Advanced Synthesis of Trifluoromethyl Enamine Intermediates: Bridging Innovation and Commercial Scale-Up for Pharmaceutical Manufacturing
The patent CN115925692A discloses a novel rhodium-catalyzed methodology for synthesizing trifluoromethyl-substituted enamine compounds, representing a significant advancement in the production of complex pharmaceutical intermediates. This innovative approach utilizes dichlorocyclopentyl rhodium(III) dimer as catalyst to facilitate carbon-hydrogen activation and subsequent molecular rearrangement, enabling the efficient construction of valuable enamine scaffolds containing both indole and trifluoromethyl moieties that are critical for developing bioactive molecules and drug candidates.
Advanced Reaction Mechanism and Purity Control
The synthetic pathway described in CN115925692A begins with rhodium-catalyzed pyrimidine nitrogen-directed carbon-hydrogen activation, where the catalyst coordinates with the indole nitrogen to facilitate selective C-H bond cleavage at the C2 position. This activated intermediate then reacts with trifluoroacetimide sulfur ylide to form a new carbon-carbon bond, followed by isomerization to generate an enamine intermediate. The process continues with an intramolecular pyrimidine group migration through a 1,5-migration mechanism, ultimately yielding the target trifluoromethyl-substituted enamine product with high regioselectivity. The reaction mechanism is illustrated in 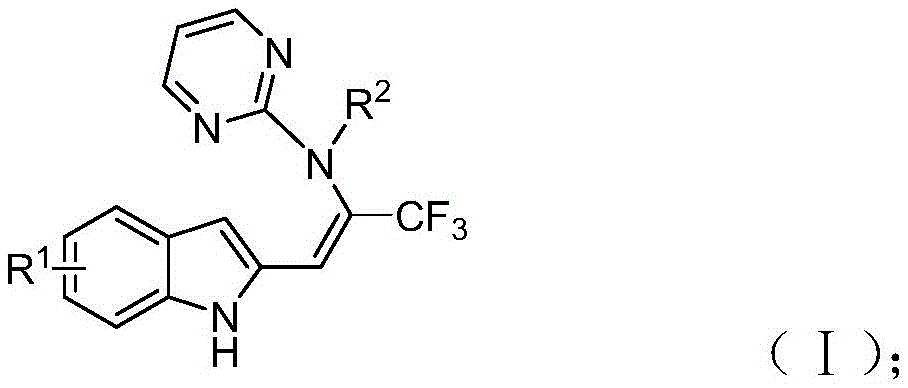 which shows the structural transformation from starting materials to final product.
which shows the structural transformation from starting materials to final product.
Purity control is achieved through careful optimization of reaction parameters and post-treatment procedures. The patent specifies that the reaction should be conducted at 110-130°C for 18-30 hours to ensure complete conversion while minimizing side reactions. The use of specific solvent systems, particularly dichloroethane and hexafluoroisopropanol mixtures, promotes high conversion rates while suppressing unwanted byproducts. The post-treatment process involves filtration, silica gel mixing, and column chromatography purification, which effectively removes catalyst residues and unreacted starting materials. The NMR data provided in the patent (as shown in  and
and  ) confirms the high purity of the final products, with characteristic peaks corresponding to the expected molecular structures and absence of significant impurities.
) confirms the high purity of the final products, with characteristic peaks corresponding to the expected molecular structures and absence of significant impurities.
Overcoming Traditional Limitations in Enamine Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing multifunctionalized enamines, particularly those containing both indole and trifluoromethyl groups, have been constrained by several significant limitations. Previous methods primarily relied on transition metal-catalyzed directed C-H activation using substrates with pre-installed directing groups, which restricted structural diversity and required additional synthetic steps to install and remove these directing groups. The limited substrate scope of conventional methods resulted in narrow application ranges for the synthesized enamine compounds, hindering their broader utility in pharmaceutical development. Furthermore, many existing protocols suffered from harsh reaction conditions, low yields, and poor functional group tolerance, making them unsuitable for large-scale production of complex intermediates required in drug manufacturing.
The Novel Approach
The methodology disclosed in CN115925692A overcomes these limitations through an innovative rhodium-catalyzed tandem reaction that combines C-H activation with directed group migration. By utilizing readily available indole compounds 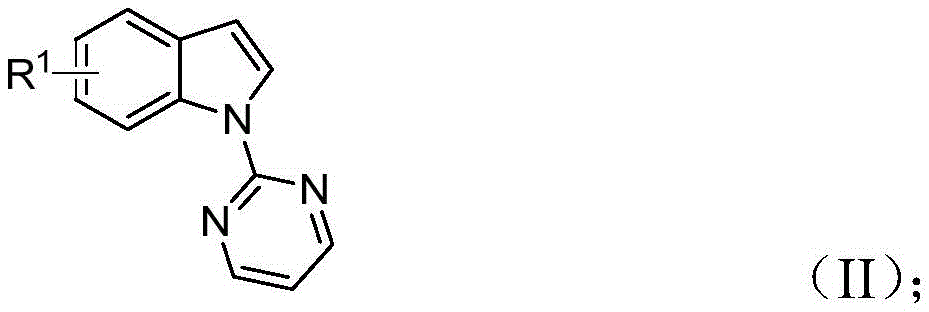 and trifluoroacetimide sulfur ylides
and trifluoroacetimide sulfur ylides  as starting materials, this approach eliminates the need for pre-functionalized substrates while maintaining high reaction efficiency. The patent demonstrates that multiple products achieve yields above 80%, with excellent functional group tolerance across a wide range of substituents including various alkyl, alkoxy, halogen, nitro, and trifluoromethyl groups. The optimized reaction conditions (110-130°C for 18-30 hours) in mixed solvent systems enable gram-scale reactions without significant loss of efficiency, addressing scalability concerns that have plagued previous methods. This breakthrough provides pharmaceutical researchers with access to diverse enamine scaffolds that can be readily incorporated into bioactive molecule development pipelines.
as starting materials, this approach eliminates the need for pre-functionalized substrates while maintaining high reaction efficiency. The patent demonstrates that multiple products achieve yields above 80%, with excellent functional group tolerance across a wide range of substituents including various alkyl, alkoxy, halogen, nitro, and trifluoromethyl groups. The optimized reaction conditions (110-130°C for 18-30 hours) in mixed solvent systems enable gram-scale reactions without significant loss of efficiency, addressing scalability concerns that have plagued previous methods. This breakthrough provides pharmaceutical researchers with access to diverse enamine scaffolds that can be readily incorporated into bioactive molecule development pipelines.
Commercial Advantages for Pharmaceutical Supply Chains
This advanced synthetic methodology addresses critical pain points in pharmaceutical intermediate supply chains by offering a streamlined production process that enhances both economic efficiency and supply reliability. The elimination of multiple synthetic steps required in conventional approaches directly translates to reduced manufacturing complexity and improved process economics for pharmaceutical manufacturers seeking high-purity intermediates.
- Cost Reduction in Chemical Manufacturing: The use of readily available starting materials, including inexpensive aromatic amines and trifluoroacetic acid derivatives, significantly lowers raw material costs compared to traditional methods requiring specialized precursors. The optimized catalyst system (dichlorocyclopentyl rhodium(III) dimer with silver hexafluoroantimonate promoter) operates at low loading (0.025:0.1 molar ratio), reducing catalyst expenses while maintaining high efficiency. The simplified reaction sequence eliminates multiple protection/deprotection steps common in conventional enamine synthesis, resulting in fewer unit operations and lower overall production costs. This cost-effective approach enables pharmaceutical manufacturers to achieve substantial savings in intermediate production without compromising quality or purity requirements.
- Reducing Lead Time for High-Purity Intermediates: The streamlined one-pot reaction process reduces manufacturing cycle time by eliminating intermediate isolation steps required in traditional multi-step syntheses. The optimized reaction duration (18-30 hours) at moderate temperatures (110-130°C) allows for faster batch turnaround compared to more demanding conventional processes that often require extended reaction times or cryogenic conditions. The straightforward post-treatment procedure (filtration followed by column chromatography) minimizes processing time while ensuring high purity levels required for pharmaceutical applications. This accelerated production timeline enables pharmaceutical companies to respond more rapidly to changing development needs and market demands for novel therapeutic compounds.
- Enhanced Supply Chain Resilience: The broad substrate scope and high functional group tolerance demonstrated in the patent provide flexibility to adapt the process to various structural requirements without significant re-optimization. The ability to scale the reaction to gram-level quantities (as documented in the patent examples) establishes a clear pathway for commercial scale-up to meet increasing demand from pharmaceutical clients. The use of common laboratory equipment and standard processing techniques ensures compatibility with existing manufacturing infrastructure, reducing implementation barriers for contract manufacturers. This robust process design supports reliable supply continuity even during periods of market volatility or raw material shortages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN115925692A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
