Advanced One-Pot Tandem Synthesis of High-Purity Oxindole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries constantly seek robust, scalable, and cost-effective methodologies for constructing privileged scaffolds. Among these, the oxindole core (2-indolinone) stands out as a critical structural motif found in numerous bioactive natural products and approved drugs such as Ropinirole, Tenidap, and Ziprasidone. A groundbreaking approach detailed in Chinese patent CN103304468A introduces a highly efficient one-pot tandem synthesis method that transforms simple, commercially available substituted o-nitrohalobenzenes into high-purity oxindoles. This technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to step economy, waste generation, and raw material costs. By integrating aromatic nucleophilic substitution, Krapcho deesterification, and reductive cyclization into a single seamless operation, this method offers a compelling value proposition for manufacturers aiming to optimize their supply chains for complex heterocyclic intermediates.
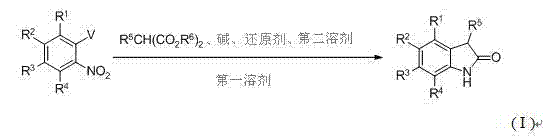
Traditionally, the construction of the oxindole framework has relied on methodologies that are often fraught with inefficiencies and operational complexities. Conventional routes, such as the oxidation of indoles or the reduction of isatins, frequently necessitate harsh reaction conditions, expensive catalysts, and multi-step sequences that degrade overall yield. For instance, methods starting from substituted o-nitrotoluene often require cumbersome multi-step transformations and the use of costly platinum catalysts for hydrogenation. Other approaches involving chloroacetanilides demand strict anhydrous and oxygen-free conditions, utilizing strong bases like LDA or potassium amide in cryogenic solvents, which poses significant safety and scalability hurdles. Furthermore, existing protocols starting from o-halogenated nitrobenzenes typically involve 2 to 4 distinct steps with intermediate isolations and column chromatography purifications, leading to substantial solvent waste and prolonged production cycles. These limitations severely impact the cost of goods sold (COGS) and hinder the ability to rapidly scale production to meet market demand.
In stark contrast, the novel one-pot tandem strategy described in the patent data revolutionizes this landscape by collapsing three distinct chemical transformations into a single reactor vessel. This approach begins with the reaction of a substituted o-nitrohalobenzene with a malonate ester in the presence of a base and a first solvent. Unlike traditional methods that would require quenching and isolating the substitution product, this process proceeds directly to a thermal deesterification step. Remarkably, the metal halide salt generated as a byproduct in the initial nucleophilic substitution serves as an intrinsic promoter for the subsequent Krapcho deesterification, eliminating the need for external additives like lithium chloride. Finally, the introduction of a reducing agent and a second solvent facilitates the reductive cyclization to form the target oxindole. This telescoped sequence not only drastically reduces processing time but also minimizes the consumption of solvents and reagents, aligning perfectly with green chemistry principles and modern manufacturing efficiency standards.
Mechanistic Insights into One-Pot Tandem Cyclization
The success of this methodology hinges on a sophisticated understanding of reaction compatibility and byproduct utilization. The mechanism initiates with an aromatic nucleophilic substitution (SNAr) where the malonate anion, generated by the deprotonation of the diester by a base such as potassium carbonate or sodium hydride, attacks the electron-deficient aromatic ring of the o-nitrohalobenzene. This step displaces the halogen atom (V), forming a carbon-carbon bond and releasing a metal halide salt (MV). In conventional synthesis, this salt is often treated as waste; however, in this innovative protocol, it plays a pivotal catalytic role. As the reaction temperature is elevated to between 110°C and 180°C, the in-situ generated metal halide facilitates the Krapcho deesterification, promoting the hydrolysis and decarboxylation of the malonate moiety without requiring additional salt promoters. This internal recycling of reagents is a masterstroke of atom economy.
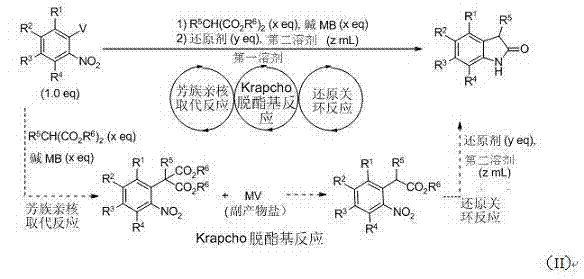
Following the deesterification, the intermediate undergoes a reductive ring-closure to finalize the oxindole structure. This step involves the reduction of the nitro group to an amine, which subsequently cyclizes onto the adjacent carbonyl. The patent highlights the versatility of reducing agents, ranging from transition metals like palladium on carbon under hydrogen atmosphere to base metals like iron, zinc, magnesium, or aluminum in acidic media. The choice of reducing system allows for fine-tuning based on the sensitivity of other functional groups present on the substrate. For example, substrates containing acid-sensitive groups might benefit from catalytic hydrogenation, while those tolerant of acidic conditions can utilize cost-effective iron powder in acetic acid. This mechanistic flexibility ensures that impurity profiles are tightly controlled, as the direct progression from one step to the next minimizes the exposure of reactive intermediates to potential degradation pathways, thereby ensuring the high purity required for pharmaceutical applications.
How to Synthesize Oxindole Efficiently
Implementing this one-pot tandem synthesis requires precise control over reaction parameters to maximize yield and minimize side reactions. The process generally involves sequentially adding the malonate ester, base, and first solvent, followed by heating to initiate the substitution. Once the starting material is consumed, the temperature is ramped up for deesterification, and finally, the reducing system is introduced for cyclization. The beauty of this method lies in its operational simplicity; there is no need for intermediate workups, extractions, or drying steps between the three major transformations. Detailed standard operating procedures, including specific stoichiometric ratios, temperature ramps, and solvent choices tailored to specific substrates, are essential for reproducible results. For a comprehensive guide on executing this synthesis with optimal parameters for your specific target molecule, please refer to the standardized protocol below.
- Perform aromatic nucleophilic substitution between substituted o-nitrohalobenzene and malonate using a base in a first solvent at 25°C to 160°C.
- Execute Krapcho deesterification by heating the reaction mixture to 110°C to 180°C, utilizing the metal halide byproduct from the previous step as a promoter.
- Complete reductive ring-closure by adding a reducing agent and a second solvent, heating to 25°C to 150°C until the intermediate is fully converted.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this one-pot tandem synthesis offers transformative benefits that directly address the pain points of procurement managers and supply chain directors. The most immediate advantage is the drastic simplification of the manufacturing process. By eliminating the need for intermediate isolation and purification, the method significantly reduces the consumption of solvents, filtration media, and labor hours associated with multi-step batch processing. This reduction in unit operations translates directly into lower manufacturing costs and a smaller environmental footprint, as less waste solvent requires treatment or disposal. Furthermore, the reliance on commodity chemicals such as substituted nitrohalobenzenes and dialkyl malonates ensures a stable and secure supply of raw materials, mitigating the risks associated with sourcing specialized or exotic reagents that often plague complex API intermediate production.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the elimination of expensive catalysts and the reduction of processing steps. Traditional methods often rely on precious metal catalysts like palladium or platinum, which represent a significant capital tie-up and require complex recovery systems to prevent loss. In contrast, this method frequently utilizes abundant base metals like iron or zinc, or catalytic amounts of cheaper alternatives, drastically lowering reagent costs. Additionally, the internal utilization of the metal halide byproduct removes the necessity of purchasing external promoters like lithium chloride, further trimming the bill of materials. The consolidation of three reaction steps into one pot also means reduced energy consumption for heating and cooling cycles across multiple vessels, contributing to substantial overall cost savings in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of this synthetic route. The tolerance of the reaction conditions to moisture and oxygen in several steps reduces the need for specialized, high-maintenance equipment such as gloveboxes or rigorous nitrogen purging systems, allowing for production in standard glass-lined reactors. This ease of operation facilitates faster technology transfer between sites and reduces the likelihood of batch failures due to environmental excursions. Moreover, the broad substrate scope means that a single manufacturing line can be adapted to produce a wide variety of oxindole derivatives simply by changing the starting nitrohalobenzene, providing agility to respond to shifting market demands without significant retooling investments.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the straightforward nature of this one-pot method mitigates many of these risks. The absence of intermediate isolations reduces the physical handling of potentially hazardous powders, improving operator safety. From an environmental standpoint, the high atom economy achieved by recycling the halide byproduct and the reduction in solvent usage align with increasingly stringent global environmental regulations. This makes the process not only compliant but also future-proof against tightening restrictions on industrial waste discharge, ensuring long-term viability for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthetic methodology is crucial for making informed sourcing decisions. The following questions address common inquiries regarding the feasibility, scope, and quality implications of adopting this one-pot oxindole synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a transparent view of what partners can expect when integrating this technology into their supply chains.
Q: What are the primary advantages of this one-pot method over conventional oxindole synthesis?
A: This method eliminates the need for intermediate isolation and purification steps required in traditional multi-step syntheses. It utilizes inexpensive starting materials like o-nitrohalobenzenes and malonates, and uniquely recycles the metal halide byproduct from the substitution step to promote the subsequent deesterification, significantly improving atom economy and reducing waste.
Q: Does this synthesis protocol support a wide range of functional groups?
A: Yes, the protocol demonstrates excellent substrate universality. It is compatible with various substituents including halogens (F, Cl, Br, I), alkyl groups, alkoxy groups, esters, amides, and carbonyls at multiple positions on the benzene ring, making it highly versatile for synthesizing diverse oxindole libraries.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process operates under relatively mild conditions without the need for strict anhydrous or oxygen-free environments for most steps. It avoids expensive noble metal catalysts like palladium in favor of cheaper metals like iron or zinc, and simplifies post-processing, which are critical factors for successful commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxindole Supplier
The technological potential of this one-pot tandem synthesis is immense, offering a pathway to high-quality oxindole derivatives that are essential for modern drug discovery and development. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of process innovation, translating such advanced academic and patent concepts into reliable commercial realities. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and chemical requirements of this tandem process, ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite you to explore how this optimized synthesis can enhance your project's economics and timeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. We encourage you to contact our technical procurement team today to request specific COA data for our oxindole portfolio and to discuss route feasibility assessments for your custom synthesis needs. Let us partner with you to drive efficiency and quality in your pharmaceutical supply chain.
