Advanced Manufacturing of Selpercatinib Intermediates for Global Pharmaceutical Supply Chains
Introduction to Next-Generation RET Inhibitor Manufacturing
The pharmaceutical landscape for oncology treatments is rapidly evolving, with a specific focus on targeting the RET proto-oncogene, a critical driver in non-small cell lung cancer (NSCLC) and medullary thyroid cancer (MTC). Central to this therapeutic revolution is Selpercatinib, a highly selective and potent oral RET inhibitor that has demonstrated exceptional clinical efficacy. However, the commercial viability of such complex small molecules hinges entirely on the robustness and efficiency of their synthetic pathways. Patent CN111057075B, filed in 2021, introduces a groundbreaking preparation method that addresses the historical bottlenecks of Selpercatinib production. This intellectual property outlines a streamlined synthetic strategy that not only enhances the total yield of reaction products but also ensures product purity surpasses 99.5 percent, a critical benchmark for regulatory approval and patient safety.
For global supply chain stakeholders, the implications of this technology are profound. Traditional synthesis routes often suffer from excessive step counts and harsh conditions that compromise scalability. In contrast, the methodology described in CN111057075B leverages mild reaction conditions and readily available intermediates to create a more resilient manufacturing pipeline. By optimizing the coupling of key structural motifs, this approach minimizes waste generation and maximizes atom economy. As we delve deeper into the technical specifics, it becomes evident that this patent represents a pivotal shift towards more sustainable and cost-effective production of high-value API intermediates, positioning manufacturers who adopt this route as leaders in the competitive oncology market.
![Chemical structure of Selpercatinib (Formula I) showing the core pyrazolo[1,5-a]pyridine scaffold](/insights/img/selpercatinib-synthesis-route-pharma-supplier-20260305104917-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN111057075B, the industrial synthesis of Selpercatinib was plagued by inefficiencies inherent in multi-step linear sequences. The conventional pathway, as illustrated in the background art, necessitates a cumbersome three-step transformation to install the critical piperazine-pyridine side chain onto the core scaffold. This legacy approach typically involves an initial halogenation step to activate the leaving group, followed by a deprotection sequence to reveal the reactive amine, and finally, a reductive amination to forge the carbon-nitrogen bond. Each of these discrete operations introduces significant opportunities for yield loss, impurity accumulation, and increased solvent consumption.
Furthermore, the reliance on halogenation reagents and subsequent reduction steps often requires stringent safety controls and specialized waste treatment protocols, driving up operational expenditures. The cumulative effect of these additional steps is a depressed overall yield, which directly impacts the cost of goods sold (COGS) and limits the ability to meet surging global demand. From a supply chain perspective, the complexity of the old route creates fragility; any disruption in the supply of specialized reagents for the halogenation or reduction steps can halt production entirely. This lack of process robustness is unacceptable for the manufacture of life-saving oncology medications where continuity of supply is paramount.
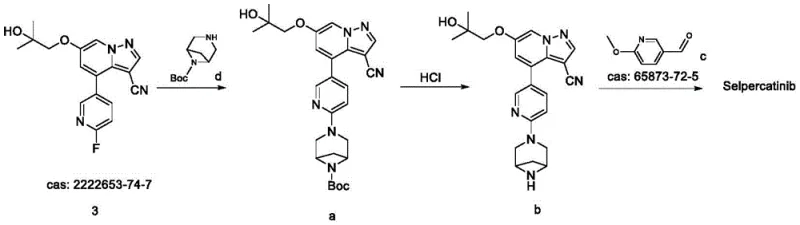
The Novel Approach
The methodology disclosed in CN111057075B fundamentally reimagines the construction of the Selpercatinib molecule by collapsing the final assembly into a direct, high-efficiency coupling reaction. Instead of the laborious three-step sequence, the novel approach utilizes a pre-functionalized intermediate, Compound 3, which reacts directly with the commercially available Compound 4 in a single pot. This strategic simplification eliminates the need for intermediate isolation, halogenation, and reductive amination, thereby drastically reducing the processing time and resource intensity. The reaction proceeds under mild thermal conditions, typically between 70°C and 80°C, utilizing potassium acetate (KOAc) as a benign base in dimethyl sulfoxide (DMSO).
This direct nucleophilic aromatic substitution not only accelerates the synthesis but also significantly enhances the purity profile of the final product. By avoiding the formation of halogenated by-products and reduction-related impurities, the process inherently produces a cleaner crude material that requires less aggressive purification. The patent data indicates that this route consistently delivers Selpercatinib with HPLC purity exceeding 99.5 percent, often reaching 99.8 percent without the need for complex chromatographic separations. For procurement managers, this translates to a reliable API intermediate supplier capable of delivering high-quality material with reduced lead times and lower environmental impact, aligning perfectly with modern green chemistry initiatives.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving the success of this novel route is a base-mediated nucleophilic aromatic substitution (SNAr), facilitated by the unique electronic properties of the pyridine ring system. In the second step of the synthesis, the secondary amine of the piperazine ring in Compound 4 acts as a potent nucleophile, attacking the electron-deficient carbon of the pyridine ring in Compound 3. The presence of the electron-withdrawing nitrogen atom within the pyridine ring activates the position ortho to it, making it susceptible to nucleophilic attack even under relatively mild conditions. The use of potassium acetate (KOAc) serves a dual purpose: it acts as a weak base to maintain the reaction medium's pH and potentially assists in the deprotonation of the ammonium species formed during the transition state, driving the equilibrium towards product formation.
Crucially, the choice of solvent plays a pivotal role in the mechanistic pathway. Dimethyl sulfoxide (DMSO) is a polar aprotic solvent that effectively solvates the cationic potassium species while leaving the nucleophilic amine 'naked' and highly reactive. This solvation effect lowers the activation energy of the rate-determining step, allowing the reaction to proceed efficiently at 75°C over a 24-hour period. The patent examples demonstrate that maintaining this specific temperature window is essential; temperatures below 70°C result in incomplete conversion, while temperatures above 85°C may promote degradation of the sensitive nitrile or ether functionalities. Understanding these kinetic parameters allows process chemists to fine-tune the reaction for maximum throughput and minimal impurity generation.
Impurity control is another critical aspect of this mechanism. The high selectivity of the SNAr reaction ensures that the nucleophile attacks exclusively at the desired fluorine-bearing carbon, minimizing regio-isomers. Furthermore, the mild basicity of KOAc prevents the hydrolysis of the nitrile group or the cleavage of the acid-labile ether linkage, which are common failure modes in harsher alkaline conditions. The workup procedure, involving the addition of water to precipitate the product followed by pulping with 30% aqueous DMSO, exploits the differential solubility of the product versus unreacted starting materials and inorganic salts. This physical purification step is highly effective at removing trace organic impurities, contributing to the exceptional >99.5% purity observed in the final isolated solid, thereby satisfying the rigorous quality standards required for pharmaceutical intermediates.
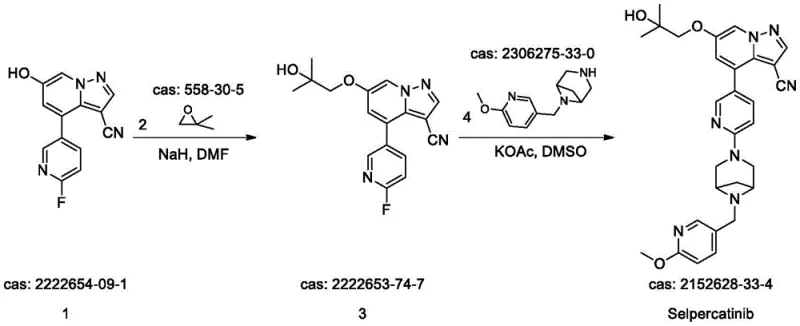
How to Synthesize Selpercatinib Efficiently
The practical execution of this synthesis requires precise control over stoichiometry and thermal profiles to replicate the high yields reported in the patent literature. The process begins with the preparation of the key intermediate, Compound 3, via an etherification reaction between a hydroxy-substituted pyrazolopyridine and a protected glycidol derivative. This step utilizes sodium hydride (NaH) in DMF, requiring careful temperature management to prevent exothermic runaway. Once Compound 3 is secured, the final coupling with Compound 4 is performed in DMSO. The detailed standardized synthetic steps below outline the exact molar ratios, temperatures, and workup procedures necessary to achieve the reported 86% yield and high purity.
- React Compound 1 with Compound 2 using NaH in DMF at 80°C for 24 hours to form Compound 3.
- Couple Compound 3 with Compound 4 using KOAc in DMSO at 75°C for 24 hours.
- Purify the final product by filtration and pulping with 30% aqueous DMSO to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN111057075B synthesis route offers transformative economic and logistical benefits. The most immediate impact is seen in the drastic simplification of the manufacturing workflow. By eliminating three distinct synthetic operations—halogenation, deprotection, and reductive amination—the process significantly reduces the consumption of raw materials, solvents, and energy. This reduction in process complexity directly correlates to a substantial decrease in manufacturing costs, as fewer unit operations mean lower labor overhead, reduced equipment occupancy time, and diminished waste disposal fees. The ability to produce high-purity material without extensive chromatographic purification further enhances cost efficiency by minimizing the use of expensive silica gel and large volumes of elution solvents.
Supply chain reliability is another major advantage conferred by this technology. The route relies on Compound 4, an intermediate that is described as 'existing and easily obtained,' suggesting a robust and mature supply base for this critical building block. This reduces the risk of supply disruptions associated with custom-synthesized specialty reagents. Moreover, the reaction conditions are exceptionally mild, operating at temperatures between 70°C and 85°C using common industrial solvents like DMF and DMSO. These conditions are easily scalable from kilogram to multi-ton batches using standard stainless steel reactors, ensuring that production capacity can be rapidly expanded to meet market demand without the need for exotic high-pressure or cryogenic equipment.
Environmental compliance and sustainability are increasingly becoming key performance indicators for pharmaceutical suppliers. This novel route aligns well with green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product). The avoidance of halogenating agents and heavy metal catalysts (often used in reductive aminations) simplifies the effluent treatment process and reduces the environmental footprint of the manufacturing site. For partners seeking a reliable API intermediate supplier, this process offers a pathway to produce Selpercatinib that is not only economically superior but also environmentally responsible, mitigating regulatory risks and enhancing the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical disclosures within CN111057075B, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating technology transfer and for procurement professionals assessing supplier qualifications.
Q: What is the primary advantage of the CN111057075B synthesis route?
A: The primary advantage is the significant reduction in reaction steps compared to prior art, eliminating halogenation and reductive amination, which improves total yield and simplifies purification.
Q: What purity levels can be achieved with this method?
A: The patented method consistently achieves HPLC purity levels exceeding 99.5%, meeting stringent requirements for pharmaceutical active ingredients.
Q: Are the reaction conditions suitable for large-scale production?
A: Yes, the process utilizes mild temperatures (70-85°C) and common industrial solvents like DMF and DMSO, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selpercatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for next-generation oncology therapeutics like Selpercatinib. Our technical team has thoroughly analyzed the CN111057075B patent and possesses the expertise to implement this advanced two-step methodology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Selpercatinib intermediate meets the >99.5% purity threshold required for downstream API synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and robust manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. Let us help you secure a stable, high-quality supply of Selpercatinib intermediates that drives your drug development programs forward with speed and confidence.
