Advanced One-Pot Synthesis of Beta-Substituted Azlactone Derivatives for Ramipril Production
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antihypertensive agents, and patent CN114478424A represents a significant technological leap in the production of key intermediates for Ramipril. This patent discloses a novel preparation method for azlactone derivatives substituted by a cycloalkenyl group at the beta position, specifically targeting the synthesis of (S,S,S)-2-azabicyclo[3,3,0]octane-3-carboxylic acid precursors. Unlike traditional multi-step processes that rely on hazardous reagents, this innovation utilizes a streamlined one-pot reaction system involving cyclic ketones, N,N-disubstituted formamide derivatives, and bis(trichloromethyl)carbonate. The technical breakthrough lies in the ability to generate the reactive electrophilic species in situ, thereby bypassing the isolation of unstable intermediates that have historically plagued this chemical space. For R&D directors and process chemists, this methodology offers a compelling alternative that aligns with modern green chemistry principles while maintaining high atom economy and product purity essential for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-substituted azlactone derivatives has been hindered by reliance on the Erlenmeyer-azlactone synthesis conditions or routes involving phosphorus oxychloride, as detailed in prior art such as WO 2011/133651. In these conventional pathways, the formation of the critical intermediate, 2-chloro-1-cyclopentenal (Structure V), requires the reaction of cyclopentanone with phosphorus oxychloride, a process that generates substantial quantities of phosphorus-containing wastewater which is environmentally burdensome and costly to treat. Furthermore, the intermediate 2-chloro-1-cyclopentenal is inherently volatile and thermally unstable, posing significant safety risks during heating and concentration steps, often leading to decomposition into tar-like substances that drastically reduce overall reaction yields. The necessity to isolate this unstable aldehyde before condensation with acylglycine adds complexity to the workflow, requiring strict temperature controls and specialized equipment to mitigate degradation, ultimately impacting the economic viability and scalability of the manufacturing process.
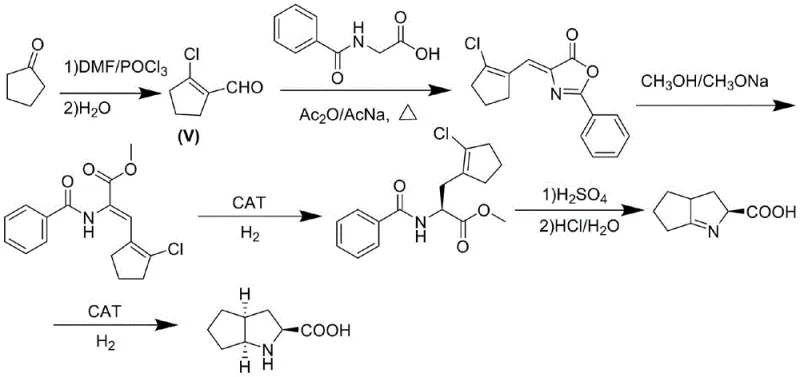
The Novel Approach
In stark contrast, the methodology described in CN114478424A introduces a sophisticated one-pot strategy that circumvents the isolation of unstable intermediates entirely by utilizing a Vilsmeier-Haack type activation system directly on the cyclic ketone. By reacting the cyclic ketone with bis(trichloromethyl)carbonate and an N,N-disubstituted formamide derivative, the process generates a reactive ammonium salt transition product in situ, which immediately undergoes condensation with the amino acid derivative or azlactone without the need for intermediate purification. This approach not only eliminates the use of phosphorus oxychloride and the associated environmental liabilities but also significantly enhances operational safety by avoiding the handling of volatile chloro-enals. Experimental data from the patent demonstrates the efficacy of this route, with Example 5 achieving a remarkable yield of 90.0% and HPLC purity of 99.5% for the target azlactone derivative, showcasing a substantial improvement over the inconsistent yields and purity profiles associated with legacy methods.
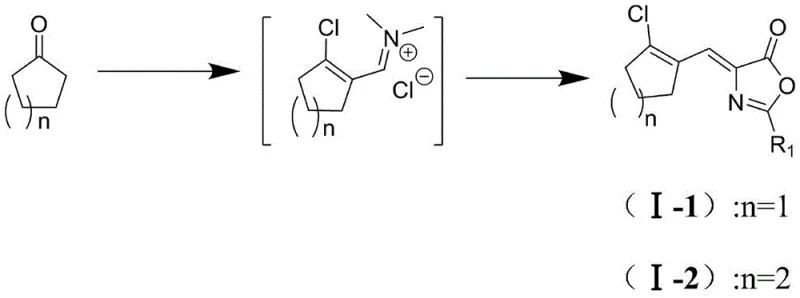
Mechanistic Insights into In Situ Vilsmeier-Haack Activation
The core mechanistic advantage of this novel synthesis lies in the generation of a highly reactive chloromethylene ammonium salt species, which acts as a potent dehydrating and activating agent within the reaction matrix. When bis(trichloromethyl)carbonate reacts with the N,N-disubstituted formamide derivative, it forms an electrophilic complex that activates the alpha-position of the cyclic ketone, effectively mimicking the reactivity of the unstable aldehyde without its physical isolation. This activated complex then facilitates a condensation reaction with the nucleophilic methylene group of the azlactone or amino acid derivative, driving the formation of the carbon-carbon double bond under mild thermal conditions ranging from 0°C to 90°C depending on the specific substrate. The reaction kinetics are carefully managed through temperature control during the dropwise addition of reagents, ensuring that the exothermic formation of the ammonium salt does not lead to side reactions or decomposition of the sensitive cyclic framework.
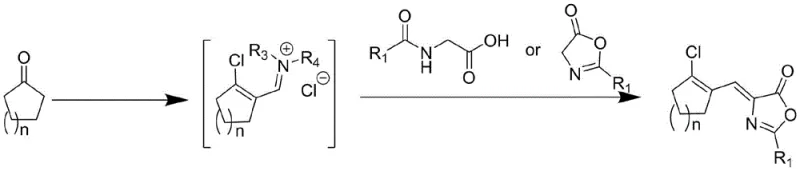
From an impurity control perspective, the one-pot nature of this reaction minimizes the exposure of reactive intermediates to external contaminants and reduces the number of unit operations where product loss typically occurs. The use of bis(trichloromethyl)carbonate as a chlorine source ensures a clean conversion profile, as the byproducts are primarily gaseous or soluble species that can be easily removed during the workup phase, unlike the phosphorus sludge generated in traditional routes. Furthermore, the choice of solvent systems, such as dichloromethane or cyclohexane, plays a critical role in stabilizing the transition states and facilitating the precipitation of the final product upon slurry purification. This mechanistic elegance allows for the synthesis of various derivatives by simply varying the cyclic ketone (n=1 to 4) and the R1 group on the azlactone, providing a versatile platform for generating diverse libraries of pharmaceutical intermediates with consistent quality attributes.
How to Synthesize Beta-Substituted Azlactone Derivatives Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, activation, and condensation steps that can be readily adapted for pilot and commercial scale operations. The process begins with the dissolution of the cyclic ketone and the formamide derivative in a suitable organic solvent, followed by the controlled addition of the carbonate activator to generate the reactive species. Once the activation is complete, the azlactone or amino acid precursor is introduced, and the mixture is heated to drive the condensation to completion, monitored via TLC or HPLC. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility and optimal yield for technical teams looking to adopt this methodology.
- Dissolve the cyclic ketone and N,N-disubstituted formamide derivative in a solvent like dichloromethane, then add bis(trichloromethyl)carbonate solution dropwise at low temperature to form the ammonium salt transition product.
- Introduce the amino acid derivative or pre-formed azlactone into the reaction mixture and maintain controlled heating until TLC monitoring confirms the disappearance of starting materials.
- Concentrate the reaction mixture to remove solvents and purify the resulting crude solid by slurry washing with appropriate solvents such as ethanol or ethyl acetate to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers transformative benefits regarding cost structure and operational reliability in the manufacturing of pharmaceutical intermediates. By eliminating the need for phosphorus oxychloride, the process removes a significant cost center associated with hazardous waste disposal and environmental compliance, leading to substantial cost savings in pharmaceutical intermediate manufacturing. The simplified one-pot workflow reduces the requirement for multiple reactors and extensive purification equipment, thereby lowering capital expenditure and energy consumption per kilogram of product produced. Additionally, the avoidance of unstable intermediates enhances supply chain reliability by reducing the risk of batch failures due to material degradation, ensuring a more consistent and predictable supply of high-quality intermediates for downstream API production.
- Cost Reduction in Manufacturing: The elimination of phosphorus-containing reagents drastically simplifies the waste treatment protocol, removing the need for expensive neutralization and disposal processes associated with phosphorus wastewater. Furthermore, the high yields achieved in this one-pot process, often exceeding 90%, maximize raw material utilization and reduce the cost of goods sold by minimizing the amount of starting material required per unit of output. The reduction in unit operations also translates to lower labor costs and reduced utility consumption, contributing to a leaner and more cost-effective manufacturing model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Utilizing stable cyclic ketones as starting materials instead of volatile and unstable chloro-enals significantly mitigates the risk of supply disruptions caused by material degradation during storage or transport. The robustness of the reaction conditions allows for flexible scheduling and longer campaign runs without the frequent equipment cleaning and maintenance required by corrosive phosphorus-based chemistries. This stability ensures a continuous and reliable flow of intermediates, enabling pharmaceutical companies to maintain steady production schedules for critical medications like Ramipril without the fear of unexpected batch rejections or delays.
- Scalability and Environmental Compliance: The green chemistry profile of this method, characterized by the absence of heavy metal catalysts and phosphorus waste, aligns perfectly with increasingly stringent global environmental regulations, facilitating easier permitting and long-term operational sustainability. The simplicity of the workup procedure, involving basic concentration and slurry purification, makes the process highly scalable from laboratory benchtop to multi-ton commercial production without significant re-engineering. This scalability ensures that suppliers can rapidly ramp up production to meet market demand surges while maintaining a minimal environmental footprint, a key factor for partners committed to sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational benefits and chemical advantages for stakeholders evaluating this process for integration into their supply chains. Understanding these details is crucial for making informed decisions about adopting this greener and more efficient manufacturing route.
Q: How does this new method improve environmental compliance compared to traditional routes?
A: The novel process completely eliminates the use of phosphorus oxychloride (POCl3), thereby preventing the generation of difficult-to-treat phosphorus-containing wastewater and significantly reducing the environmental footprint of the manufacturing process.
Q: What are the stability advantages of avoiding the 2-chloro-1-cyclopentenal intermediate?
A: By generating the reactive species in situ rather than isolating the volatile and unstable 2-chloro-1-cyclopentenal, the process avoids safety hazards associated with heating and concentration, leading to higher operational safety and consistent product quality.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the one-pot strategy simplifies unit operations by combining activation and condensation steps, which reduces energy consumption and processing time, making it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azlactone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value pharmaceutical intermediates like the beta-substituted azlactone derivatives described in CN114478424A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of azlactone intermediate meets the exacting standards required for the synthesis of life-saving antihypertensive drugs like Ramipril.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production needs, along with specific COA data and route feasibility assessments. Let us help you optimize your manufacturing process, reduce environmental impact, and secure a reliable supply of high-purity intermediates for your critical drug development programs.
