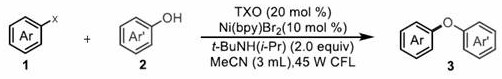
Advanced Nickel-Photoredox Catalysis for Scalable Diaryl Ether Production
The chemical manufacturing landscape is continuously evolving towards more sustainable and cost-efficient methodologies, particularly in the synthesis of complex heteroatom-linked scaffolds essential for modern drug discovery. A significant breakthrough in this domain is documented in Chinese Patent CN113717038A, which discloses a novel method for synthesizing diaryl ether compounds through a nickel/ketone double-catalysis reaction. This technology represents a paradigm shift from traditional transition metal cross-coupling strategies by leveraging the synergistic power of photoredox catalysis and earth-abundant nickel chemistry. By utilizing 9-Thioxanthone (TXO) as a photosensitizer in conjunction with a nickel bromide complex, the process enables the efficient coupling of halogenated aromatic hydrocarbons with aryl phenols under remarkably mild conditions. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent offers a compelling alternative to legacy methods, promising enhanced operational safety and reduced raw material costs without compromising on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of diaryl ether motifs, which are ubiquitous structural units in bioactive natural products and medicinal compounds, has relied heavily on copper or palladium-catalyzed cross-coupling reactions. While effective, these conventional pathways often impose severe constraints on process scalability and economic viability. Traditional Ullmann-type couplings typically necessitate the use of stoichiometric amounts of copper salts, strongly basic conditions, and elevated temperatures that can exceed 100°C, leading to significant energy consumption and potential decomposition of thermally sensitive substrates. Similarly, palladium-catalyzed Buchwald-Hartwig etherifications, though milder, suffer from the prohibitive cost of precious metal catalysts and the requirement for sophisticated, air-sensitive ligand systems that complicate supply chain logistics. Furthermore, the removal of residual heavy metals from the final active pharmaceutical ingredient (API) to meet stringent regulatory limits adds additional downstream processing steps, increasing both production time and waste generation.
The Novel Approach
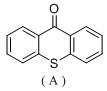
In stark contrast to these energy-intensive protocols, the methodology described in patent CN113717038A introduces a transformative dual-catalytic system that operates efficiently at room temperature under visible light irradiation. This innovative approach replaces expensive palladium catalysts with a cost-effective nickel complex, specifically Ni(bpy)Br2, and employs an organic ketone, 9-Thioxanthone (TXO), to harness light energy for catalytic turnover. The reaction proceeds in common solvents like acetonitrile under inert gas protection, utilizing a mild amine base rather than harsh alkoxides or hydrides. This shift not only drastically simplifies the operational requirements but also expands the substrate scope to include sensitive functional groups that would otherwise be incompatible with high-temperature conditions. The ability to drive the reaction using a standard 45W compact fluorescent lamp (CFL) underscores the practicality of this method for large-scale commercial adoption, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nickel/Photoredox Dual Catalysis
The success of this synthetic strategy lies in the intricate interplay between the photo-excited organic catalyst and the nickel redox cycle. Upon irradiation with visible light, the 9-Thioxanthone (TXO) photosensitizer absorbs photons to reach an excited triplet state, possessing sufficient energy to engage in single-electron transfer (SET) processes. This excited species interacts with the nickel catalyst, facilitating the generation of low-valent nickel species capable of oxidative addition into the carbon-halogen bond of the aryl halide substrate. Simultaneously, the phenol substrate is deprotonated by the amine base to form a nucleophilic phenoxide, which subsequently coordinates to the nickel center. The catalytic cycle culminates in a reductive elimination step that forms the desired carbon-oxygen bond, regenerating the active nickel species for further turnover. This mechanistic pathway bypasses the high activation energy barriers associated with thermal two-electron processes, allowing the reaction to proceed smoothly at ambient temperatures.
From an impurity control perspective, this mechanism offers distinct advantages over traditional thermal methods. The mild reaction environment minimizes side reactions such as homocoupling of the aryl halide or degradation of the phenolic component, which are common pitfalls in high-temperature copper catalysis. Additionally, the use of a well-defined nickel-bipyridine complex ensures consistent catalytic activity, reducing the formation of ill-defined metal aggregates that can act as sinks for reactants or sources of contamination. The specificity of the photoredox activation also means that the reaction can be precisely controlled by simply switching the light source on or off, providing an extra layer of process safety and control that is unavailable in purely thermal systems. This level of control is critical for maintaining high purity profiles in complex pharmaceutical intermediates, ensuring that the final product meets the rigorous specifications required for downstream drug synthesis.
How to Synthesize Diaryl Ether Efficiently
The implementation of this nickel-mediated etherification protocol is straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation by oxygen. Key to the success of the reaction is the precise molar ratio of the halogenated aromatic hydrocarbon to the aryl phenol, typically maintained at 1:3 to drive the equilibrium towards product formation. The detailed standardized synthesis steps, including specific workup procedures and purification techniques to achieve high isolated yields, are outlined in the guide below.
- Charge a dry reaction vessel with halogenated aromatic hydrocarbon, aryl phenol, TXO photocatalyst, Ni(bpy)Br2 catalyst, and t-BuNH(i-Pr) base under inert gas protection.
- Add acetonitrile (MeCN) as the solvent and ensure the mixture is thoroughly stirred to create a homogeneous suspension.
- Irradiate the reaction mixture with a 45W CFL lamp at room temperature for 72 to 120 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel/photoredox technology presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The most immediate advantage is the substantial cost savings derived from replacing precious metal catalysts with abundant nickel salts, which are orders of magnitude cheaper and less subject to geopolitical supply volatility. Furthermore, the elimination of high-temperature heating requirements translates into significant energy savings and reduces the load on facility cooling systems, contributing to a lower overall carbon footprint for the manufacturing process. The mild conditions also extend the lifespan of reactor vessels and reduce maintenance costs associated with corrosion from harsh bases, enhancing the long-term economic viability of the production line.
- Cost Reduction in Manufacturing: The substitution of palladium with nickel eliminates the need for expensive ligand systems and reduces the cost of goods sold (COGS) significantly. Since the catalyst loading is low and the metal is inexpensive, the financial burden of catalyst recovery or disposal is minimized. Additionally, the use of simple amine bases instead of specialized strong bases reduces raw material procurement costs and simplifies inventory management. The overall process efficiency, characterized by high yields and minimal byproduct formation, ensures that raw materials are utilized effectively, further driving down the cost per kilogram of the final diaryl ether product.
- Enhanced Supply Chain Reliability: Relying on earth-abundant nickel and commercially available organic photocatalysts like TXO mitigates the risk of supply chain disruptions often associated with precious metals. The reagents required for this process are widely sourced from multiple global suppliers, ensuring continuity of supply even during market fluctuations. Moreover, the robustness of the reaction conditions allows for flexible scheduling and batch processing without the need for specialized high-pressure or high-temperature equipment, making it easier to scale up production to meet sudden increases in demand from downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with modern environmental, social, and governance (ESG) goals. Operating at room temperature reduces energy consumption, while the use of acetonitrile, a solvent with established recycling protocols, facilitates waste management. The absence of toxic heavy metals like palladium in the final product streamlines the regulatory approval process for new drug applications, as residual metal limits are easier to meet. This environmental compliance not only avoids potential fines but also enhances the brand reputation of the manufacturer as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dual-catalysis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams evaluating this route for commercial adoption.
Q: What are the primary advantages of this nickel-catalyzed method over traditional palladium coupling?
A: This method utilizes earth-abundant nickel instead of expensive palladium, operates at room temperature rather than requiring high heat, and avoids the strongly basic conditions often needed for copper-catalyzed Ullmann reactions, resulting in a greener and more cost-effective process.
Q: Is this protocol suitable for sensitive functional groups like esters or aldehydes?
A: Yes, the mild reaction conditions allow for excellent tolerance of various functional groups including esters, nitriles, trifluoromethyl groups, and aldehydes, which might otherwise decompose under harsh thermal conditions.
Q: What is the role of 9-Thioxanthone (TXO) in this reaction system?
A: TXO acts as an organic photosensitizer that absorbs visible light to generate excited states, which then facilitate the single-electron transfer processes necessary to activate the nickel catalytic cycle for carbon-oxygen bond formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the nickel/photoredox system described in CN113717038A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with the necessary photochemical reactors and stringent purity specifications to handle sensitive catalytic cycles, supported by rigorous QC labs that guarantee every batch meets the highest international standards for quality and consistency.
We invite forward-thinking pharmaceutical companies and chemical manufacturers to collaborate with us to leverage this cost-effective and sustainable synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how this technology can optimize your supply chain. We encourage you to contact us today to request specific COA data for our diaryl ether portfolio and discuss route feasibility assessments that can accelerate your drug development timelines while reducing overall manufacturing costs.
