Advanced Iron-Catalyzed Route for Scalable Production of 2-Trifluoromethyl Quinazolinone Derivatives
Advanced Iron-Catalyzed Route for Scalable Production of 2-Trifluoromethyl Quinazolinone Derivatives
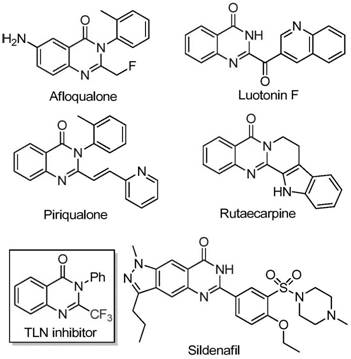
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring systems are ubiquitous in medicinal chemistry, found in numerous natural products and synthetic drug molecules exhibiting potent anti-cancer, anticonvulsant, anti-inflammatory, antifungal, and antimalarial activities. The strategic introduction of a trifluoromethyl group into these heterocyclic cores offers profound advantages, including significantly improved electronegativity, enhanced bioavailability, superior metabolic stability, and optimized lipophilicity of the target molecule. This patent presents a transformative approach that addresses long-standing challenges in synthesizing these valuable intermediates, leveraging a cost-effective iron-catalyzed cyclization strategy that promises to reshape supply chains for reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has been fraught with significant technical and economic hurdles that impede efficient manufacturing. Literature reports indicate that traditional pathways primarily rely on the cyclization reaction of specific synthons containing trifluoromethyl groups, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide, anthranilic acid, or isatoic anhydride. While chemically feasible, these conventional methods are severely limited by a constellation of disadvantages that render them suboptimal for modern industrial applications. The reaction conditions are often severe, requiring harsh reagents that pose safety risks and complicate process control. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, are frequently expensive and not readily available in bulk quantities, driving up the overall cost of goods. Perhaps most critically, these legacy routes often suffer from low yields and narrow substrate scope, meaning they fail to tolerate diverse functional groups, thereby restricting the chemical space accessible to medicinal chemists and limiting the designability of the final drug candidates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel methodology disclosed in the patent introduces a streamlined and economically viable pathway utilizing readily available trifluoroethylimidoyl chloride and isatin as the foundational starting raw materials. This innovative route employs a series of cyclization reactions catalyzed by cheap metal iron, specifically ferric chloride, which acts as a potent yet inexpensive Lewis acid promoter. The process is characterized by its operational simplicity and remarkable functional group tolerance, allowing for the synthesis of a wide array of derivatives with varying substituents on the aryl rings. By shifting away from precious metal catalysts and expensive anhydrides, this approach drastically simplifies the reaction setup and post-treatment procedures. The method demonstrates excellent reaction efficiency and applicability, capable of being expanded to the gram level with ease, thus providing a tangible possibility for seamless industrial scale application. This shift represents a paradigm change in how these critical heterocycles are manufactured, offering a sustainable alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway facilitated by the ferric chloride catalyst. The reaction initiates with the interaction between the trifluoroethylimidoyl chloride and isatin in the presence of a base, typically sodium hydride. During the initial phase, alkali-promoted carbon-nitrogen bond formation occurs between the imidoyl chloride and the isatin substrate, leading to the generation of a transient trifluoroacetamidine intermediate. This step is crucial as it sets the stage for the subsequent ring closure. Following this initial coupling, the system undergoes an iron-catalyzed decarbonylation and cyclization reaction. The ferric chloride likely coordinates with the carbonyl oxygen or the nitrogen centers, activating the molecule towards intramolecular nucleophilic attack and facilitating the loss of carbon monoxide. This isomerization process ultimately yields the stable 2-trifluoromethyl-substituted quinazolinone core. The use of 4A molecular sieves in the reaction mixture plays a vital role in scavenging moisture, ensuring that the sensitive imidoyl chloride and the reactive sodium hydride remain effective throughout the prolonged heating period, thereby maximizing conversion rates.
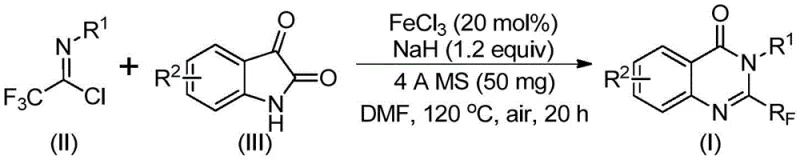
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-mediated cyclizations. The mild nature of the iron catalyst compared to strong Bronsted acids minimizes side reactions such as hydrolysis of the trifluoromethyl group or degradation of the sensitive isatin moiety. The specific stoichiometry employed, with a molar ratio of ferric chloride to sodium hydride optimized at approximately 0.2 to 1.2, ensures that the basic environment is sufficient to drive the initial deprotonation and coupling without causing excessive decomposition of the starting materials. Furthermore, the two-stage temperature profile—starting at a lower temperature of 40°C to allow for the initial coupling and then ramping to 120°C to drive the cyclization and decarbonylation—provides precise kinetic control. This thermal gradient helps in managing the exothermicity of the reaction and prevents the formation of polymeric byproducts or tar, which are common issues in high-temperature heterocycle synthesis. Consequently, the crude reaction mixture is cleaner, simplifying the downstream purification process and enhancing the overall purity profile of the final active pharmaceutical ingredient intermediate.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling in a standard laboratory or pilot plant setting. The protocol involves charging a reaction vessel with the catalyst system and drying agents before introducing the organic substrates. The choice of solvent is critical, with polar aprotic solvents like DMF, acetonitrile, or dioxane being preferred; among these, DMF has shown superior performance in effectively dissolving the starting materials and promoting the reaction to high conversion. The reaction timeline is carefully balanced, typically involving an initial stir at 40°C for 8 to 10 hours followed by heating to 120°C for 18 to 20 hours under an air atmosphere. This extended duration ensures complete consumption of the isatin and full cyclization. Post-reaction, the workup is straightforward, involving simple filtration to remove the molecular sieves and inorganic salts, followed by standard silica gel chromatography. For those seeking the detailed standardized operating procedures and specific stoichiometric ratios for various substrates, please refer to the comprehensive guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for an additional 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed synthesis route presents a compelling value proposition centered on cost optimization and supply security. The transition from expensive, specialized reagents to commodity chemicals fundamentally alters the cost structure of producing these high-value intermediates. By eliminating the dependency on scarce resources and complex multi-step precursor syntheses, manufacturers can achieve substantial cost savings that can be passed down the supply chain or reinvested into further R&D. Moreover, the robustness of the reaction conditions implies a lower risk of batch failures, which is a critical metric for maintaining consistent inventory levels and meeting delivery commitments to downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the replacement of costly catalysts and reagents. Traditional methods often rely on precious metals or expensive fluorinating agents that contribute significantly to the bill of materials. In this novel process, ferric chloride is utilized, which is abundant and inexpensive compared to palladium or rhodium alternatives. Additionally, the starting materials—isatin and trifluoroethylimidoyl chloride—are commercially available in large quantities at competitive prices. The elimination of expensive heavy metal removal steps, which are mandatory when using precious metal catalysts to meet regulatory limits for residual metals in APIs, further reduces processing costs. This streamlined approach allows for a drastic simplification of the purification workflow, directly translating to lower operational expenditures and a more favorable margin structure for the final product.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability of raw materials. The substrates used in this method, such as isatin and various aromatic amines required to make the imidoyl chlorides, are produced on a massive global scale for the dye and agrochemical industries. This widespread availability ensures that there are multiple qualified sources for these inputs, mitigating the risk of supply disruptions caused by single-source dependencies. Furthermore, the reaction tolerates a wide range of functional groups, meaning that a single robust platform technology can be used to produce a diverse library of analogues without needing to requalify entirely new synthetic routes for each derivative. This flexibility allows suppliers to respond rapidly to changing market demands or specific client requests for novel analogues without incurring significant lead time penalties.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this method is inherently designed for scalability. The use of common solvents like DMF and the absence of highly hazardous reagents like gaseous chlorine or pyrophoric organometallics simplify the engineering controls required for large-scale reactors. The reaction proceeds under air, removing the need for expensive inert gas blanketing systems often required for sensitive organometallic chemistry. From an environmental standpoint, the use of iron, a non-toxic and environmentally benign metal, aligns well with green chemistry principles and increasingly stringent environmental regulations. The simplified workup reduces the volume of solvent waste and silica gel usage, contributing to a lower environmental footprint and easier compliance with waste disposal regulations, which is a growing concern for sustainable manufacturing operations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential integration into their supply chains. The following questions address common inquiries regarding the practical implementation and capabilities of this technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this iron-catalyzed method over traditional synthesis?
A: This method utilizes inexpensive and readily available starting materials like isatin and trifluoroethylimidoyl chloride, avoiding expensive trifluoroacetic anhydride. It employs cheap ferric chloride instead of precious metal catalysts, significantly reducing raw material costs while maintaining high functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application. The use of common solvents like DMF and simple post-treatment procedures like filtration and column chromatography supports scalability.
Q: What represents the key structural feature of the products synthesized?
A: The core structure is a quinazolinone ring substituted with a trifluoromethyl group at the 2-position. This modification enhances electronegativity, bioavailability, and metabolic stability, making these compounds valuable scaffolds for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the demand for fluorinated heterocycles continues to surge in the development of novel therapeutics, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2-trifluoromethyl quinazolinone meets the exacting standards required for clinical and commercial applications. We understand that consistency is key in pharmaceutical manufacturing, and our robust quality management systems are designed to deliver that reliability consistently.
We invite you to leverage our technical capabilities to optimize your supply chain for these critical intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact our technical procurement team to request specific COA data for our catalog items or to discuss route feasibility assessments for custom synthesis projects. Let us collaborate to bring your next-generation drug candidates to market faster and more efficiently.
