Advanced Catalytic Strategy for Scalable 2-Pyridone Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access complex heterocyclic scaffolds, particularly the 2-pyridone motif which is prevalent in numerous bioactive natural products and drug candidates. A significant breakthrough in this domain is detailed in patent CN111925320A, which discloses a highly efficient method for synthesizing polysubstituted 2-pyridone compounds. This innovative approach leverages a palladium-catalyzed strategy involving norbornene derivatives to achieve multi-functionalization in a single operational step. By utilizing simple 4-iodo-2-pyridone precursors alongside various electrophiles and nucleophiles, this technology overcomes the limitations of classical synthesis, offering a robust pathway for generating high-value pharmaceutical intermediates. The ability to construct multiple chemical bonds simultaneously represents a paradigm shift in how chemists approach the assembly of these critical molecular architectures.
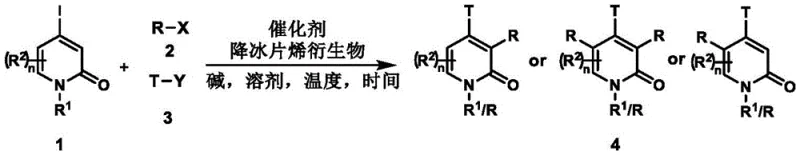
For R&D directors and process chemists, understanding the limitations of conventional methods is crucial for evaluating new technologies. Traditionally, the synthesis of 2-pyridone derivatives has relied on methods such as the hydrolysis of pyridines or classical electrophilic substitution reactions. However, these legacy approaches often suffer from significant drawbacks, including limited substrate scope, poor regioselectivity, and harsh reaction conditions that can degrade sensitive functional groups. Furthermore, strategies based on acyclic precursors often require specialized starting materials that are not commercially available, increasing the complexity and cost of the supply chain. In contrast, the novel approach described in the patent utilizes a convergent synthesis strategy. By employing a palladium catalyst in conjunction with a norbornene co-catalyst, the method enables the simultaneous formation of carbon-carbon and carbon-heteroatom bonds. This not only streamlines the synthetic route but also significantly enhances the overall yield and purity of the final product, addressing the critical need for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd/Norbornene Cooperative Catalysis
The core of this technological advancement lies in the sophisticated mechanism of palladium/norbornene cooperative catalysis, often referred to as Catellani-type chemistry. In this catalytic cycle, the palladium species initially undergoes oxidative addition with the 4-iodo-2-pyridone substrate. The presence of the norbornene derivative is pivotal, as it facilitates the ortho-C-H activation of the pyridone ring, a step that is typically challenging to achieve with high selectivity using standard cross-coupling protocols. This transient palladacycle intermediate then reacts with the electrophile, followed by the insertion of the nucleophile, before undergoing reductive elimination to release the polysubstituted product and regenerate the active catalyst. This intricate dance of organometallic steps allows for the precise installation of substituents at specific positions on the pyridone ring, providing chemists with unprecedented control over molecular architecture. The mechanism ensures that side reactions are minimized, leading to a cleaner reaction profile and simplifying downstream purification processes.
From an impurity control perspective, this mechanism offers distinct advantages over traditional multi-step syntheses. Because the reaction constructs multiple bonds in a single pot, there are fewer isolation steps where impurities can accumulate or where yield losses typically occur. The high chemoselectivity of the palladium-norbornene system means that sensitive functional groups present on the electrophiles or nucleophiles, such as esters, nitriles, or halogens, remain intact throughout the transformation. This tolerance is essential for late-stage functionalization of complex drug molecules. Moreover, the absence of additional phosphine ligands, which are often required in standard palladium catalysis, eliminates a common source of metal contamination and difficult-to-remove byproducts. This results in a final product with a superior impurity profile, reducing the burden on quality control laboratories and ensuring that the material meets the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Polysubstituted 2-Pyridones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize efficiency and yield. The process begins with the selection of appropriate starting materials, specifically the 4-iodo-2-pyridone derivative, which serves as the central scaffold. The reaction is typically conducted under an inert atmosphere, such as nitrogen, to prevent oxidation of the catalyst and reagents. A combination of a palladium source, such as palladium acetate, and a norbornene derivative is dissolved in a suitable organic solvent like 1,4-dioxane or dimethoxyethane. The addition of a base, such as potassium carbonate, is critical to neutralize the acid byproducts generated during the coupling. The detailed standardized synthesis steps are outlined in the guide below, providing a clear roadmap for technical teams to replicate the high yields reported in the patent data.
- Prepare the reaction mixture by combining 4-iodo-2-pyridone derivatives, electrophiles, nucleophiles, palladium catalyst, norbornene derivative, and base in an organic solvent under inert atmosphere.
- Heat the reaction mixture to a temperature between 50°C and 150°C and stir for up to 48 hours to facilitate the coupling and C-H activation processes.
- Upon completion, cool the mixture, filter through celite, remove solvents under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial simplification of the raw material supply chain. Since the method utilizes readily available commercial reagents such as alkyl halides, aryl halides, and simple alkenes or alkynes, there is no dependency on exotic or custom-synthesized starting materials that often carry long lead times and high price tags. This accessibility ensures a more stable and resilient supply chain, mitigating the risks associated with raw material shortages. Furthermore, the elimination of expensive phosphine ligands, which are often air-sensitive and costly to store and handle, directly contributes to cost reduction in API manufacturing. The operational simplicity of the process also means that training requirements for production staff are reduced, and the risk of batch failure due to handling errors is minimized.
Enhanced supply chain reliability is another critical factor driven by the robustness of this chemical process. The reaction conditions are relatively mild, operating effectively between 50°C and 150°C, which reduces the energy consumption compared to high-temperature processes. This thermal efficiency not only lowers utility costs but also extends the lifespan of reactor equipment. Additionally, the high step economy—building two or three bonds in one go—drastically reduces the total processing time. Fewer reaction steps mean fewer unit operations, less solvent usage, and reduced waste generation, aligning perfectly with modern environmental compliance standards. This green chemistry profile facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing site. Consequently, companies can achieve commercial scale-up of complex pharmaceutical intermediates with greater confidence and speed, ensuring that production timelines are met without compromising on quality or sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, based on the detailed data provided in the patent documentation. These insights are designed to clarify the feasibility and benefits of implementing this route for your specific project needs. Understanding these aspects will help stakeholders make informed decisions about integrating this methodology into their existing production workflows.
Q: What are the key advantages of this synthesis method over traditional approaches?
A: This method allows for the construction of two or three chemical bonds in a single step, offering superior step economy compared to traditional methods that often require multiple sequential reactions. It also exhibits excellent functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available commercial reagents, mild reaction conditions (50-150°C), and avoids expensive phosphine ligands, making it highly adaptable for commercial scale-up and industrial manufacturing.
Q: What types of substituents can be introduced using this methodology?
A: The methodology supports a wide range of electrophiles such as alkyl halides and aryl halides, and nucleophiles including alkenes, alkynes, and boronic esters, allowing for diverse structural modifications of the 2-pyridone core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyridone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111925320A for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-pyridone intermediate delivered meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this Pd-catalyzed process for your specific molecule, ensuring maximum yield and minimal impurity levels.
We invite you to collaborate with us to unlock the full commercial potential of your drug candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating how this efficient synthesis route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timeline and secure a reliable supply of high-quality intermediates for your global operations.
