Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
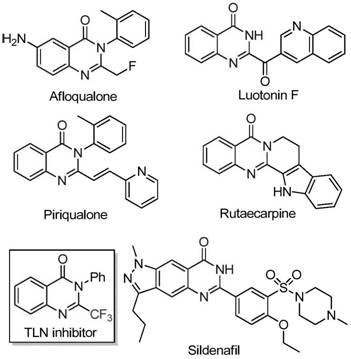
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing fluorinated motifs that enhance biological efficacy. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, utilizing an inexpensive iron-catalyzed cyclization strategy. This technology represents a significant leap forward for manufacturers of pharmaceutical intermediates, addressing critical pain points related to catalyst cost and substrate availability. Quinazolinone scaffolds are ubiquitous in medicinal chemistry, serving as the core structure for numerous bioactive molecules with anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group further amplifies these properties by improving metabolic stability, lipophilicity, and bioavailability, making the efficient synthesis of these derivatives a high-priority objective for R&D teams globally.
The significance of this patent lies not only in the chemical transformation but in its potential to streamline the supply chain for complex heterocyclic building blocks. By shifting away from precious metal catalysis and expensive fluorinating reagents, this process aligns perfectly with the industry's drive towards sustainable and economically viable manufacturing. For procurement managers and supply chain directors, understanding the nuances of this synthetic route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the volatility associated with scarce raw materials. The following analysis dissects the technical merits and commercial implications of this novel methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with trifluoroacetic anhydride or ethyl trifluoroacetate. While these methods are chemically feasible, they suffer from severe practical limitations that hinder their application in large-scale cost reduction in pharmaceutical intermediates manufacturing. The reaction conditions are often harsh, requiring strong acids or bases and elevated temperatures that can degrade sensitive functional groups. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, can be prohibitively expensive and difficult to source in bulk quantities. Yields are frequently inconsistent, and the substrate scope is often narrow, limiting the ability to generate diverse libraries of analogs for drug discovery programs. These factors collectively contribute to high production costs and extended lead times, creating bottlenecks in the development of new therapeutic agents.
The Novel Approach
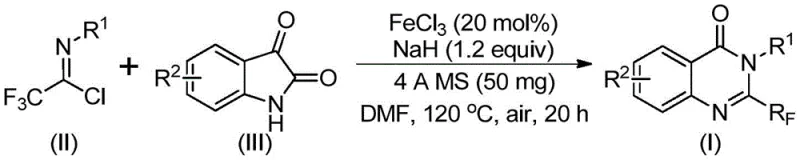
In stark contrast, the method disclosed in CN111675662B utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials, catalyzed by cheap and abundant ferric chloride. This novel approach fundamentally alters the economic landscape of producing these valuable heterocycles. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This pathway avoids the need for pre-functionalized, expensive anthranilic acid derivatives. The operational simplicity is remarkable; the reaction can be conducted in common aprotic solvents like DMF, with a straightforward two-stage heating profile (40°C followed by 120°C). This robustness allows for the synthesis of a wide array of substituted quinazolinones, including those with electron-donating and electron-withdrawing groups, thereby offering unparalleled flexibility for medicinal chemists.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic elegance of this transformation is a key driver of its efficiency and selectivity. The process initiates with the deprotonation of the isatin nitrogen by sodium hydride, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a crucial trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst facilitates a decarbonylation event, effectively removing the carbonyl oxygen from the isatin moiety while promoting the cyclization to form the quinazolinone ring system. This iron-catalyzed step is critical; it operates under milder conditions than traditional thermal cyclizations, minimizing side reactions such as polymerization or decomposition of the trifluoromethyl group. The presence of 4A molecular sieves in the reaction mixture plays a vital role in scavenging moisture, which could otherwise hydrolyze the sensitive imidoyl chloride or deactivate the sodium hydride, ensuring high conversion rates and consistent product quality.
From an impurity control perspective, this mechanism offers distinct advantages. The specificity of the iron catalyst for the decarbonylation-cyclization sequence reduces the formation of regioisomers or over-reacted byproducts that are common in non-catalytic thermal methods. The tolerance for various substituents on the aromatic rings (R1 and R2) suggests that the electronic nature of the substrate does not drastically impede the catalytic cycle. Whether the substrate bears a strongly electron-withdrawing nitro group or an electron-donating methoxy group, the reaction maintains high efficiency. This broad functional group tolerance is essential for high-purity pharmaceutical intermediates, as it simplifies downstream purification. The ability to achieve high yields (often exceeding 80-90% for optimized substrates) with minimal byproduct formation translates directly to reduced solvent usage and waste generation during the isolation phase, a critical metric for green chemistry compliance.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The protocol involves mixing ferric chloride (20 mol%), sodium hydride (1.2 equivalents), 4A molecular sieves, trifluoroethylimidoyl chloride, and the isatin derivative in DMF. The mixture is initially stirred at 40°C for 8-10 hours to allow the initial coupling to occur without triggering premature decomposition. Following this induction period, the temperature is ramped to 120°C for 18-20 hours to drive the cyclization to completion. Post-reaction processing is equally streamlined, involving simple filtration to remove solids, followed by silica gel treatment and column chromatography. This straightforward workflow minimizes unit operations, reducing both labor costs and equipment downtime. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, refer to the standardized synthesis steps provided below.
- Combine ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an organic solvent such as DMF.
- Stir the reaction mixture at 40°C for 8-10 hours to initiate the alkali-promoted bond formation.
- Heat the mixture to 120°C and react for 18-20 hours to complete the iron-catalyzed decarbonylation and cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost stability and supply security. The shift from precious metal catalysts to commodity chemicals like ferric chloride eliminates the price volatility associated with rare earth metals. Additionally, the starting materials—isatin and aromatic amines used to make the imidoyl chloride—are bulk commodities with established global supply chains, ensuring consistent availability even during market fluctuations. This resilience is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by multinational pharmaceutical clients. The simplified workup procedure further enhances operational efficiency, reducing the consumption of auxiliary materials and shortening the overall batch cycle time.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts and specialized fluorinating reagents with inexpensive iron salts and readily available chlorides results in a drastic reduction in raw material costs. The elimination of complex purification steps required to remove heavy metal residues further lowers the cost of goods sold (COGS). By utilizing a catalytic system that operates efficiently at moderate loadings, the process minimizes the quantity of catalyst required per kilogram of product, driving down the overall chemical cost significantly.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key reagents are not subject to the geopolitical or logistical constraints often faced by specialty fluorine reagents. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, reducing the risk of batch failures. This reliability ensures a steady flow of commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to commit to long-term supply agreements with confidence.
- Scalability and Environmental Compliance: The use of DMF as a solvent, while requiring careful handling, is well-understood in industrial settings with established recovery protocols. The absence of toxic heavy metals simplifies wastewater treatment and solid waste disposal, aligning with increasingly stringent environmental regulations. The reaction's ability to proceed under air (as noted in some embodiments) or with simple inert gas protection reduces the engineering complexity of the reactor setup, facilitating easier reducing lead time for high-purity pharmaceutical intermediates during technology transfer from lab to plant.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers a substantial cost reduction compared to precious metals like palladium or rhodium. Furthermore, iron is environmentally benign, simplifying waste disposal and regulatory compliance for large-scale manufacturing.
Q: Can this synthesis method tolerate diverse functional groups on the aromatic ring?
A: Yes, the patent demonstrates excellent functional group tolerance. Substituents such as methyl, halogen (F, Cl, Br, I), methoxy, and nitro groups at ortho-, meta-, or para-positions are well-tolerated, yielding products with high purity.
Q: Is the reaction scalable for industrial production of pharmaceutical intermediates?
A: The method is designed for scalability. It utilizes readily available starting materials like isatin and operates under relatively standard thermal conditions (up to 120°C), making it suitable for transition from gram-level laboratory synthesis to multi-kilogram commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the FeCl3-catalyzed synthesis route for 2-trifluoromethyl quinazolinones. As a leading CDMO partner, we possess the technical expertise to optimize this pathway for your specific project needs, ensuring that the transition from bench-scale discovery to commercial manufacturing is seamless. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to handle the specific thermal and safety requirements of this exothermic cyclization process. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of residual iron and other impurities, guaranteeing a product that meets the highest pharmacopeial standards.
We invite you to collaborate with us to leverage this cost-effective technology for your next drug development program. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in delivering high-quality, affordable chemical solutions.
