Advanced C-F Bond Activation Technology for Scalable Diaryl Ether Production
Advanced C-F Bond Activation Technology for Scalable Diaryl Ether Production
The chemical industry is currently witnessing a paradigm shift towards sustainable synthesis methodologies that leverage abundant feedstocks like carbon dioxide. A groundbreaking development in this arena is detailed in patent CN112125840B, which discloses a novel method for preparing diaryl ether compounds based on C-F bond activation with CO2 participation. This technology represents a significant leap forward for the production of high-value heterocyclic intermediates, specifically addressing the historical challenges associated with activating thermodynamically stable carbon-fluorine bonds. By utilizing CO2 not merely as a greenhouse gas but as a valuable oxygen resource, this process offers a green, efficient, and economically viable route to complex diaryl ethers. For R&D directors and procurement strategists, understanding the implications of this patent is crucial for optimizing supply chains and reducing the environmental footprint of pharmaceutical and agrochemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of diaryl ethers has relied heavily on nucleophilic aromatic substitution or transition-metal catalyzed cross-coupling reactions, such as the Ullmann condensation or Buchwald-Hartwig etherification. These conventional pathways typically require the coupling of aryl halides with phenolate salts or boronic acids. However, these methods often suffer from significant limitations when applied to fluorinated substrates. The carbon-fluorine bond is one of the strongest single bonds in organic chemistry, possessing high thermodynamic stability and kinetic inertness, which makes it resistant to standard oxidative addition processes used in palladium catalysis. Consequently, synthesizing fluorinated diaryl ethers often necessitates harsh reaction conditions, including elevated temperatures, strong bases, and expensive ligands, which can lead to poor selectivity and the formation of difficult-to-remove impurities. Furthermore, the reliance on pre-functionalized phenol derivatives increases the step count and overall cost of goods, creating bottlenecks in the supply chain for key pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN112125840B introduces a transformative strategy that directly activates the C-F bond using carbon dioxide as the oxygen source. This approach bypasses the need for pre-formed phenols, instead constructing the C-O bond through a unique multi-component coupling involving a 2-fluoropyridine derivative, CO2, and phenylsilane. The reaction proceeds under remarkably mild conditions, specifically at room temperature (25°C) and atmospheric pressure (1atm), which drastically reduces energy consumption and safety risks associated with high-pressure operations. This method not only expands the scope of accessible chemical space by enabling the functionalization of stable fluoro-heterocycles but also aligns perfectly with green chemistry principles by valorizing CO2. The ability to generate complex diaryl ether scaffolds from simple, readily available starting materials positions this technology as a superior alternative for modern process chemistry.
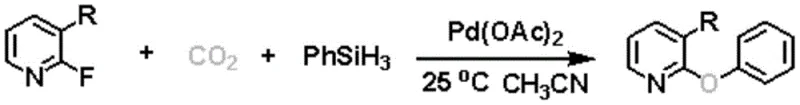
Mechanistic Insights into Pd-Catalyzed C-F Activation with CO2
The core of this innovation lies in the sophisticated palladium-catalyzed cycle that facilitates the cleavage of the robust C-F bond and the subsequent insertion of oxygen derived from carbon dioxide. While the exact mechanistic details involve complex organometallic steps, the process fundamentally relies on the ability of the palladium catalyst, specifically palladium acetate (Pd(OAc)2), to mediate the activation of the fluoro-pyridine substrate. The presence of phenylsilane (PhSiH3) plays a dual critical role: it acts as a reducing agent to regenerate the active catalytic species and serves as the source of the phenyl group that ultimately forms the ether linkage. The cesium carbonate base assists in deprotonation steps and stabilizes intermediates, ensuring the smooth progression of the catalytic cycle. This synergistic interaction between the transition metal catalyst, the silane reductant, and the CO2 molecule allows for the construction of the C-O-C bridge under neutral to mild basic conditions, avoiding the degradation of sensitive functional groups often seen in harsher protocols.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity intermediates. The high selectivity of the C-F activation ensures that side reactions, such as homocoupling or defluorination without oxygen insertion, are minimized. The mild reaction temperature of 25°C further suppresses thermal decomposition pathways that often plague high-temperature couplings. For R&D teams, this means a cleaner crude reaction profile, which simplifies downstream purification processes like silica gel column chromatography or crystallization. The patent data indicates yields of up to 83% for cyano-substituted substrates and 75% for nitro-substituted analogs, demonstrating robust performance across different electronic environments. This level of control is essential for meeting the stringent purity specifications required in the manufacture of active pharmaceutical ingredients (APIs) and advanced agrochemicals.
How to Synthesize Diaryl Ethers Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and reaction atmosphere to maximize yield and reproducibility. The protocol involves dissolving the 2-fluoropyridine compound in acetonitrile, followed by the sequential addition of the palladium catalyst, cesium carbonate, and phenylsilane. A critical operational step is the thorough displacement of the reaction vessel with CO2 gas to ensure an adequate concentration of the oxygen source throughout the 24-hour reaction period. The detailed standardized synthesis steps, including specific workup procedures like ice-water quenching and dichloromethane extraction, are outlined below to guide process engineers in replicating this high-efficiency transformation.
- Prepare the reaction vessel by adding an acetonitrile solution of the 2-fluoropyridine substrate.
- Sequentially add the catalyst palladium acetate, cesium carbonate base, and phenylsilane reductant to the mixture.
- Displace the system with CO2 gas three times and maintain reaction at 25°C and 1atm pressure for 24 hours before workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CO2-participated C-F activation technology presents compelling economic and logistical benefits. The shift from traditional high-energy coupling methods to this ambient condition process directly translates to significant operational cost savings. By eliminating the need for specialized high-pressure reactors and extensive heating or cooling infrastructure, manufacturers can reduce capital expenditure (CAPEX) and lower utility costs. Furthermore, the use of CO2 as a raw material leverages an abundant and inexpensive feedstock, potentially insulating production costs from the volatility associated with specialized phenolic reagents. This process intensification allows for more flexible manufacturing schedules and reduced lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the simplification of reaction conditions and the use of commodity chemicals. Since the reaction runs at 25°C and 1atm, the energy load is minimal compared to traditional refluxing or高压 (high-pressure) reactions. Additionally, the avoidance of expensive ligand systems often required for difficult C-F activations reduces the raw material cost per kilogram. The high atom economy of incorporating CO2 and the silane directly into the product structure minimizes waste generation, which in turn lowers waste disposal costs. These factors combine to create a leaner, more cost-effective manufacturing process that enhances overall margin potential for the final API or intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the availability of the key starting materials. 2-Fluoropyridines are widely produced commodity chemicals, and phenylsilane is a standard reagent in the fine chemical industry. Unlike specialized boronic acids or stannanes which can suffer from supply shortages or long lead times, the reagents for this process are robustly sourced. The mild conditions also mean that the process is less sensitive to minor fluctuations in utility supply (e.g., steam or chilled water), ensuring consistent batch-to-batch quality. This reliability is critical for maintaining continuous production lines and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is inherently safer and more straightforward due to the absence of hazardous high-pressure gases or extreme temperatures. The use of CO2, a non-toxic gas, aligns with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste. The process generates fewer byproducts, simplifying effluent treatment and reducing the environmental burden. This 'green' credential is increasingly valued by downstream customers who are under pressure to reduce their Scope 3 emissions, making suppliers who adopt such technologies preferred partners in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-F activation technology. These answers are derived directly from the experimental data and technical disclosures within patent CN112125840B, providing clarity on reaction scope, safety, and scalability for potential adopters.
Q: What is the primary advantage of using CO2 in this diaryl ether synthesis?
A: The primary advantage is the utilization of CO2 as a sustainable and inexpensive oxygen source for C-F bond activation, replacing traditional harsh oxidants or pre-functionalized phenols, thereby enhancing atom economy and reducing environmental impact.
Q: Can this method tolerate sensitive functional groups like nitriles and nitro groups?
A: Yes, the patent data explicitly demonstrates high compatibility with electron-withdrawing groups such as cyano (CN) and nitro (NO2) substituents on the pyridine ring, yielding target products with good selectivity under mild conditions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely. The reaction operates at ambient temperature (25°C) and atmospheric pressure (1atm), eliminating the need for expensive high-pressure reactors or cryogenic cooling, which significantly simplifies scale-up and reduces capital expenditure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the CO2-participated C-F activation technology described in patent CN112125840B for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise to translate such innovative academic and patent literature into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle complex catalytic cycles involving sensitive organometallic species, ensuring that the high yields and selectivity reported in the lab are maintained during scale-up. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality that meets global regulatory standards.
We invite R&D directors and procurement leaders to collaborate with us to evaluate the feasibility of integrating this advanced synthesis route into your supply chain. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data for similar diaryl ether structures and to discuss route feasibility assessments that could accelerate your project timelines and reduce overall manufacturing costs.
