Advancing Isoquinoline Production: A Green Copper-Catalyzed Cyclization Strategy for Commercial Scale-up
Introduction to Green Isoquinoline Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign methodologies for constructing heterocyclic scaffolds. Patent CN114478378A introduces a transformative approach to synthesizing isoquinoline compounds, a privileged structure found in numerous bioactive alkaloids such as berberine, morphine, and papaverine. This technology leverages a copper-catalyzed cyclization of trans-2-alkynyl aromatic oxime ethers in an aqueous medium, marking a significant departure from traditional methods that rely on hazardous organic solvents and harsh reagents. By utilizing water as the sole reaction medium, this process not only aligns with the principles of green chemistry but also simplifies downstream processing, offering a compelling value proposition for reliable isoquinoline supplier networks aiming to optimize their production pipelines.
The core innovation lies in the unique interaction between the substrate and the solvent. The trans-2-alkynyl aromatic oxime ether contains both nitrogen and oxygen atoms capable of forming hydrogen bonds with water molecules. This interaction unexpectedly enhances the solubility of the organic substrate in the aqueous phase, facilitating efficient contact with the copper catalyst without the need for phase-transfer agents or co-solvents. The reaction proceeds smoothly at temperatures ranging from 80°C to 120°C under air atmosphere, demonstrating remarkable operational simplicity. For R&D directors focused on process safety and sustainability, this water-based protocol represents a paradigm shift in pharmaceutical intermediate manufacturing, reducing the reliance on volatile organic compounds (VOCs) and minimizing the carbon footprint associated with solvent recovery and disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinoline skeleton has relied on classical named reactions such as the Gabriel-Colman rearrangement, the Pomeranz-Fritsch reaction, and the Bischler-Napieralski cyclization. While these methods are well-documented, they suffer from significant drawbacks that hinder their applicability in modern, large-scale commercial scale-up of complex pharmaceutical intermediates. The Bischler-Napieralski reaction, for instance, typically requires strong dehydrating agents like phosphorus oxychloride or polyphosphoric acid, which are highly corrosive and generate substantial amounts of acidic waste. Similarly, the Pomeranz-Fritsch synthesis often necessitates strong acidic conditions and high temperatures, leading to poor functional group tolerance and potential decomposition of sensitive substrates.
Furthermore, many transition-metal catalyzed alternatives reported in recent literature utilize expensive noble metals such as palladium, rhodium, iridium, or silver. These processes frequently depend on anhydrous organic solvents like toluene, tetrahydrofuran, dichloromethane, or dimethylformamide (DMF). The use of such solvents poses serious environmental and safety challenges, including toxicity, flammability, and the high energy cost associated with their distillation and recycling. For procurement managers, the volatility of noble metal prices and the regulatory pressure to limit residual heavy metals in API intermediates add layers of complexity and cost. Consequently, there is an urgent industry demand for a methodology that combines high efficiency with economic and environmental sustainability.
The Novel Approach
The method disclosed in CN114478378A addresses these pain points by employing a base metal catalyst, specifically copper iodide (CuI), in conjunction with water as a green solvent. This novel approach eliminates the need for strong acids, bases, or toxic organic media, thereby drastically simplifying the reaction setup and workup procedures. The use of copper, an earth-abundant and inexpensive metal, significantly lowers the catalyst cost compared to palladium or rhodium systems. Moreover, the reaction exhibits broad substrate scope, accommodating various substituents on the aromatic ring, including halogens, alkyl groups, alkoxy groups, and even heterocycles like thiophene and pyridine.
From a process engineering perspective, the ability to run the reaction in water at moderate temperatures (80-120°C) under air atmosphere is a major advantage. It removes the necessity for inert gas protection (argon or nitrogen) and rigorous drying of reagents, which are standard requirements for many organometallic transformations. This robustness translates directly into reduced operational expenditures (OPEX) and improved safety profiles for plant operators. The high yields reported across numerous examples, often exceeding 80-90%, indicate that this method is not merely a laboratory curiosity but a viable candidate for industrial adoption, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing through simplified unit operations and waste management.
Mechanistic Insights into Copper-Catalyzed Cyclization
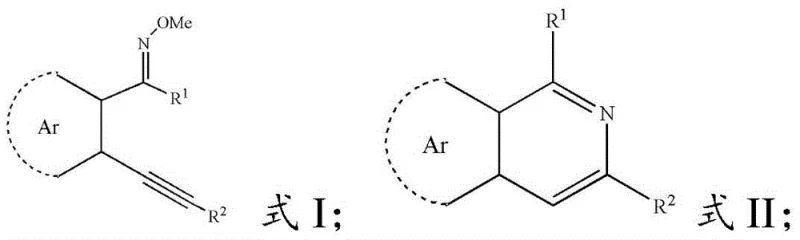
The success of this transformation hinges on the synergistic effect between the copper catalyst and the aqueous medium. Mechanistically, the copper species likely activates the alkyne moiety of the trans-2-alkynyl aromatic oxime ether, rendering it susceptible to nucleophilic attack by the nitrogen atom of the oxime ether group. This intramolecular cyclization forms the dihydroisoquinoline intermediate, which subsequently undergoes oxidation to yield the fully aromatic isoquinoline system. The presence of water plays a dual role: firstly, as discussed, it enhances substrate solubility through hydrogen bonding networks involving the oxime ether oxygen and nitrogen; secondly, it may participate in the proton transfer steps essential for the aromatization process. The use of air as the oxidant suggests that molecular oxygen serves as the terminal oxidant, regenerating the active copper species and completing the catalytic cycle without the need for stoichiometric chemical oxidants.
Impurity control is another critical aspect where this mechanism offers benefits. Traditional acid-mediated cyclizations often lead to polymerization or rearrangement byproducts due to the harsh conditions. In contrast, the mild, neutral-to-slightly-basic conditions of the copper-water system minimize side reactions. The high chemoselectivity observed, even in the presence of sensitive functional groups like bromides or trifluoromethyl groups, underscores the gentle nature of this catalytic system. For quality control teams, this means a cleaner crude reaction profile, which simplifies purification and ensures that the final high-purity isoquinoline products meet stringent regulatory specifications with minimal effort. The detailed understanding of this mechanism allows process chemists to fine-tune parameters such as catalyst loading (1-10 mol%) and temperature to optimize the balance between reaction rate and selectivity.

How to Synthesize Isoquinoline Compounds Efficiently
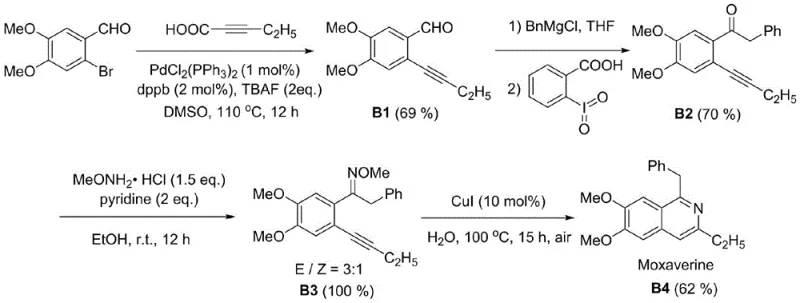
The synthetic route described in the patent provides a straightforward two-stage process: first, the preparation of the key trans-2-alkynyl aromatic oxime ether intermediate, and second, the copper-catalyzed cyclization. The precursor synthesis involves a Sonogashira coupling between an ortho-halo ketone and a terminal alkyne, followed by condensation with methoxylamine hydrochloride. This modular approach allows for the rapid assembly of diverse libraries of isoquinoline derivatives by varying the alkyne or the aromatic ketone component. The subsequent cyclization step is operationally trivial, requiring only the mixing of the substrate, copper iodide, and water in a sealed vessel followed by heating. Detailed standardized synthesis steps for specific derivatives can be found in the guide below, ensuring reproducibility and consistency across different batches.
- Prepare the trans-2-alkynyl aromatic oxime ether substrate by coupling an ortho-halo ketone with a terminal alkyne, followed by oximation with methoxylamine hydrochloride.
- Mix the substrate with a copper-based catalyst (such as CuI, 1-10 mol%) and water in a sealed reaction vessel.
- Heat the mixture to 80-120°C for 15-24 hours under air atmosphere to facilitate the cyclization reaction, then extract and purify the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere technical feasibility. The shift from organic solvents to water fundamentally alters the cost structure of the manufacturing process. Organic solvents represent a significant portion of the material cost in fine chemical synthesis, not just in terms of purchase price but also in terms of storage, handling, and disposal fees. By replacing solvents like DMF, DCM, or toluene with water, the process eliminates these costs entirely. Furthermore, the removal of volatile organic compounds reduces the risk of fire and explosion, potentially lowering insurance premiums and safety compliance costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts (Pd, Rh, Ir) with inexpensive copper iodide results in a drastic reduction in catalyst expenditure. Copper is orders of magnitude cheaper than palladium, and its lower toxicity profile simplifies the removal of residual metals from the final product, a critical requirement for pharmaceutical applications. Additionally, the elimination of organic solvents removes the need for complex solvent recovery systems and reduces the volume of hazardous waste generated, leading to substantial savings in waste treatment and environmental compliance. The high atom economy of the cyclization reaction further contributes to cost efficiency by maximizing the conversion of raw materials into the desired product.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as ortho-halo acetophenones and terminal alkynes, are commodity chemicals available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply chain disruptions caused by single-source dependencies. The robustness of the reaction conditions—tolerating air and moisture—means that the process is less sensitive to variations in raw material quality or environmental conditions during transport and storage. This reliability ensures consistent production schedules and shorter lead times, enabling supply chain managers to maintain optimal inventory levels and respond quickly to market demands for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling up reactions that use water as a solvent is inherently safer and easier than scaling up those using flammable or toxic organic solvents. Heat transfer is more efficient in aqueous media, reducing the risk of thermal runaway incidents. From an environmental standpoint, the process generates minimal hazardous waste, aligning with increasingly strict global regulations on industrial emissions and effluent discharge. This 'green' credential is not just a regulatory checkbox but a market differentiator, appealing to end-users who prioritize sustainability in their supply chains. The ease of scale-up from gram to kilogram and potentially ton scales ensures that the technology can support commercial production volumes without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed isoquinoline synthesis. These answers are derived directly from the experimental data and scope defined in patent CN114478378A, providing clarity on substrate compatibility, reaction conditions, and product isolation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows.
Q: What are the advantages of using water as a solvent in this isoquinoline synthesis?
A: Using water eliminates the need for toxic organic solvents like DMF or dichloromethane, significantly reducing environmental impact and waste disposal costs while leveraging hydrogen bonding to enhance substrate solubility.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent tolerance for halogens (F, Cl, Br), electron-donating groups (methoxy, methyl), and electron-withdrawing groups (trifluoromethyl), making it suitable for diverse pharmaceutical scaffolds.
Q: Is the copper catalyst expensive compared to traditional noble metals?
A: Copper iodide is substantially more cost-effective than palladium, rhodium, or iridium catalysts often used in similar cyclizations, leading to significant raw material cost reductions for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Supplier
The technological advancements detailed in CN114478378A underscore the potential for more sustainable and cost-effective production of isoquinoline derivatives. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of isoquinoline intermediate meets the highest standards required by the global pharmaceutical industry. We understand the critical importance of supply continuity and cost efficiency in today's competitive market.
We invite you to collaborate with us to leverage this green synthesis technology for your specific project needs. Whether you require custom synthesis of novel isoquinoline analogs or large-scale supply of established intermediates like Moxaverine precursors, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive value and efficiency in your supply chain.