Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Novel Molybdenum-Copper Catalysis
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Novel Molybdenum-Copper Catalysis
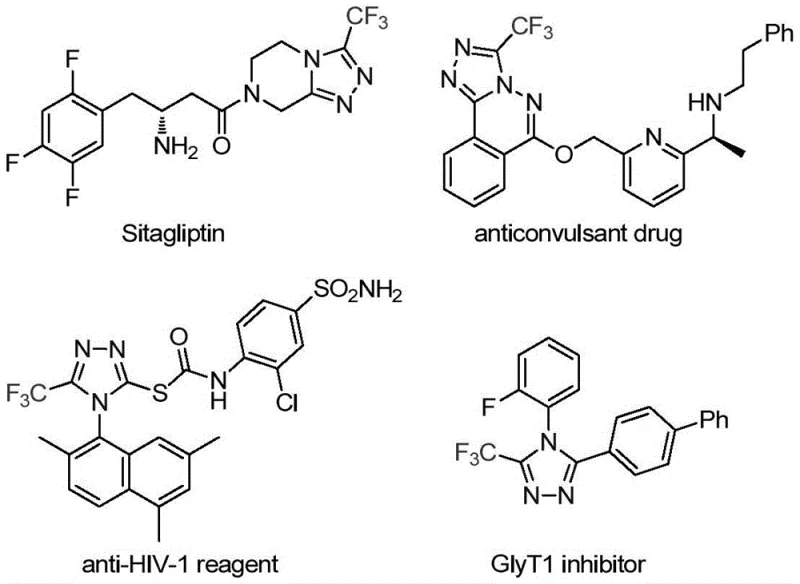
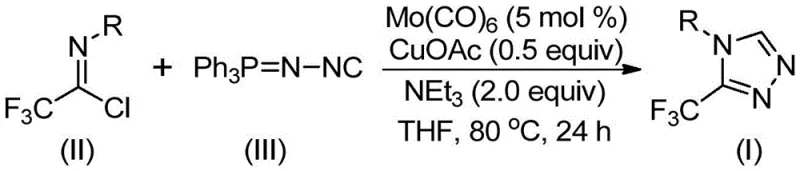
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. A groundbreaking development in this sector is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward for manufacturers aiming to produce high-purity pharmaceutical intermediates, as it overcomes the limitations of traditional synthetic routes that often suffer from harsh conditions and poor atom economy. The disclosed method utilizes a unique dual-catalytic system involving molybdenum hexacarbonyl and cuprous acetate, enabling the direct cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. For R&D directors and procurement specialists, this innovation offers a pathway to streamline the supply chain for critical drug scaffolds, ensuring consistent quality and reduced operational complexity in the manufacturing of bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that present significant challenges for large-scale industrial application. Traditional routes often involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently require multiple synthetic steps and generate substantial chemical waste. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose serious safety hazards due to the explosive nature of diazo compounds, creating unacceptable risks for commercial scale-up. These legacy methods often struggle with narrow substrate scope, failing to accommodate diverse functional groups without significant yield penalties, which complicates the synthesis of analog libraries required for modern drug discovery. Consequently, the industry has faced a persistent bottleneck in accessing these valuable heterocyclic building blocks efficiently and safely.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach described in the patent utilizes a streamlined one-pot cycloaddition strategy that dramatically simplifies the synthetic workflow. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as readily available starting materials, the reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C, eliminating the need for cryogenic temperatures or extreme pressures. The core of this innovation lies in the synergistic catalytic effect of molybdenum and copper, which facilitates the formation of the triazole ring with high regioselectivity and efficiency. As illustrated in the reaction scheme below, this method not only tolerates a wide array of substituents including halogens, alkyl groups, and electron-withdrawing moieties but also achieves impressive conversion rates that translate directly to higher throughput in a manufacturing setting.
Mechanistic Insights into Mo/Cu Co-catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to optimize the process for specific API intermediates. The reaction mechanism is believed to initiate with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a transient metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Subsequently, the cuprous acetate promoter facilitates a [3+2] cycloaddition between this activated species and the trifluoroethylimidoyl chloride, leading to the formation of a five-membered ring intermediate. This cooperative catalysis is essential, as neither metal alone achieves the same level of efficiency, highlighting the sophisticated design of the catalytic system. The final step involves the elimination of triphenylphosphine oxide, driven by the presence of water in the system or during workup, to yield the stable 3-trifluoromethyl-substituted 1,2,4-triazole product.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions common in radical-based or high-energy pathways. The mild thermal conditions prevent the decomposition of sensitive functional groups, thereby reducing the formation of complex byproduct profiles that are difficult to separate. The use of triethylamine as a base further ensures that acidic byproducts are neutralized effectively, maintaining a clean reaction environment. For quality control laboratories, this translates to a simpler purification profile where the target compound can be isolated with high purity using standard silica gel chromatography, significantly reducing the burden on downstream processing units and ensuring the final material meets stringent pharmaceutical specifications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed to be accessible for both laboratory research and pilot plant operations, requiring standard equipment and commercially available reagents. The procedure involves charging a reaction vessel with the catalytic system and substrates in an aprotic solvent like THF, followed by heating to the specified temperature range. The robustness of the method allows for flexibility in reaction times, typically spanning 18 to 30 hours, providing operators with a wide window to ensure complete conversion without risking product degradation. Detailed standardized operating procedures for this synthesis are outlined below to assist technical teams in replicating these results.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. By shifting away from hazardous diazonium salts and expensive proprietary reagents, manufacturers can significantly reduce raw material costs and mitigate the regulatory burdens associated with handling dangerous chemicals. The reliance on earth-abundant metals like copper and molybdenum, rather than scarce precious metals, further insulates the supply chain from volatile market fluctuations and sourcing bottlenecks. Additionally, the simplified workup procedure reduces solvent consumption and waste generation, aligning with modern green chemistry initiatives and lowering the overall cost of goods sold.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of cheap, commodity-grade starting materials drastically lowers the input cost per kilogram of the final intermediate. Furthermore, the high reaction efficiency minimizes the loss of valuable fluorine-containing precursors, ensuring that every gram of raw material contributes maximally to the final output. This economic efficiency is compounded by the reduced need for complex purification steps, which traditionally consume significant amounts of silica gel and solvents, thereby driving down operational expenditures substantially.
- Enhanced Supply Chain Reliability: Since all key reagents, including trifluoroethylimidoyl chloride and functionalized isonitriles, are commercially available from multiple global suppliers, the risk of single-source dependency is effectively eliminated. This diversity in sourcing options ensures continuous production capability even during regional supply disruptions, providing a strategic buffer for long-term manufacturing contracts. The stability of the reagents also allows for extended storage periods without significant degradation, facilitating better inventory management and just-in-time delivery models for downstream clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of explosive intermediates make this process inherently safer and easier to scale from gram-level experiments to multi-ton commercial production. The reduced generation of hazardous waste simplifies compliance with environmental regulations, avoiding the costly disposal fees associated with heavy metal contaminants or toxic byproducts. This scalability ensures that the technology can grow alongside demand, supporting the rapid expansion of new drug candidates from clinical trials to full-scale commercial launch without requiring process re-engineering.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this technology, we have compiled a set of answers based on the specific data and findings within the patent documentation. These insights are intended to clarify the operational parameters and scope of the reaction for potential partners evaluating this route for their own pipelines. Understanding these nuances is critical for assessing the feasibility of integrating this method into existing manufacturing frameworks.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing a dual catalytic system of Mo(CO)6 and CuOAc with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, nitro, and tert-butyl substituents with yields ranging from moderate to excellent.
Q: Is the purification process complex for industrial scale-up?
A: The post-treatment is straightforward, involving simple filtration followed by standard silica gel column chromatography, making it highly amenable to process optimization and scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Mo/Cu co-catalyzed technology for the next generation of pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global marketplace.
