Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses the growing demand for reliable pharmaceutical intermediate suppliers capable of delivering complex heterocyclic scaffolds with high purity and consistency. The 1,2,4-triazole core is a privileged structure found in numerous bioactive molecules, including notable drugs such as Sitagliptin, as illustrated in the structural diversity of modern therapeutics. By integrating a trifluoromethyl group at the 3-position, chemists can drastically alter the physicochemical properties of the parent molecule, including lipophilicity and electronegativity, thereby optimizing drug candidates for clinical success.
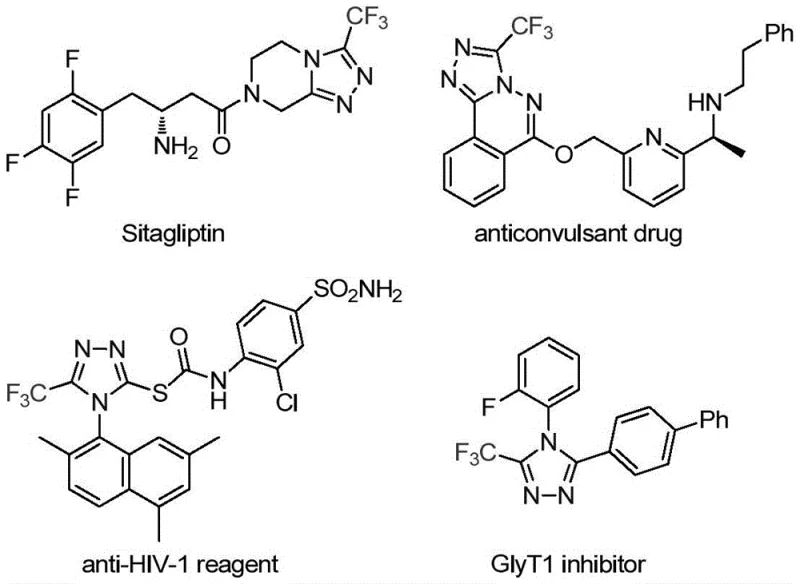
The strategic importance of this patent lies in its ability to streamline the synthesis of these valuable intermediates. Traditional routes often suffer from limitations such as multi-step sequences, hazardous reagents, or poor atom economy. In contrast, the methodology described in CN113307778A utilizes a novel transition metal co-catalytic system involving molybdenum and copper. This approach not only simplifies the operational workflow but also expands the applicability of the reaction to a wide range of substrates. For R&D directors and procurement managers alike, understanding the nuances of this synthetic route is essential for evaluating potential supply chain partners who can offer cost reduction in API manufacturing through innovative chemistry. The ability to access diverse 3-trifluoromethyl-1,2,4-triazoles efficiently positions this technology as a cornerstone for next-generation drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on several classical strategies that present significant challenges for industrial application. One common pathway involves the cyclization reaction of trifluoroacetyl hydrazine with amidine compounds, a process that often requires stringent conditions and generates substantial waste. Another prevalent method is the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, which can be cumbersome due to the sensitivity of the intermediates and the need for careful temperature control. Furthermore, copper-catalyzed multi-component reactions utilizing diazonium salts, trifluorodiazoethane, and nitriles have been reported, but these often involve hazardous diazo compounds that pose safety risks on a large scale. Additionally, methods employing the cyclization of trifluoroethylimidoyl chloride with aldehyde hydrazones and hydrazides frequently result in fully substituted triazoles, limiting the ability to synthesize specific 3,4-disubstituted variants required for certain biological targets. These conventional approaches often lack the versatility and safety profile demanded by modern green chemistry standards, leading to higher production costs and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The invention disclosed in patent CN113307778A introduces a paradigm shift by employing a molybdenum and copper co-catalyzed cycloaddition reaction. This novel approach utilizes trifluoroethylimidoyl chloride and functionalized isonitrile (specifically Ph3P=N-NC) as the primary building blocks. The reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C, in common organic solvents such as tetrahydrofuran (THF). Unlike previous methods that might require exotic or unstable reagents, this strategy leverages commercially available starting materials, significantly lowering the barrier to entry for production. The use of molybdenum hexacarbonyl as a metal activator in conjunction with cuprous acetate creates a synergistic catalytic environment that promotes the formation of the five-membered triazole ring with high efficiency. This method is particularly advantageous because it allows for the design and synthesis of various functionalized 1,2,4-triazole compounds simply by modifying the R group on the imidoyl chloride, thus offering unparalleled flexibility for medicinal chemists exploring structure-activity relationships.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic pathway of this transformation is a sophisticated interplay between the molybdenum and copper catalysts, which orchestrate the assembly of the triazole ring from simple precursors. Initially, molybdenum hexacarbonyl interacts with the functionalized isonitrile to form a reactive metal-isocyanide complex. This activation step is crucial as it enhances the nucleophilicity of the isonitrile carbon, preparing it for the subsequent cycloaddition event. Concurrently, the cuprous acetate acts as a promoter for the [3+2] cycloaddition reaction between the activated isonitrile species and the trifluoroethylimidoyl chloride. This step leads to the formation of a five-membered ring intermediate, effectively stitching together the carbon and nitrogen atoms into the desired heterocyclic framework. The presence of triethylamine as a base facilitates the deprotonation steps necessary to drive the equilibrium forward, while also neutralizing any acidic byproducts generated during the reaction. Finally, the intermediate undergoes elimination of triphenylphosphine oxide, likely assisted by trace water in the system or during workup, to yield the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This elegant mechanism avoids the formation of toxic hydrazine byproducts and ensures a cleaner reaction profile.
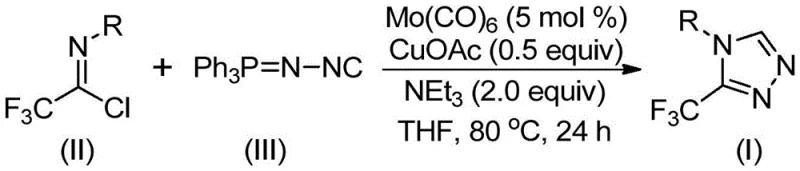
From an impurity control perspective, this catalytic system offers distinct advantages over stoichiometric methods. The use of catalytic amounts of metals (5 mol % Mo and 0.5 equiv Cu) minimizes the metal load in the final product, simplifying downstream purification processes. The reaction demonstrates high tolerance for various functional groups, including halogens (fluoro, chloro), alkyl groups (methyl, ethyl, t-butyl), and electron-withdrawing groups (nitro), without significant side reactions or decomposition. The patent data indicates that even sterically hindered substrates, such as those with ortho-substitution or bulky t-butyl groups, can be converted to the corresponding triazoles, albeit with varying yields. This robustness suggests that the catalytic cycle is resilient against steric congestion, likely due to the flexible coordination geometry of the molybdenum center. Furthermore, the choice of THF as the solvent ensures good solubility for both polar and non-polar intermediates, preventing precipitation that could otherwise halt the reaction or trap impurities. For quality control teams, this means a more predictable impurity profile and easier validation of the manufacturing process.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory research and pilot plant production. The standard procedure involves charging a reaction vessel, such as a Schlenk tube, with the catalysts, base, and solvent under an inert atmosphere to prevent oxidation of the sensitive metal centers. The reactants, trifluoroethylimidoyl chloride and the functionalized isonitrile, are then introduced, and the mixture is heated to the optimal temperature range of 80°C for approximately 24 hours. Monitoring the reaction progress via TLC or HPLC ensures complete conversion before proceeding to workup. The post-treatment is straightforward, involving filtration to remove solid residues followed by silica gel mixing and column chromatography purification. This simplicity reduces the technical burden on operators and minimizes the risk of product loss during isolation. For detailed standardized operating procedures and specific stoichiometric ratios tailored to different substrates, please refer to the comprehensive guide below.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in anhydrous THF.
- Add trifluoroethylimidoyl chloride (1.0 equiv) and functionalized isonitrile (1.5 equiv) to the mixture under inert atmosphere.
- Heat the reaction mixture to 80°C and stir for 24 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The shift towards this Mo/Cu co-catalyzed method represents a strategic move away from expensive and hazardous reagents towards a more sustainable and cost-effective manufacturing model. By utilizing readily available starting materials like trifluoroethylimidoyl chloride and functionalized isonitriles, companies can mitigate the risks associated with raw material scarcity and price volatility. The elimination of complex multi-step sequences reduces the overall processing time and labor costs, allowing for faster turnaround times on custom synthesis orders. Moreover, the mild reaction conditions (70-90°C) lower energy consumption compared to high-temperature or cryogenic processes, contributing to a smaller carbon footprint and reduced utility expenses. These factors collectively enhance the competitiveness of suppliers who can leverage this technology to offer better pricing and reliability to their clients.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive catalysts such as cuprous acetate and triethylamine, which are commodity chemicals available in bulk quantities. Unlike methods requiring precious metals like palladium or rhodium, the copper and molybdenum system significantly lowers the catalyst cost per kilogram of product. Additionally, the high reaction efficiency and yields (often exceeding 90% for optimized substrates) minimize waste generation and maximize raw material utilization. The simplified workup procedure, which avoids complex extractions or crystallizations in favor of standard chromatography, further reduces solvent consumption and disposal costs. Consequently, manufacturers can achieve substantial cost savings in API manufacturing, passing these benefits on to downstream customers through competitive pricing structures.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, and this synthesis method bolsters reliability by relying on a robust portfolio of commercial reagents. The starting materials, including various substituted aromatic amines and trifluoroacetic acid derivatives, are produced by multiple global vendors, reducing dependency on single-source suppliers. The scalability of the reaction, demonstrated from milligram to gram scales in the patent, indicates a clear path to tonnage production without fundamental changes to the process chemistry. This scalability ensures that suppliers can meet fluctuating demand volumes without compromising on quality or delivery schedules. Furthermore, the stability of the intermediates and the final triazole products facilitates easier storage and transportation, reducing the risk of degradation during logistics and ensuring consistent quality upon arrival at the customer's facility.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental protection intensify, the green chemistry attributes of this method become increasingly valuable. The reaction operates in THF, a solvent that can be efficiently recovered and recycled, minimizing volatile organic compound (VOC) emissions. The absence of highly toxic hydrazines or explosive diazo compounds simplifies safety compliance and reduces the need for specialized containment infrastructure. The high atom economy of the cycloaddition reaction means less chemical waste is generated per unit of product, aligning with corporate sustainability goals. For facilities aiming to expand capacity, the straightforward nature of the reaction allows for easy adaptation to larger reactors, supporting the commercial scale-up of complex pharmaceutical intermediates. This alignment with environmental and safety standards future-proofs the supply chain against tightening regulations.
Frequently Asked Questions (FAQ)
To assist stakeholders in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the technical details provided in the patent literature. These inquiries address common concerns regarding reaction scope, safety, and scalability, providing clarity for decision-makers. Understanding these aspects is crucial for integrating this synthesis route into existing production pipelines or R&D workflows. The answers below reflect the empirical data and observations recorded during the development of this method, ensuring accuracy and relevance.
Q: What are the key advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods requiring harsh hydrazinolysis or unstable diazonium salts, this patent describes a mild one-pot cycloaddition at 70-90°C using commercially available imidoyl chlorides and isonitriles, significantly improving safety and operational simplicity.
Q: Can this synthesis method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates excellent substrate scope, successfully synthesizing derivatives with electron-donating groups (methyl, methoxy, t-butyl) and electron-withdrawing groups (fluoro, chloro, nitro) with yields ranging from moderate to excellent (up to 99%).
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly states the method is scalable to gram-level reactions with simple post-processing (filtration and chromatography), utilizing cheap catalysts like cuprous acetate and common solvents like THF, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate seamlessly into industrial reality. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole intermediates that meet stringent purity specifications required by global regulatory bodies. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing consistency and reliability for our partners. By leveraging the innovative Mo/Cu co-catalyzed technology described in CN113307778A, we can offer customized solutions that optimize both cost and quality for your specific project needs.
We invite you to collaborate with us to explore the full potential of this cutting-edge synthesis platform. Whether you require small quantities for preclinical studies or large volumes for commercial manufacturing, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and securing a stable supply of critical pharmaceutical intermediates.
