Advanced Iodine-Promoted Cyclization for High-Purity 3-Trifluoromethyl-1,2,4-Triazole Derivatives
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing fluorinated heterocycles, driven by the critical role these motifs play in enhancing metabolic stability and bioavailability. Patent CN114920707B, published in April 2023, introduces a transformative approach to synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, a core scaffold found in blockbuster drugs such as Sitagliptin and various Factor IXa inhibitors. This innovation addresses long-standing synthetic challenges by leveraging dimethylformamide (DMF) not merely as a solvent, but as an active carbon source in an iodine-promoted tandem cyclization. For R&D directors and procurement specialists, this represents a paradigm shift towards more atom-economical and operationally simple processes that can be seamlessly integrated into existing manufacturing lines without requiring specialized anhydrous or anaerobic infrastructure.
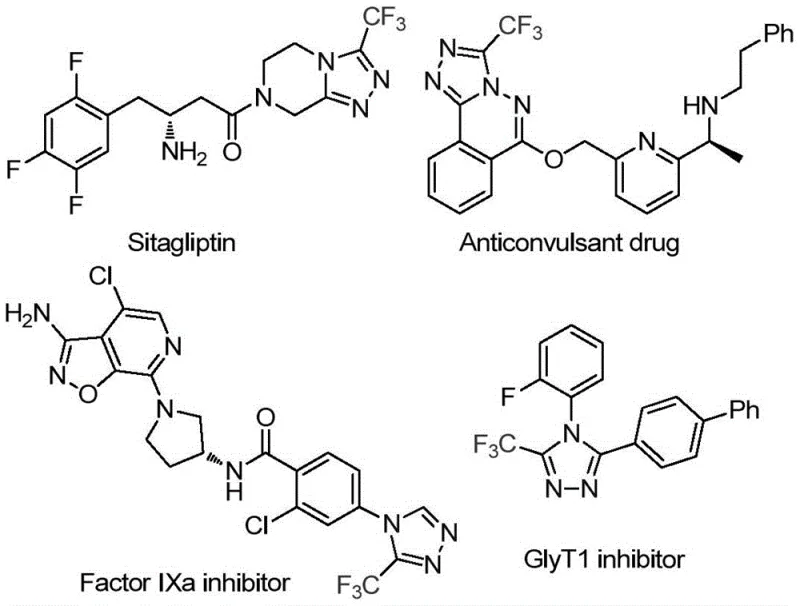
The strategic value of this technology lies in its ability to access diverse substitution patterns at the 4-position of the triazole ring through simple substrate design. As illustrated in the provided structural data, the presence of the trifluoromethyl group significantly modulates the physicochemical properties of the parent molecule, often leading to improved pharmacokinetics. By establishing a reliable supply chain for these high-purity intermediates, manufacturers can accelerate the development of next-generation therapeutics while mitigating the risks associated with complex multi-step syntheses that traditionally plague this chemical class.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on harsh reaction conditions that pose significant safety and scalability hurdles for industrial applications. Traditional routes often necessitate the use of expensive transition metal catalysts, strictly anhydrous environments, and hazardous reagents that generate substantial toxic waste streams. These conventional methods frequently suffer from poor atom economy, requiring multiple protection and deprotection steps that drastically reduce overall yield and increase the cost of goods sold (COGS). Furthermore, the sensitivity of many fluorinated intermediates to moisture and oxygen demands sophisticated reactor setups and rigorous quality control measures, creating bottlenecks in the supply chain that can delay time-to-market for critical API projects.
The Novel Approach
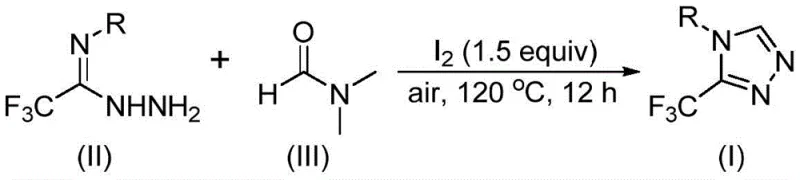
In stark contrast, the methodology disclosed in CN114920707B offers a streamlined, one-pot solution that utilizes molecular iodine as a mild promoter under ambient air conditions. The core innovation involves the activation of DMF, a ubiquitous and inexpensive industrial solvent, which acts as a C1 synthon to construct the triazole ring directly from trifluoroethyliminohydrazide precursors. This eliminates the need for external formylating agents and reduces the number of unit operations required for purification. The reaction operates efficiently at temperatures between 110°C and 130°C, demonstrating remarkable tolerance to various functional groups on the aromatic ring. This robustness allows for the synthesis of a wide array of derivatives, including those with electron-rich and electron-deficient substituents, without compromising yield or purity.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic pathway is crucial for optimizing this process for commercial scale-up. The reaction proceeds through a fascinating dual-pathway mechanism depending on which fragment of the DMF molecule participates. In the primary pathway, the formyl group of DMF undergoes condensation with the trifluoroethyliminohydrazide to generate a hydrazone intermediate. This is followed by an intramolecular cyclization event where a molecule of dimethylamine is eliminated, directly yielding the 3-trifluoromethyl-substituted 1,2,4-triazole product. Alternatively, the N-methyl group of DMF can serve as the carbon source; here, DMF reacts with iodine to form an amine salt, which subsequently undergoes nucleophilic addition and elimination steps to generate an azadiene intermediate before oxidative aromatization occurs. This mechanistic flexibility ensures high conversion rates even with varying substrate electronic properties.
From an impurity control perspective, the use of molecular iodine is particularly advantageous as it facilitates clean oxidative aromatization without generating heavy metal residues that are difficult to remove in downstream processing. The reaction profile indicates that side reactions are minimized due to the specific activation of the DMF C-H bonds by the iodine species. For quality assurance teams, this means the impurity profile is predictable and manageable, primarily consisting of unreacted starting materials that can be easily removed via standard silica gel chromatography or crystallization. The absence of transition metals simplifies the regulatory filing process, as there is no need for extensive testing for residual palladium or copper, which are common pain points in traditional cross-coupling approaches.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemistry teams looking to transfer technology from the lab to the pilot plant. The procedure involves charging a reaction vessel with the hydrazide substrate, molecular iodine, and DMF, followed by heating under air. The detailed standardized synthesis steps, including precise stoichiometry and workup protocols, are outlined below to ensure reproducibility and safety during implementation.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel purification to isolate the target triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based cyclization strategy offers tangible benefits that extend beyond mere chemical elegance. The primary driver for cost optimization is the dual functionality of DMF, which serves as both the reaction medium and a reactant, thereby reducing the total volume of chemicals required and minimizing solvent recovery costs. Since DMF is a commodity chemical produced on a massive global scale, its price stability and availability are far superior to specialized reagents used in alternative synthetic routes. This inherent raw material security insulates the manufacturing process from supply shocks that often affect niche catalysts or fluorinating agents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the removal of stringent inert atmosphere requirements lead to substantial operational expenditure savings. By conducting the reaction in air rather than under nitrogen or argon, facilities can utilize standard glass-lined reactors without modification, significantly lowering capital expenditure barriers. Furthermore, the simplified workup procedure, which typically involves filtration and chromatography, reduces labor hours and solvent consumption during purification, directly impacting the bottom line through improved process efficiency.
- Enhanced Supply Chain Reliability: The starting materials, specifically the trifluoroethyliminohydrazides, are readily accessible or can be synthesized from common aromatic amines and trifluoroacetic acid derivatives. This reliance on bulk chemicals ensures a stable supply chain with multiple qualified vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor fluctuations in environmental controls, ensuring consistent on-time delivery for downstream API synthesis campaigns.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the use of high-boiling solvents like DMF which allow for safe operation at elevated temperatures without excessive pressure buildup. The waste stream is significantly cleaner compared to metal-catalyzed alternatives, as it primarily contains organic salts and iodine byproducts that are easier to treat. This aligns with increasingly strict environmental regulations regarding heavy metal discharge, positioning manufacturers who adopt this technology as leaders in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis method. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear framework for evaluating feasibility within your specific project requirements.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive formylating agents and significantly simplifying the reaction setup.
Q: Does this process require strict anaerobic conditions?
A: No, the patent explicitly states that the reaction proceeds efficiently under an air atmosphere, removing the operational complexity and cost associated with inert gas protection like nitrogen or argon.
Q: What is the substrate scope for the R group in the hydrazide?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like halogens and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your upcoming projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this iodine-promoted route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
