Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharmaceutical Manufacturing
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral scaffolds that serve as critical building blocks for novel therapeutics. Patent CN112209947A introduces a groundbreaking synthetic route for chiral indoxazinone compounds, a class of molecules characterized by their fused indole-oxazinone core structure which exhibits significant potential in oncology. This technology leverages a sophisticated dual-catalytic system involving chiral phosphoric acids and hexafluoroisopropanol to achieve exceptional stereocontrol under remarkably mild conditions. For R&D directors and procurement specialists, this represents a pivotal advancement, offering a pathway to high-purity intermediates without the need for harsh reagents or expensive transition metals. The structural versatility of the resulting compounds, as defined by the general formula where R groups can vary widely across alkyl, aryl, and heteroaryl substituents, allows for extensive library generation for structure-activity relationship studies.
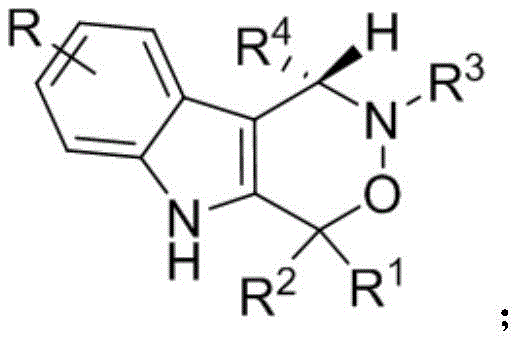
The core innovation lies in the ability to access these biologically active motifs through a direct cycloaddition strategy that bypasses the limitations of traditional multi-step syntheses. By utilizing readily available 2-indolemethanols and nitrones as starting materials, the process ensures a high degree of atom economy and operational simplicity. The resulting products have demonstrated potent cytotoxicity against PC-3 human prostate cancer cell lines, validating their utility as lead compounds for anti-tumor drug discovery. This patent not only secures a novel chemical space but also provides a scalable manufacturing protocol that aligns with modern green chemistry principles, making it an attractive asset for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral indole-fused ring systems has been plagued by significant synthetic challenges that hinder efficient commercial production. Traditional approaches often rely on stoichiometric amounts of chiral auxiliaries or expensive transition metal catalysts, which introduce heavy metal contamination risks that are strictly regulated in pharmaceutical manufacturing. Furthermore, many existing protocols require cryogenic temperatures or highly reactive reagents that pose safety hazards and increase energy consumption drastically. The lack of generality in older methods often means that changing a single substituent on the aromatic ring can lead to a catastrophic drop in yield or enantioselectivity, forcing process chemists to re-optimize conditions for every new analog. These inefficiencies result in prolonged development timelines and inflated costs, creating bottlenecks in the supply chain for critical drug candidates.
The Novel Approach
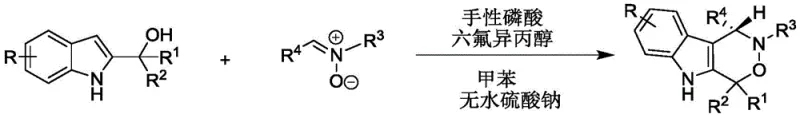
In stark contrast, the methodology disclosed in CN112209947A utilizes an organocatalytic cascade that operates at near-ambient temperatures, specifically between 20°C and 30°C, eliminating the need for energy-intensive cooling systems. The reaction employs a synergistic combination of a chiral phosphoric acid and hexafluoroisopropanol (HFIP) in toluene, creating a highly organized hydrogen-bonding network that directs the stereochemical outcome with precision. This approach tolerates a broad spectrum of functional groups, including halogens, alkoxy groups, and various heteroaryl moieties, without compromising the integrity of the final product. The use of anhydrous sodium sulfate as a simple additive further streamlines the process by managing water content effectively, ensuring consistent reaction performance. This robustness translates directly to reduced operational complexity and enhanced reliability for large-scale manufacturing environments.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cycloaddition
The success of this synthesis hinges on the intricate interplay between the chiral phosphoric acid catalyst and the hexafluoroisopropanol co-catalyst, which together activate the electrophilic and nucleophilic partners simultaneously. The chiral phosphoric acid, typically derived from a binaphthyl or spiro skeleton, acts as a Brønsted acid to activate the nitrone dipole through hydrogen bonding, while the HFIP enhances the acidity and stabilizes the transition state through a cooperative hydrogen-bonding network. This dual activation lowers the energy barrier for the [3+2] or similar cycloaddition pathway, facilitating the formation of the new C-C and C-O bonds with high fidelity. The steric bulk of the catalyst's 3,3'-substituents, such as the 9-phenanthryl group, creates a confined chiral pocket that strictly controls the facial selectivity of the attack, thereby dictating the absolute configuration of the newly formed stereocenters.
Impurity control is inherently built into this mechanism due to the high specificity of the organocatalytic cycle. Unlike radical-based or metal-catalyzed reactions that often generate diverse side products through non-selective pathways, this proton-transfer mediated process follows a well-defined trajectory. The mild reaction conditions prevent the decomposition of sensitive functional groups on the indole or nitrone substrates, which is a common source of impurities in harsher protocols. Additionally, the use of toluene as a solvent ensures good solubility of the organic components while allowing for easy removal during workup, minimizing the risk of solvent-derived adducts. The result is a crude reaction mixture with a clean profile, significantly reducing the burden on downstream purification steps and improving the overall mass balance of the process.
How to Synthesize Chiral Indoxazinone Efficiently
The practical execution of this synthesis is designed for ease of handling, requiring standard laboratory equipment and commercially available reagents. The process begins with the precise weighing of 2-indolemethanol and nitrone substrates, which are then dissolved in dry toluene. Anhydrous sodium sulfate is added to the mixture to scavenge any trace moisture that could deactivate the acid catalyst, followed by the introduction of the chiral phosphoric acid and hexafluoroisopropanol. The reaction is allowed to proceed with stirring at a controlled temperature of 30°C, monitored by thin-layer chromatography (TLC) until the starting materials are fully consumed. Upon completion, the solid additive is removed by filtration, and the filtrate is concentrated under reduced pressure. The final product is isolated via silica gel column chromatography using a petroleum ether and dichloromethane gradient, yielding the target chiral indoxazinone as a high-purity white solid.
- Combine 2-indolemethanol and nitrone reactants in toluene solvent with anhydrous sodium sulfate.
- Add chiral phosphoric acid and hexafluoroisopropanol catalysts and stir the mixture at 20-30°C.
- Monitor reaction via TLC, then filter, concentrate, and purify the product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of precious metal catalysts removes a major cost driver and simplifies the regulatory compliance landscape, as there is no need for rigorous heavy metal testing or specialized scavenging resins. The reliance on commodity chemicals like toluene and simple indole derivatives ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or single-source reagents. Furthermore, the ambient temperature operation significantly reduces the facility's energy footprint, contributing to lower utility costs and aligning with corporate sustainability goals. These factors combine to create a manufacturing process that is not only economically viable but also robust enough to withstand the demands of global pharmaceutical supply networks.
- Cost Reduction in Manufacturing: The process achieves substantial cost optimization by replacing expensive transition metal catalysts with organocatalysts that are used in low loadings. The mild reaction conditions eliminate the need for specialized cryogenic equipment, leading to significant savings in capital expenditure and energy consumption. Additionally, the high yields and selectivity reduce the amount of raw material wasted on side products, improving the overall cost-efficiency of the production line.
- Enhanced Supply Chain Reliability: By utilizing broadly available starting materials such as substituted indoles and nitrones, the method reduces dependency on complex custom synthesis providers. The robustness of the reaction conditions means that production can be maintained consistently even with minor variations in raw material quality, ensuring uninterrupted supply for downstream drug formulation. This stability is crucial for maintaining long-term contracts and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The use of toluene, a standard industrial solvent, facilitates easy scale-up from gram to kilogram scales without the need for process re-engineering. The absence of toxic heavy metals simplifies waste treatment protocols, reducing the environmental impact and disposal costs associated with hazardous waste streams. This alignment with green chemistry principles enhances the company's environmental profile and ensures compliance with increasingly stringent global regulations regarding pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral indoxazinone synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on performance metrics and application scope. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production pipelines or R&D programs.
Q: What is the enantioselectivity achieved in this synthesis method?
A: The method achieves extremely high enantioselectivity, with ee values reaching up to 96% depending on the specific substrate substituents, ensuring high optical purity for drug development.
Q: Does the synthesized compound exhibit biological activity?
A: Yes, the chiral indoxazinone compounds demonstrate cytotoxic activity against PC-3 human prostate cancer cells, with IC50 values as low as 40.08 μg/mL for specific derivatives.
Q: What are the optimal reaction conditions for this process?
A: The reaction operates under mild conditions, typically at 20-30°C in toluene, utilizing a dual catalytic system of chiral phosphoric acid and hexafluoroisopropanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation anti-cancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral indoxazinone meets the highest standards of optical purity and chemical integrity required by global regulatory bodies. Our commitment to excellence makes us the ideal partner for companies seeking to secure a stable supply of these valuable pharmaceutical building blocks.
We invite you to contact our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this organocatalytic route. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and accelerate your drug development timeline.
