Advanced Rhodium-Catalyzed Synthesis of Trifluoromethyl Enamines for Pharmaceutical Applications
Advanced Rhodium-Catalyzed Synthesis of Trifluoromethyl Enamines for Pharmaceutical Applications
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and versatile methodologies to construct complex molecular architectures. A significant breakthrough in this domain is documented in Chinese Patent CN115925692A, which discloses a novel preparation method for trifluoromethyl-substituted enamine compounds. This technology leverages a sophisticated rhodium-catalyzed carbon-hydrogen (C-H) activation strategy coupled with a unique directing group migration tandem reaction. For R&D directors and procurement specialists in the fine chemical industry, this patent represents a pivotal shift away from laborious multi-step syntheses towards a more streamlined, atom-economical approach. The ability to directly install both an indole skeleton and a trifluoromethyl group—a pharmacophore known for enhancing metabolic stability and lipophilicity—into a single enamine scaffold offers immense value for drug discovery programs targeting various therapeutic areas.
The core innovation lies in the utilization of readily available indole derivatives and trifluoroacetimide sulfur ylides as starting materials. Unlike conventional methods that often require pre-functionalized substrates or harsh reaction conditions, this protocol operates under relatively mild thermal conditions using a robust catalytic system. The reaction employs a dichlorocyclopentyl rhodium(III) dimer as the catalyst, supported by silver hexafluoroantimonate as a promoter and a buffer system of sodium acetate and acetic acid. This specific combination facilitates the cleavage of inert C-H bonds and the subsequent formation of carbon-carbon bonds with high regioselectivity. For a reliable pharmaceutical intermediate supplier, mastering such transformations is critical to maintaining a competitive edge in the global market, ensuring that clients receive high-purity building blocks essential for the development of next-generation bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of enamine functional groups onto indole scaffolds has been a challenging endeavor in organic synthesis. Traditional literature reports predominantly rely on indole substrates that are pre-equipped with specific directing groups to facilitate transition metal-catalyzed C-H activation. While effective in academic settings, these methods often suffer from significant limitations when translated to industrial scales. The requirement for specialized, often expensive, directing groups adds extra synthetic steps, thereby increasing the overall cost of goods and reducing the overall yield due to cumulative losses at each stage. Furthermore, many existing protocols utilize ketene imines as substrates, which can be unstable or difficult to handle, limiting the structural diversity of the final enamine products. The reliance on manganese or other less abundant metals in some prior art can also introduce supply chain vulnerabilities and complicate downstream purification processes due to heavy metal residue concerns. Consequently, the application scope of these conventional enamines remains restricted, hindering the rapid exploration of chemical space required for modern drug discovery.
The Novel Approach
In stark contrast, the methodology outlined in CN115925692A offers a transformative solution by employing trifluoroacetimide sulfur ylides as versatile synthetic building blocks. These ylides serve a dual purpose: they act as precursors for imine carbenes and provide the essential trifluoromethyl moiety in a single operation. The reaction proceeds through a tandem sequence involving rhodium-catalyzed pyrimidine nitrogen-directed C-H activation, followed by isomerization and a remarkable intramolecular 1,5-migration of the pyrimidine group. This cascade mechanism not only constructs the desired carbon-carbon bond efficiently but also regenerates the indole nitrogen functionality without the need for additional deprotection steps. The result is a highly efficient process that delivers multifunctionalized enamine compounds with yields frequently exceeding 80%. By utilizing cheap and commercially available starting materials, this approach drastically simplifies the synthetic route, making it an ideal candidate for cost reduction in API manufacturing and accelerating the timeline from benchtop discovery to pilot plant production.
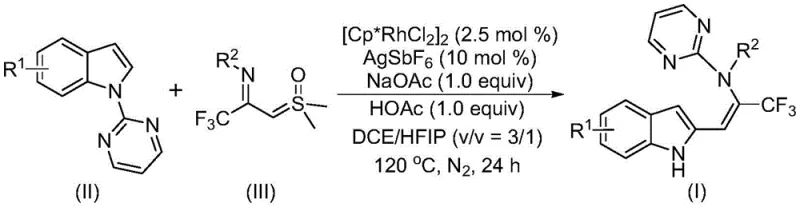
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Migration
To fully appreciate the technical sophistication of this process, one must delve into the proposed mechanistic pathway which underpins its high efficiency and selectivity. The catalytic cycle initiates with the coordination of the active rhodium(III) species to the pyrimidine nitrogen atom present on the indole substrate (Compound II). This coordination directs the metal center to the proximal C-H bond, facilitating a concerted metalation-deprotonation (CMD) process assisted by the acetate additive. This step generates a stable five-membered rhodacycle intermediate, effectively activating the inert C-H bond for subsequent functionalization. The trifluoroacetimide sulfur ylide (Compound III) then enters the cycle, reacting with the rhodacycle to form a new carbon-carbon bond. This step is crucial as it installs the trifluoromethyl-containing side chain directly onto the indole core. Following the C-C bond formation, the intermediate undergoes isomerization to generate an initial enamine species. However, the true elegance of this chemistry lies in the subsequent migration step.
The pyrimidine group, which initially served as the directing group for C-H activation, undergoes an intramolecular 1,5-migration to the nitrogen atom of the newly formed enamine chain. This migration restores the aromaticity of the indole system and releases the final trifluoromethyl-substituted enamine product (Compound I), while regenerating the active rhodium catalyst to continue the cycle. This mechanism explains the high functional group tolerance observed across a wide range of substrates, including those with electron-donating groups like methoxy and methyl, as well as electron-withdrawing halogens like fluorine, chlorine, and bromine. The robustness of this catalytic cycle ensures consistent impurity profiles, a critical factor for R&D teams focused on purity specifications. By understanding these mechanistic nuances, process chemists can better optimize reaction parameters such as temperature and solvent ratios to maximize throughput and minimize waste, ensuring a sustainable and economically viable production process for high-purity pharmaceutical intermediates.
How to Synthesize Trifluoromethyl Substituted Enamine Efficiently
Implementing this advanced synthetic route requires precise control over reaction conditions to achieve the reported high yields and purity levels. The protocol specifies a molar ratio of indole compound to sulfur ylide of approximately 1:2, ensuring that the ylide is in excess to drive the reaction to completion. The catalyst loading is kept low at 2.5 mol% for the rhodium dimer and 10 mol% for the silver salt, demonstrating the high turnover number of the catalytic system. The choice of solvent is also critical; a mixture of 1,2-dichloroethane (DCE) and hexafluoroisopropanol (HFIP) in a 3:1 volume ratio is preferred to solubilize all reagents effectively and promote the reaction kinetics. The detailed standardized synthesis steps below outline the exact procedure for replicating this transformation in a laboratory or pilot plant setting.
- Combine catalyst [Cp*RhCl2]2, promoter AgSbF6, additives NaOAc/HOAc, indole compound (II), and trifluoroacetimide sulfur ylide (III) in a Schlenk tube.
- Add organic solvent mixture of DCE and HFIP (3: 1 v/v) to ensure complete dissolution of reactants.
- Heat the reaction mixture to 120°C under nitrogen atmosphere for 24 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the significant simplification of the supply chain for raw materials. Since the method utilizes inexpensive and widely available indoles and aromatic amines (precursors for the ylide), there is no dependency on exotic or custom-synthesized starting materials that often carry long lead times and high price volatility. This accessibility ensures a stable supply base, reducing the risk of production delays caused by raw material shortages. Furthermore, the elimination of multiple synthetic steps required in traditional routes means fewer unit operations, less solvent consumption, and reduced energy usage per kilogram of product. This streamlined process inherently lowers the manufacturing cost base, allowing for more competitive pricing structures in a crowded marketplace without sacrificing margin.
- Cost Reduction in Manufacturing: The economic impact of this methodology is profound due to the drastic reduction in step count and the use of earth-abundant catalytic systems relative to precious metals like palladium in some alternative couplings. By avoiding the need for pre-functionalized directing groups that must be installed and later removed, the process saves substantial amounts of reagents and labor hours. The high reaction efficiency, with yields consistently reaching up to 82% for various substrates, minimizes the loss of valuable intermediates, thereby maximizing the return on investment for every batch produced. Additionally, the simplified post-treatment process, which involves basic filtration and standard column chromatography, reduces the complexity of purification, leading to lower operational expenditures and faster turnaround times for order fulfillment.
- Enhanced Supply Chain Reliability: Stability in the supply chain is paramount for pharmaceutical manufacturers who cannot afford interruptions in their API production lines. This synthesis route enhances reliability by relying on commodity chemicals that are sourced from a broad network of global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, tolerating a wide array of functional groups, means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with different batches of starting materials. This resilience allows for better inventory planning and forecasting, enabling supply chain teams to maintain optimal stock levels of critical intermediates like trifluoromethyl enamines without the need for excessive safety stocks.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, the scalability of this process offers a distinct advantage. The patent explicitly notes the successful expansion to gram-level reactions, indicating a clear path towards kilogram and ton-scale production. The use of a defined solvent system and the potential for catalyst recycling further align with environmental compliance standards by reducing hazardous waste generation. The ability to produce complex heterocycles with high atom economy supports sustainability goals, making this technology attractive for companies aiming to reduce their carbon footprint. Moreover, the straightforward workup procedure facilitates easier waste management and solvent recovery, contributing to a cleaner and more compliant manufacturing environment that meets rigorous international regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this synthesis route for their specific applications. Understanding these details is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional enamine synthesis?
A: This method utilizes a directing group migration strategy that allows for the direct functionalization of inexpensive indole substrates without pre-activation, significantly simplifying the synthetic route and improving atom economy compared to traditional cross-coupling methods.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates that the reaction can be expanded to gram-level scales with high efficiency and yields often exceeding 80%, indicating strong potential for commercial scale-up in API manufacturing.
Q: What is the substrate scope regarding the indole and ylide components?
A: The methodology exhibits broad substrate tolerance, accommodating various substituents on the indole ring (such as halogens, alkyl, and alkoxy groups) and diverse aryl groups on the sulfur ylide, enabling the synthesis of a wide library of bioactive analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Enamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN115925692A for the development of advanced pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this rhodium-catalyzed route are fully realized in a practical, industrial setting. We are committed to delivering high-purity trifluoromethyl enamine compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic synthesis allows us to offer customized solutions that align perfectly with your drug development timelines and quality requirements.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs, helping you optimize your budget without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary molecules. Let us be your trusted partner in navigating the complexities of modern chemical synthesis and securing a reliable supply of critical building blocks for the future of medicine.
