Scalable Synthesis of Trifluoromethyl-1,2,4-Triazines for Advanced Pharmaceutical Applications
Scalable Synthesis of Trifluoromethyl-1,2,4-Triazines for Advanced Pharmaceutical Applications
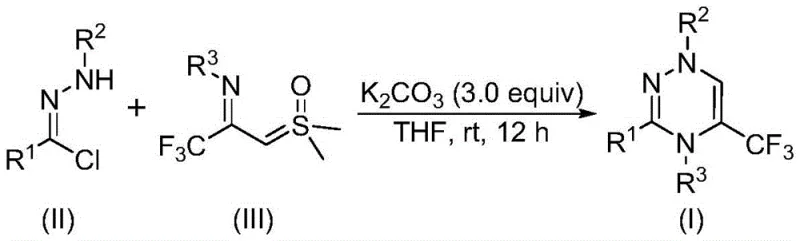
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current synthetic workflows. This technology leverages a novel cycloaddition strategy that bypasses the need for transition metal catalysis, offering a greener and more economically viable pathway for producing high-value intermediates. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline the supply chain for complex heterocyclic scaffolds used in anticancer, antifungal, and antihypertensive drug development. The ability to introduce a trifluoromethyl group efficiently while maintaining structural diversity is a key differentiator in modern medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied on classical condensation reactions involving amidrazones and 1,2-diketones or alkynes, often requiring stringent conditions and multiple steps. Traditional multicomponent reactions utilizing hydrazides and dicarbonyl compounds frequently suffer from poor atom economy and limited functional group tolerance, leading to complex purification challenges. Furthermore, many existing protocols necessitate the use of expensive transition metal catalysts or harsh acidic/basic environments that can degrade sensitive substrates. These limitations result in lower overall yields, higher production costs due to extensive downstream processing, and significant environmental burdens associated with heavy metal waste disposal. For supply chain heads, these factors translate into longer lead times and increased volatility in the availability of critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN116253692A utilizes a direct [3+3] cycloaddition between chlorohydrazones and trifluoroacetyl thio ylides, promoted by simple inorganic bases. This innovative route operates under remarkably mild conditions, specifically at temperatures between 20°C and 40°C in an ambient air atmosphere, eliminating the need for energy-intensive heating or inert gas protection. The use of potassium carbonate as a cheap, non-toxic promoter not only reduces raw material costs but also simplifies the workup procedure significantly. By avoiding heavy metal catalysts entirely, the process ensures a cleaner impurity profile, which is paramount for regulatory compliance in API manufacturing. This approach exemplifies a shift towards sustainable chemistry that does not compromise on efficiency or product quality.
Mechanistic Insights into Metal-Free [3+3] Cycloaddition
The mechanistic pathway of this transformation is both elegant and efficient, driven by the in situ generation of highly reactive intermediates. Under the promotion of potassium carbonate, the chlorohydrazone precursor undergoes dehydrohalogenation to form a nitrile imine intermediate, a crucial 1,3-dipole species. This reactive intermediate then engages in a synergistic [3+3] cycloaddition with the trifluoroacetyl thio ylide, which acts as the dipolarophile. The reaction proceeds through a concerted mechanism that constructs the six-membered triazine ring while simultaneously eliminating a molecule of dimethyl sulfoxide. This cascade sequence avoids the formation of stable byproducts that typically complicate purification, ensuring a high degree of convergence in the synthetic design. The absence of metal coordination steps further reduces the risk of metal-induced side reactions or catalyst poisoning.
From an impurity control perspective, this mechanism offers distinct advantages for process chemists aiming for high-purity outputs. Since the reaction does not involve radical pathways or unstable organometallic species, the formation of unpredictable side products is minimized. The primary byproduct, dimethyl sulfoxide, is highly soluble in aqueous workups and easily removed, leaving the target triazine compound relatively pure even before chromatographic purification. The structural integrity of sensitive functional groups, such as halogens or ethers on the aromatic rings, is preserved throughout the process due to the mild basicity of potassium carbonate. This selectivity allows for the late-stage functionalization of complex molecules, providing R&D teams with greater flexibility in designing structure-activity relationship (SAR) studies without worrying about protecting group strategies.
How to Synthesize Trifluoromethyl-Substituted 1,2,4-Triazines Efficiently
The operational simplicity of this protocol makes it accessible for both laboratory-scale optimization and pilot plant operations. The standard procedure involves mixing the chlorohydrazone and trifluoroacetyl sulfur ylide in a polar aprotic solvent like tetrahydrofuran (THF), followed by the addition of potassium carbonate. The reaction mixture is stirred at room temperature for approximately 10 to 14 hours, after which conversion is typically complete. Detailed standardized synthesis steps, including precise molar ratios and purification parameters, are outlined in the technical guide below to ensure reproducibility across different manufacturing sites.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran (THF).
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to allow the cycloaddition to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl-substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of precious metal catalysts removes a major cost driver and supply risk associated with fluctuating prices of palladium or rhodium. Additionally, the reliance on commodity chemicals like potassium carbonate and widely available organic solvents ensures a stable and resilient supply chain, unaffected by the geopolitical constraints often seen with specialized reagents. The mild reaction conditions also reduce energy consumption and infrastructure requirements, allowing for production in facilities that may not be equipped for high-pressure or high-temperature processes. This flexibility enhances the overall agility of the manufacturing network.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive catalytic systems and the simplification of purification workflows. Without the need for metal scavengers or complex extraction protocols to remove trace metals, the downstream processing costs are drastically reduced. The high atom economy of the cycloaddition reaction means less raw material is wasted, directly improving the cost-per-kilogram metric for the final intermediate. Furthermore, the use of inexpensive inorganic bases instead of organic amines or strong bases lowers the recurring expenditure on reagents, contributing to substantial long-term savings in API manufacturing budgets.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of starting materials that are commercially abundant and easy to source globally. Chlorohydrazones and thio ylides can be synthesized from basic feedstocks like acyl chlorides and hydrazines, which are produced at massive scales for various industries. This reduces the dependency on single-source suppliers for niche reagents, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction to air and moisture further decreases the likelihood of batch failures caused by environmental factors, ensuring consistent delivery schedules to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is straightforward due to the absence of exothermic hazards or toxic gas evolution. The reaction can be safely conducted in standard stainless steel reactors without the need for specialized lining or pressure ratings. From an environmental standpoint, the process aligns with green chemistry principles by minimizing hazardous waste generation and avoiding persistent heavy metal contaminants. This facilitates easier regulatory approval and reduces the costs associated with waste treatment and disposal, making it an attractive option for companies aiming to improve their sustainability metrics and carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing portfolios.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive heavy metal catalysts and harsh reaction conditions. It operates at room temperature in air, uses inexpensive potassium carbonate as a promoter, and utilizes readily available starting materials, significantly simplifying post-treatment and reducing production costs.
Q: What is the structural scope of the substituents allowed in this reaction?
A: The reaction demonstrates broad substrate tolerance. R1 can be alkyl, phenyl, naphthyl, or furyl groups with various substituents like methyl, methoxy, or halogens. R2 and R3 can similarly be substituted or unsubstituted phenyl or naphthyl groups, allowing for the design of diverse libraries of bioactive molecules.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. The use of stable, non-toxic reagents like potassium carbonate, the absence of inert gas requirements (air atmosphere), and mild thermal conditions (20-40°C) make it ideal for scaling from gram-level laboratory synthesis to multi-ton industrial production without significant safety hazards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the lab bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering trifluoromethyl-substituted 1,2,4-triazines that meet the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced technology for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for maximum efficiency and reliability.
