Revolutionizing Indole Synthesis: Advanced Ferrous Complex Catalysis for Commercial Scale
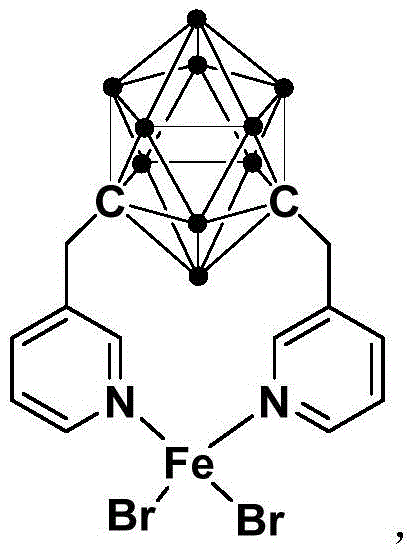
The chemical industry is currently witnessing a paradigm shift towards sustainable and cost-effective catalytic systems, particularly in the synthesis of high-value heterocyclic compounds. Patent CN113185444A introduces a groundbreaking method for synthesizing indole derivatives utilizing a novel ferrous complex catalyst. This technology represents a significant departure from traditional methodologies that rely on expensive precious metals or harsh reaction conditions. By employing a ferrous complex containing a meta-carborane methyl pyridine structure, this innovation enables the efficient coupling of arylamine and acetaldehyde at room temperature. For R&D Directors and Procurement Managers seeking a reliable indole derivative supplier, this patent data underscores a pathway to substantial cost reduction in pharmaceutical intermediates manufacturing. The ability to achieve high yields under ambient conditions not only enhances process safety but also drastically simplifies the engineering requirements for commercial production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole derivatives has been plagued by significant technical and economic hurdles that impede efficient commercial scale-up of complex heterocyclic compounds. Prior art often necessitates the use of costly transition metal catalysts such as copper, cobalt, or ruthenium, which introduce heavy metal contamination risks and require extensive downstream purification processes. Furthermore, many established protocols demand rigorous reaction conditions, including high temperatures and the use of hazardous solvents like chlorobenzene, which complicate waste management and increase operational expenditures. The reliance on expensive alkyne raw materials in some conventional routes further exacerbates the cost burden, making the final API intermediates less competitive in the global market. These factors collectively create bottlenecks in supply chain reliability and extend the lead time for high-purity indole derivatives, posing challenges for manufacturers aiming to optimize their production pipelines.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data leverages an earth-abundant iron-based catalyst to overcome these longstanding deficiencies. By utilizing a specifically designed ferrous complex with a meta-carborane methyl pyridine ligand, the reaction proceeds efficiently at room temperature in toluene, a common and manageable organic solvent. This method eliminates the need for expensive alkynes, instead using readily available acetaldehyde as a key building block, which significantly lowers raw material costs. The catalytic system demonstrates remarkable versatility, accommodating a wide range of arylamine substrates with varying electronic properties without compromising yield or selectivity. This technological leap facilitates cost reduction in pharmaceutical intermediates manufacturing by streamlining the synthetic route and minimizing energy consumption, thereby offering a robust solution for modern chemical production needs.
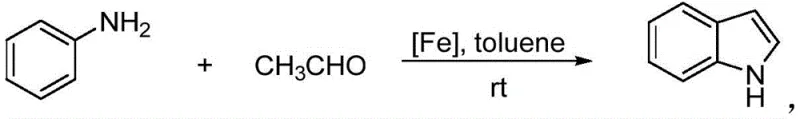
Mechanistic Insights into Ferrous Complex-Catalyzed Cyclization
The core of this technological advancement lies in the unique structural properties of the ferrous complex catalyst, which features a meta-carborane methyl pyridine framework. This specific ligand architecture provides exceptional stability to the iron center, preventing premature decomposition and ensuring sustained catalytic activity throughout the reaction cycle. The carborane cage acts as an electron reservoir, modulating the electronic density at the metal center to facilitate the activation of arylamine and acetaldehyde under mild thermal conditions. Such mechanistic precision allows for the formation of the indole skeleton with high regioselectivity, minimizing the generation of unwanted by-products and simplifying the purification workflow. For technical teams, understanding this mechanism is crucial for optimizing reaction parameters and ensuring consistent batch-to-batch quality in large-scale operations.
Furthermore, the impurity profile of the resulting indole derivatives is significantly improved due to the high specificity of the ferrous catalyst. Traditional methods often suffer from side reactions such as polymerization or over-oxidation, which generate difficult-to-remove impurities that can compromise the safety and efficacy of the final pharmaceutical product. The mild nature of this iron-catalyzed process suppresses these deleterious pathways, leading to cleaner reaction mixtures and higher crude purity. This inherent advantage reduces the burden on QC labs and shortens the overall production cycle time. By controlling the electronic and steric environment around the iron atom, the catalyst ensures that the cyclization proceeds smoothly even with sterically hindered substrates, demonstrating broad applicability across diverse chemical spaces required for drug discovery and development.
How to Synthesize Indole Derivatives Efficiently
The synthesis of high-purity indole derivatives via this ferrous complex catalysis involves a streamlined procedure that is amenable to both laboratory optimization and industrial manufacturing. The process begins with the preparation of the catalyst itself, followed by the direct coupling of arylamine and acetaldehyde in a toluene medium. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are critical for maximizing yield and ensuring operator safety. The following section outlines the standardized synthesis steps derived from the patent data, providing a clear roadmap for technical implementation. Please refer to the specific injection point below for the comprehensive step-by-step guide tailored for process engineers.
- Preparation of the ferrous complex catalyst involving m-carborane, n-BuLi, chloromethylpyridine, and FeBr2.
- Mixing arylamine and acetaldehyde in toluene solvent with the prepared ferrous complex catalyst.
- Reacting the mixture at room temperature for 60-240 minutes followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this ferrous complex catalysis technology offers transformative benefits for supply chain resilience and cost management. The shift from precious metal catalysts to iron-based systems fundamentally alters the cost structure of indole derivative production, removing dependency on volatile commodity markets for metals like ruthenium or palladium. Additionally, the operation at room temperature significantly reduces energy consumption associated with heating and cooling cycles, contributing to lower utility costs and a smaller carbon footprint. These factors combine to create a more predictable and stable supply chain, reducing the risk of production delays caused by equipment failure or raw material scarcity. For Supply Chain Heads, this translates to enhanced supply chain reliability and the ability to meet tight delivery schedules with greater confidence.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts and the use of inexpensive acetaldehyde as a feedstock drive down the direct material costs substantially. Moreover, the simplified purification process resulting from high selectivity reduces solvent usage and waste disposal expenses, leading to significant overall cost savings. The robustness of the catalyst also means lower catalyst loading is required to achieve high conversion, further optimizing the economic efficiency of the manufacturing process without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing earth-abundant iron and common solvents like toluene mitigates the risk of supply disruptions often associated with specialized reagents. The mild reaction conditions reduce the stress on manufacturing equipment, extending asset life and minimizing unplanned maintenance downtime. This operational stability ensures a continuous flow of high-purity indole derivatives, allowing procurement teams to secure long-term contracts with greater assurance of fulfillment and consistency in product specifications.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of extreme temperatures or pressures, making it easier to transfer from pilot plants to full commercial production. The reduced generation of hazardous waste and the avoidance of toxic solvents align with stringent environmental regulations, facilitating smoother regulatory approvals. This green chemistry approach not only enhances corporate sustainability profiles but also future-proofs the manufacturing process against tightening environmental legislation globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrous complex catalysis technology. These insights are derived directly from the patent data to provide accurate and actionable information for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines. Please see the dynamic content injection below for specific answers tailored to your operational concerns.
Q: What are the primary advantages of using ferrous complexes over traditional catalysts for indole synthesis?
A: The use of ferrous complexes offers significant advantages including mild reaction conditions at room temperature, elimination of expensive precious metals like ruthenium or copper, and higher catalytic activity with yields ranging from 85% to 96%.
Q: Is this catalytic method suitable for large-scale commercial production?
A: Yes, the method is highly suitable for scale-up due to the stability of the ferrous complex, the use of common solvents like toluene, and the avoidance of harsh high-temperature or high-pressure conditions.
Q: What is the substrate scope for this indole synthesis method?
A: The method demonstrates a wide substrate range, effectively catalyzing reactions with various arylamines including those with electron-donating or electron-withdrawing groups such as methyl, bromo, methoxy, and nitro substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative catalytic technologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this ferrous complex method are successfully translated into robust industrial processes. We are committed to delivering high-purity indole derivatives that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. Our infrastructure is designed to handle complex synthetic routes with precision, guaranteeing the consistency and quality required by top-tier pharmaceutical and agrochemical companies worldwide.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this iron-catalyzed route for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate how we can support your goals for cost efficiency and supply security. Let us help you optimize your manufacturing strategy with our proven expertise in catalytic process development and commercial scale-up.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
