignment with environmental, social, and governance (ESG) goals makes the process attractive for companies aiming to reduce their carbon footprint and adhere to stricter environmental regulations without compromising on production volume or speed.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes inexpensive potassium carbonate as a promoter, completely eliminating the need for costly heavy metal catalysts and subsequent removal steps.
Q: What are the optimal reaction conditions for scalability?
A: The reaction proceeds efficiently at room temperature (20-40°C) in an air atmosphere using THF as the solvent, making it highly suitable for large-scale manufacturing without specialized inert gas equipment.
Q: What is the structural diversity achievable with this method?
A: The method supports a wide range of substituents including alkyl, phenyl, naphthyl, and furyl groups with various electron-donating or withdrawing groups, allowing for extensive library generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of trifluoromethyl triazine intermediate meets the highest global standards for pharmaceutical use. We understand that consistency and quality are paramount in the supply of critical intermediates, and our team is dedicated to delivering products that facilitate your regulatory filings and clinical trials.
We invite you to collaborate with us to leverage this innovative chemistry for your specific pipeline needs. Whether you require custom synthesis of novel analogs or bulk manufacturing of established intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your development timelines and optimize your manufacturing costs.
