Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
Introduction to Patent CN114920707B
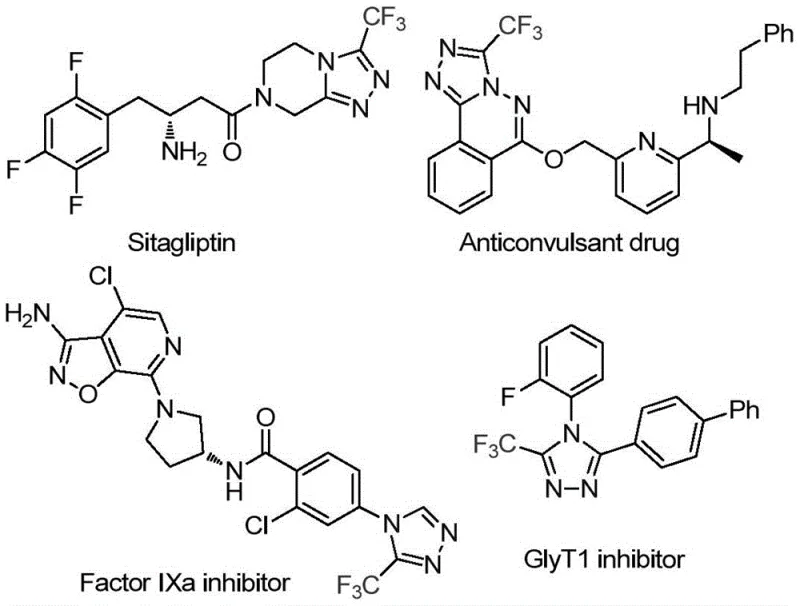
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, a structural motif prevalent in high-value bioactive molecules such as Factor IXa inhibitors and GlyT1 inhibitors. This innovation addresses a significant gap in current organic synthesis by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source. By leveraging molecular iodine as a promoter under aerobic conditions, this technology enables the construction of complex fluorinated heterocycles with remarkable simplicity and efficiency. For R&D directors and procurement specialists, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of essential pharmaceutical intermediates, eliminating the reliance on exotic reagents or苛刻 reaction conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has often involved multi-step sequences requiring specialized reagents that are both costly and hazardous to handle on a large scale. Conventional methodologies frequently necessitate the use of pre-functionalized C1 synthons or harsh cyclization conditions that demand strictly anhydrous and oxygen-free environments, thereby increasing operational complexity and infrastructure costs. Furthermore, many existing protocols suffer from limited substrate scope, failing to accommodate diverse functional groups without significant yield erosion or extensive purification efforts. These limitations create bottlenecks in the supply chain for high-purity API intermediates, leading to extended lead times and inflated production budgets that ultimately impact the commercial viability of new drug candidates targeting metabolic disorders or neurological conditions.
The Novel Approach
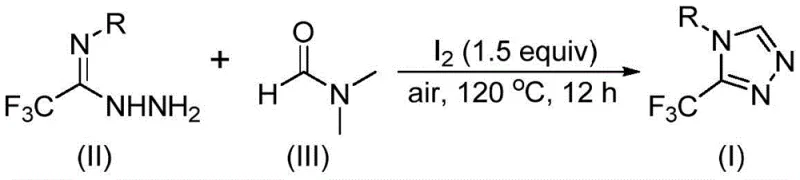
In stark contrast, the methodology disclosed in CN114920707B offers a streamlined, one-pot tandem cyclization strategy that drastically simplifies the synthetic landscape. The core innovation lies in the dual functionality of DMF, which acts simultaneously as the polar aprotic solvent and the source of the methine carbon required for ring closure. As illustrated in the reaction scheme below, the process utilizes readily available trifluoroethyliminohydrazide and molecular iodine under ambient air, reacting at moderate temperatures of 110-130°C. This approach not only reduces the number of unit operations but also mitigates safety risks associated with handling pyrophoric reagents. The ability to synthesize a wide array of 4-substituted 3-trifluoromethyl-1,2,4-triazoles through simple substrate design underscores the versatility of this method, making it an ideal candidate for the rapid generation of compound libraries and the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, this transformation is a fascinating example of solvent-participating catalysis where molecular iodine plays a pivotal role in activating the DMF molecule. The reaction pathway bifurcates depending on which moiety of the DMF molecule participates; when the formyl group acts as the carbon source, it undergoes condensation with the hydrazide to form a hydrazone intermediate, followed by intramolecular cyclization and elimination of dimethylamine. Alternatively, when the N-methyl group serves as the carbon source, iodine facilitates the formation of an amine salt, which subsequently reacts via nucleophilic addition and oxidative aromatization. This dual-pathway capability ensures high conversion rates across a broad spectrum of electronic environments on the aromatic ring, providing R&D teams with a reliable tool for optimizing reaction yields without needing to switch catalysts or solvents.
Furthermore, the impurity profile generated by this method is exceptionally clean, primarily consisting of volatile byproducts like dimethylamine or iodinated species that are easily removed during standard aqueous workups. The absence of transition metal catalysts, such as palladium or copper, which are common in cross-coupling approaches, eliminates the risk of heavy metal contamination in the final API. This is a critical quality attribute for regulatory compliance, as it simplifies the purification process and ensures that the resulting high-purity 1,2,4-triazole derivatives meet stringent pharmacopeial standards. The robustness of the oxidative aromatization step under air atmosphere further confirms the thermodynamic favorability of the triazole ring formation, ensuring consistent batch-to-batch reproducibility essential for GMP manufacturing.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires minimal specialized equipment, relying instead on standard glassware and heating mantles capable of sustaining temperatures up to 130°C. The procedure begins with the precise weighing of trifluoroethyliminohydrazide and molecular iodine, typically in a molar ratio of 1:1.5, which are then dissolved in excess DMF to drive the equilibrium towards product formation. The reaction mixture is stirred vigorously under open air conditions for a duration of 10 to 15 hours, allowing sufficient time for the tandem cyclization and aromatization processes to reach completion. Following the reaction, the crude product is isolated through simple filtration or extraction, followed by purification via silica gel column chromatography to afford the target compound in high purity. For detailed standardized operating procedures and safety guidelines regarding this specific transformation, please refer to the technical guide below.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel under air atmosphere.
- Heat the reaction mixture to 110-130°C and maintain stirring for 10-15 hours to ensure complete cyclization.
- Perform post-treatment via filtration, washing, and silica gel column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic reduction of raw material costs, as DMF is one of the most inexpensive and widely available industrial solvents, effectively replacing costly specialized reagents. By eliminating the need for noble metal catalysts and inert gas protection systems, the process significantly lowers the capital expenditure required for reactor setup and maintenance, translating into substantial cost savings in API manufacturing. Additionally, the simplified workup procedure reduces solvent consumption and waste generation, aligning with green chemistry principles and reducing the environmental burden associated with chemical production.
- Cost Reduction in Manufacturing: The utilization of DMF as a dual-purpose reagent removes the necessity for purchasing separate C1 building blocks, which are often priced at a premium due to their specialized nature. Moreover, the use of molecular iodine as a promoter is far more economical than transition metal catalysts, and its recovery or disposal is less regulated and costly. This combination of low-cost inputs and high atom economy directly contributes to a lower cost of goods sold (COGS), enabling competitive pricing strategies for generic drug manufacturers and contract development organizations seeking to optimize their margins without compromising quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various substituted aromatic amines and hydrazines, are commodity chemicals with established global supply chains, minimizing the risk of shortages or delivery delays. Unlike methods relying on custom-synthesized precursors with long lead times, this approach allows for rapid procurement and inventory turnover. The robustness of the reaction under air atmosphere further enhances reliability by removing the dependency on nitrogen or argon supply lines, ensuring that production can continue uninterrupted even in facilities with limited utility infrastructure, thereby securing the continuity of supply for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is straightforward due to the absence of exothermic hazards associated with strong bases or pyrophoric reagents. The reaction operates at moderate temperatures and atmospheric pressure, reducing the engineering controls needed for high-pressure reactors. From an environmental standpoint, the process generates minimal hazardous waste, as the primary byproducts are manageable organic salts and volatiles. This ease of waste treatment facilitates compliance with increasingly stringent environmental regulations, avoiding costly fines and shutdowns, and positioning the manufacturing site as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this novel triazole synthesis method. These insights are derived directly from the experimental data and scope defined in the patent literature, aiming to clarify feasibility for potential licensees or manufacturing partners. Understanding these nuances is crucial for project planning and risk assessment when integrating new chemistries into existing production pipelines.
Q: What are the key advantages of using DMF as a carbon source in this synthesis?
A: Using DMF eliminates the need for separate, expensive C1 building blocks. It serves a dual role as both the reaction medium and the reactant, significantly simplifying the supply chain and reducing raw material costs while maintaining high reaction efficiency.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the major operational benefits of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly facilitating industrial scale-up.
Q: What is the substrate scope for the R group in the trifluoroethyliminohydrazide?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituted aryl groups including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, trifluoromethyl) substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted cyclization technology for the production of high-value fluorinated heterocycles. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and material requirements of this DMF-based process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cost-effective synthesis for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and economic efficiency.
