Optimizing Nicorandil Production: A Safe and Scalable Nitration Strategy for Global Supply Chains
Optimizing Nicorandil Production: A Safe and Scalable Nitration Strategy for Global Supply Chains
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for safer, more efficient, and cost-effective manufacturing processes for critical active pharmaceutical ingredients (APIs). A recent technological breakthrough disclosed in patent CN110845403A introduces a novel preparation method for Nicorandil, a potent vasodilator widely used in the treatment of coronary heart disease and angina pectoris. This innovation addresses long-standing challenges in the nitration of organic compounds, shifting away from hazardous mixed-acid systems toward a milder, propionic anhydride-mediated esterification process. By leveraging N-(2-hydroxyethyl)nicotinamide as a stable starting material, this route offers a robust alternative that enhances both operational safety and product purity. 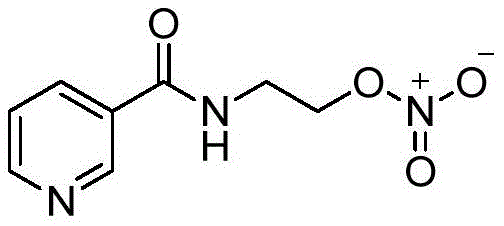
Nicorandil represents a unique pharmacological profile, combining the properties of a nitrate donor with those of an ATP-sensitive potassium channel opener. Its clinical efficacy in managing ischemic heart disease has established it as a priority medication for physicians worldwide. However, the commercial viability of such complex molecules often hinges on the efficiency of their synthetic routes. The method detailed in CN110845403A utilizes a specific combination of dilute nitric acid, propionic acid, and propionic anhydride to achieve high conversion rates under温和 conditions. This approach not only mitigates the risks associated with traditional nitration reagents but also streamlines the downstream purification process, ensuring a reliable supply of high-purity Nicorandil for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nicorandil has been plagued by significant technical and safety hurdles that impede efficient large-scale production. One prominent prior art method, referenced in Bioorganic & Medicinal Chemistry, relies on the direct nitration of N-(2-hydroxyethyl)nicotinamide using concentrated nitric acid or a concentrated nitric/sulfuric acid mixture. 
As illustrated in the reaction scheme above, this conventional approach suffers from poor reaction selectivity, often resulting in yields as low as 40%. The use of strong mixed acids generates intense exothermic reactions that are difficult to control, posing severe safety risks in an industrial setting. Furthermore, the generation of large volumes of spent acid waste creates a substantial environmental burden, complicating compliance with modern green chemistry standards and driving up disposal costs for manufacturers.
Alternative strategies have attempted to circumvent these issues but introduced new complexities. For instance, another literature method employs tert-butyl nitrite as a nitration reagent in the presence of a specific metal catalyst and oxygen. While this avoids mixed acids, it necessitates the use of expensive catalytic systems and requires operation under aerobic conditions, which introduces potential explosion hazards. Additionally, the raw materials for this route are often costly and difficult to source in bulk quantities, rendering it economically unfeasible for cost-sensitive generic drug production. Another patented route involves the condensation of unstable low-carbon-chain nitrate intermediates, which are prone to decomposition and explosion, further highlighting the urgent need for a safer, more stable synthetic pathway.
The Novel Approach
In stark contrast to these problematic legacy methods, the novel process disclosed in CN110845403A utilizes a sophisticated nitration system based on dilute nitric acid and propionic anhydride. This method capitalizes on the superior esterification capability of the acid anhydride-nitric acid complex, which allows the reaction to proceed efficiently at significantly lower temperatures, typically between 10°C and 30°C. By avoiding the use of concentrated sulfuric acid, the process eliminates the formation of sulfonated byproducts and drastically reduces the corrosivity of the reaction medium. The use of propionic acid as a solvent further enhances the homogeneity of the reaction mixture, ensuring consistent heat transfer and mixing efficiency throughout the batch.
The strategic selection of N-(2-hydroxyethyl)nicotinamide as the precursor is pivotal to the success of this route. 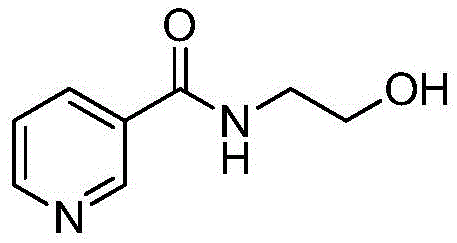
This intermediate is commercially available and chemically stable, providing a solid foundation for the subsequent nitration step. The reaction conditions are remarkably mild, requiring only standard cooling equipment rather than specialized cryogenic setups. Post-reaction workup is simplified to a quenching step with dilute ammonia water, which neutralizes residual acids and facilitates the crystallization of the product. This streamlined workflow not only improves the overall yield to over 80% but also ensures that the final product meets stringent purity specifications with minimal effort, representing a paradigm shift in the manufacturing of nitrate-based cardiovascular drugs.
Mechanistic Insights into Propionic Anhydride-Mediated Nitration
The core chemical transformation in this optimized synthesis is the esterification of the hydroxyl group on the ethanolamine side chain with the nitrate group. Mechanistically, the interaction between dilute nitric acid and propionic anhydride generates a highly reactive acetyl nitrate or propionyl nitrate species in situ. This mixed anhydride acts as a potent nitrating agent that is significantly more selective than free nitronium ions generated in strong mineral acids. The reaction proceeds through a nucleophilic attack by the alcohol oxygen of the N-(2-hydroxyethyl)nicotinamide on the nitrogen center of the activated nitrate species. This pathway is favored entropically and enthalpically under the mild thermal conditions employed, minimizing side reactions such as oxidation of the pyridine ring or hydrolysis of the amide bond.
Impurity control is inherently built into this mechanistic design. In traditional mixed-acid nitrations, the harsh acidic environment often promotes the degradation of the sensitive nitrate ester product or the formation of oxidative byproducts. However, the buffered nature of the propionic acid system maintains a pH environment that stabilizes the product once formed. Furthermore, the absence of heavy metal catalysts, which are required in some alternative green chemistry routes, eliminates the risk of metal contamination in the final API. This is critical for pharmaceutical applications where residual metal limits are strictly regulated. The high selectivity of the anhydride-mediated nitration ensures that the primary impurity profile is clean, facilitating easier purification and reducing the load on downstream chromatography or recrystallization steps.
How to Synthesize Nicorandil Efficiently
The synthesis of Nicorandil via this novel route is characterized by its operational simplicity and robustness, making it highly attractive for process chemists aiming to transfer technology from the lab to the pilot plant. The procedure involves the careful preparation of the nitrating mixture followed by the controlled addition of the substrate. Detailed standardized operating procedures regarding stoichiometry, addition rates, and temperature ramps are essential to maximize the yield and ensure batch-to-batch consistency. The following guide outlines the critical stages of this synthesis based on the optimized parameters found in the patent literature.
- Prepare the nitrating mixture by adding 68% dilute nitric acid to a reactor, cooling to 0-10°C, and slowly滴adding propionic acid and propionic anhydride while maintaining temperature.
- Add N-(2-hydroxyethyl)nicotinamide to the reaction system in batches, then allow the temperature to rise to 10-30°C and stir for 3-4 hours until HPLC monitoring shows completion.
- Quench the reaction by pouring the solution into 10% dilute ammonia water, cool to 10-15°C to induce crystallization, and filter to obtain the pure Nicorandil product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The shift away from hazardous reagents and complex catalytic systems translates directly into reduced operational risk and lower overhead costs. By utilizing commodity chemicals such as dilute nitric acid and propionic anhydride, manufacturers can decouple their supply chains from the volatility of specialized reagent markets. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients without interruption.
- Cost Reduction in Manufacturing: The elimination of expensive metal catalysts and the avoidance of cryogenic cooling requirements result in significant capital and operational expenditure savings. Traditional methods often require specialized reactors capable of withstanding extreme corrosion or low temperatures, whereas this process can be run in standard glass-lined or stainless steel vessels. Furthermore, the high atom economy of the reaction minimizes raw material waste, ensuring that a greater proportion of input costs are converted into valuable saleable product. The simplified workup procedure also reduces labor hours and solvent consumption during the isolation phase, contributing to a leaner overall manufacturing cost structure.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk-grade raw materials ensures a resilient supply chain that is less susceptible to geopolitical disruptions or supplier shortages. Unlike routes dependent on proprietary catalysts or unstable intermediates that require cold-chain logistics, the reagents for this process are stable and easily sourced from multiple vendors globally. This redundancy allows procurement teams to negotiate better pricing and secure long-term contracts with confidence. Additionally, the robustness of the reaction conditions means that production is less likely to be halted due to minor fluctuations in utility supplies or environmental conditions, guaranteeing a steady flow of material to downstream formulation units.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a substantial improvement over legacy technologies. The absence of concentrated sulfuric acid eliminates the generation of large volumes of spent acid waste, which is costly and difficult to treat. The mild reaction temperatures reduce the energy footprint of the process, aligning with corporate sustainability goals and carbon reduction targets. The process generates primarily aqueous waste streams that are easier to neutralize and treat, simplifying regulatory compliance and reducing the risk of environmental fines. This 'green' profile enhances the marketability of the final product to environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Nicorandil synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this route for industrial production.
Q: What are the safety advantages of this Nicorandil synthesis method compared to traditional mixed acid nitration?
A: This method avoids the use of concentrated sulfuric acid and concentrated nitric acid, which are highly corrosive and generate significant exothermic heat. By utilizing 68% dilute nitric acid combined with propionic anhydride, the reaction proceeds under much milder conditions (10-30°C), significantly reducing the risk of thermal runaway and simplifying waste acid treatment.
Q: How does the yield of this new process compare to prior art methods?
A: While traditional methods reported in literature often suffer from low selectivity and yields around 40%, this optimized protocol achieves yields exceeding 80% (specifically 82.3% to 85.9% in examples). This substantial improvement is due to the enhanced esterification capability of the nitric acid/anhydride system.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scale-up. It uses cheap and easily obtained raw materials, operates at safe temperatures without requiring cryogenic cooling or high pressure, and generates minimal environmental pollution, making it ideal for commercial manufacturing of cardiovascular intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicorandil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO specializing in complex pharmaceutical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our engineering teams are adept at optimizing reaction parameters to ensure that the theoretical benefits of the propionic anhydride nitration method are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Nicorandil meets the highest international quality standards.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing their cardiovascular drug supply chains. By leveraging our advanced process development capabilities, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can drive value for your organization and secure your position in the competitive global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
