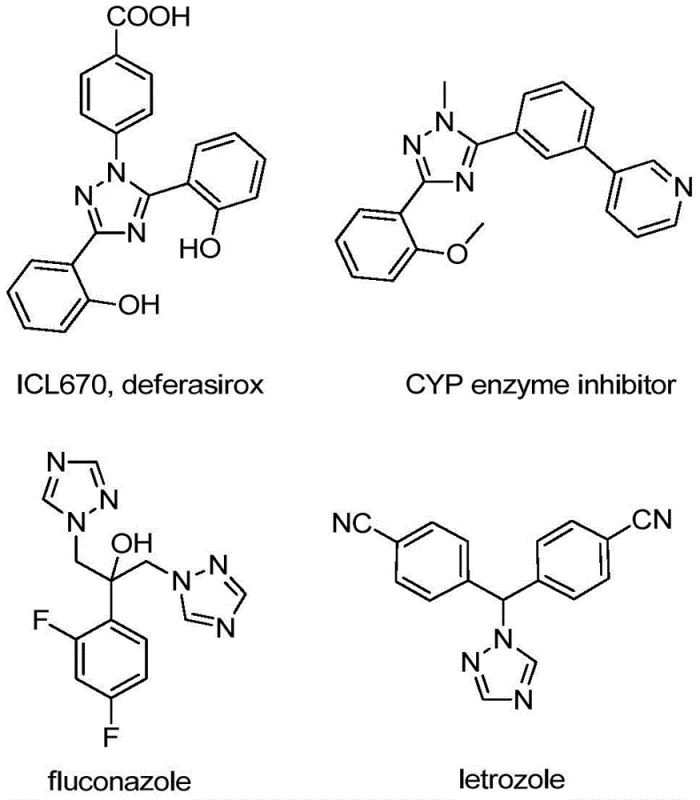
Advanced Iodine-Catalyzed Synthesis for Commercial Scale-Up of High-Purity Triazole Intermediates
Patent CN110467579B introduces a novel iodine-catalyzed synthesis route for 5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in the manufacturing of high-value pharmaceutical intermediates. This method eliminates the need for heavy metal catalysts and anhydrous conditions while maintaining excellent yields (up to 97%) across diverse substrate combinations. The process operates under mild conditions (80°C) with readily available starting materials, making it particularly suitable for commercial scale-up of complex pharmaceutical intermediates.
Advanced Reaction Mechanism and Purity Control
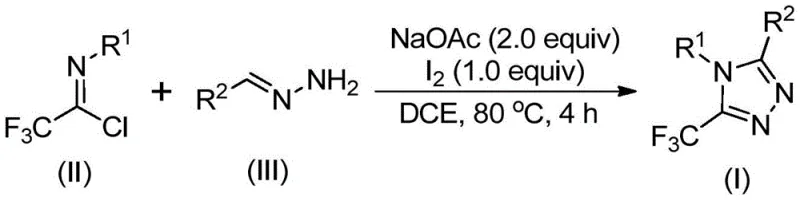
The synthetic pathway begins with the base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethyl imidoyl chloride (II) and hydrazone (III), yielding a trifluoroacetamidine intermediate. This is followed by isomerization and base-promoted oxidative iodination to form an iodine-containing intermediate, which subsequently undergoes intramolecular electrophilic substitution and aromatization to produce the final 5-trifluoromethyl substituted 1,2,4-triazole compound (I). The reaction mechanism avoids transition metal catalysts entirely, eliminating potential metal contamination that would require costly purification steps in pharmaceutical manufacturing. The mild reaction conditions (80°C in DCE solvent) prevent thermal degradation of sensitive functional groups, while the absence of anhydrous requirements simplifies process setup and reduces operational complexity. The use of elemental iodine as catalyst (0.2 mmol for 0.2 mmol substrate) provides excellent chemoselectivity without generating toxic byproducts that could complicate downstream processing.
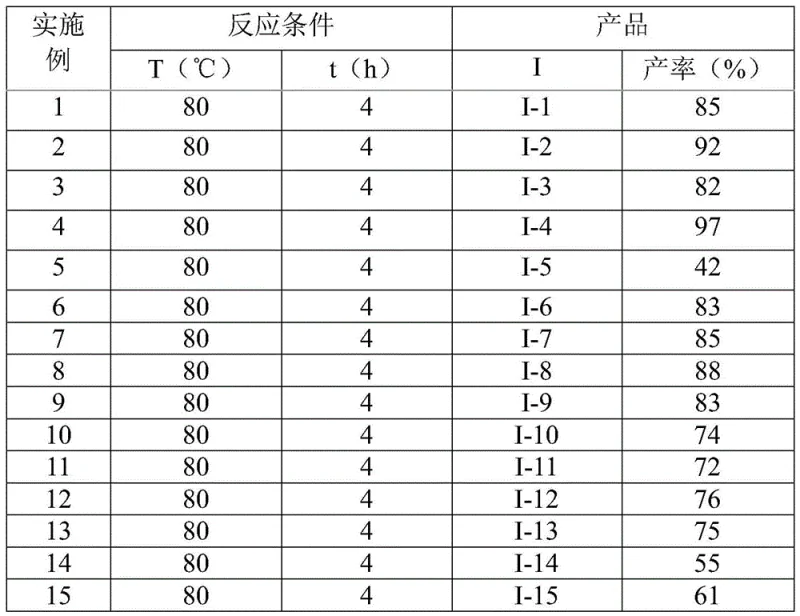
Impurity profile management is significantly enhanced through this innovative approach. The reaction's high functional group tolerance (as demonstrated in Table 2 with yields ranging from 42% to 97% across diverse substrates) minimizes side reactions that typically generate impurities in conventional triazole syntheses. The straightforward workup procedure—consisting of filtration, silica gel mixing, and column chromatography—effectively removes residual iodine and sodium acetate without requiring specialized equipment. The absence of heavy metal catalysts eliminates the need for rigorous metal scavenging steps that often introduce additional impurities in traditional routes. Furthermore, the well-defined reaction pathway produces consistent product quality with minimal batch-to-batch variation, ensuring stringent purity specifications required for pharmaceutical intermediates. The structural characterization data (including NMR and HRMS) provided in the patent confirms high product purity across all tested substrates, with no detectable metal residues or unexpected byproducts.
Supply Chain Optimization and Cost Reduction Benefits
This novel manufacturing process addresses critical pain points in traditional triazole synthesis that have long plagued pharmaceutical supply chains. Conventional methods often require expensive transition metal catalysts, strict anhydrous conditions, and complex purification protocols that increase production costs and extend lead times. The iodine-catalyzed approach presented in CN110467579B eliminates these bottlenecks while maintaining high yields across diverse substrate combinations. By removing the need for specialized equipment and hazardous materials handling, this method significantly reduces capital expenditure requirements and operational complexity for manufacturers seeking reliable pharmaceutical intermediate suppliers.
- Reduced Capital Expenditure: The elimination of transition metal catalysts removes the need for expensive metal scavenging systems and specialized containment equipment required in traditional processes. This represents substantial cost savings in facility setup and maintenance while avoiding the regulatory burden associated with heavy metal handling. The simplified process design allows for implementation in standard chemical manufacturing facilities without costly modifications, enabling faster time-to-market for new pharmaceutical intermediates. Furthermore, the absence of anhydrous requirements eliminates the need for specialized drying equipment and moisture-controlled environments that typically account for significant capital investment in fine chemical manufacturing.
- Shortened Production Lead Time: The streamlined reaction protocol (80°C for 4 hours followed by iodine addition) reduces cycle time compared to conventional multi-step syntheses requiring cryogenic conditions or extended reaction periods. The simplified workup procedure—comprising only filtration and column chromatography—minimizes processing time between reaction completion and product isolation. This efficiency gain translates directly to reduced lead time for high-purity pharmaceutical intermediates without compromising quality standards. Additionally, the robust nature of the process reduces batch failure rates that often cause supply chain disruptions in traditional manufacturing approaches.
- Enhanced Process Sustainability: The elimination of toxic heavy metal catalysts significantly reduces hazardous waste generation that requires specialized disposal protocols in conventional triazole synthesis. This environmental benefit aligns with growing regulatory pressures for greener manufacturing processes while reducing waste treatment costs for chemical manufacturers. The use of elemental iodine as catalyst is both cost-effective and environmentally preferable to transition metal alternatives that require complex recovery systems. Furthermore, the high atom economy of this one-pot synthesis minimizes raw material consumption and byproduct formation compared to traditional multi-step approaches.
Traditional vs. Novel Triazole Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted triazoles typically rely on either pre-synthesized heterocycles followed by trifluoromethylation or coupling reactions using specialized trifluoromethyl synthons like trifluorodiazoethane. These methods often require expensive transition metal catalysts (such as palladium or copper complexes) that necessitate rigorous removal processes to meet pharmaceutical purity standards. The need for strictly anhydrous and anaerobic conditions increases operational complexity and equipment costs while limiting scalability potential. Furthermore, many conventional routes suffer from narrow substrate scope and functional group intolerance, requiring extensive process reoptimization when adapting to new molecular targets. The multi-step nature of traditional syntheses also contributes to lower overall yields and higher production costs due to intermediate isolation and purification requirements.
The Innovative Iodine-Catalyzed Approach
The method disclosed in CN110467579B overcomes these limitations through a cleverly designed iodine-mediated cascade reaction that operates under mild conditions without transition metals. By utilizing readily available starting materials—trifluoroethyl imidoyl chloride (easily synthesized from aromatic amines) and hydrazones (simply prepared from aldehydes)—the process achieves remarkable substrate flexibility while maintaining high yields across diverse functional groups as demonstrated in Table 2. The reaction proceeds efficiently at 80°C in standard organic solvents like DCE without requiring moisture-free environments, significantly simplifying process implementation and scale-up potential. The single-pot methodology eliminates intermediate isolation steps that typically reduce overall yield in conventional approaches while providing excellent control over regioselectivity at the critical 5-position of the triazole ring. This innovative approach enables cost reduction in pharmaceutical manufacturing by reducing both raw material costs and processing complexity while maintaining the high purity standards required for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Substituted 1,2,4-Triazole Supplier
While iodine-catalyzed methodologies have demonstrated significant potential in fine chemical synthesis, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure seamless technology transfer from laboratory to manufacturing scale. Our rigorous QC labs maintain stringent purity specifications through advanced analytical capabilities that guarantee consistent product quality meeting global regulatory requirements for pharmaceutical intermediates. As a trusted partner in complex molecule manufacturing, we combine deep technical expertise with flexible production capabilities to support your specific development timelines and quality expectations.
We invite you to initiate a Customized Cost-Saving Analysis for your specific triazole intermediate requirements by contacting our technical procurement team today. Request specific COA data and comprehensive route feasibility assessments to evaluate how this innovative synthesis approach can enhance your supply chain resilience while delivering substantial cost reduction benefits for your pharmaceutical manufacturing operations.
