Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
The landscape of modern pharmaceutical discovery is increasingly reliant on complex heterocyclic scaffolds that offer unique biological activities and metabolic stability. Patent CN115353511A introduces a groundbreaking multi-component methodology for the efficient construction of carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technological advancement addresses a critical bottleneck in organic synthesis by enabling the one-pot assembly of these privileged structures under remarkably mild conditions. By leveraging a transition metal palladium-catalyzed cascade reaction, the process circumvents the traditional reliance on hazardous carbon monoxide gas, instead utilizing a safe in-situ generation system. For R&D directors and process chemists, this represents a significant leap forward in accessing diverse chemical space for drug discovery programs, particularly for targets requiring fluorinated heterocycles which are known to enhance bioavailability and binding affinity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged biheterocyclic systems has been fraught with significant operational challenges and safety concerns. Traditional carbonylation reactions typically necessitate the use of pressurized carbon monoxide gas, which poses severe toxicity risks and requires specialized high-pressure equipment that is often unavailable in standard laboratory settings. Furthermore, conventional routes often involve multi-step sequences with poor atom economy, where the introduction of the carbonyl bridge requires pre-functionalized substrates and harsh reaction conditions that limit functional group tolerance. The direct coupling of two distinct heterocyclic units frequently suffers from low regioselectivity and requires expensive coupling reagents, leading to substantial waste generation and increased production costs. These limitations have historically restricted the widespread adoption of carbonyl-bridged scaffolds in early-stage drug discovery, forcing medicinal chemists to settle for simpler, less potent analogues due to synthetic inaccessibility.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this synthetic landscape by employing a safe, efficient, and highly modular multi-component reaction strategy. Instead of external CO gas, the process utilizes a mixture of formic acid and acetic anhydride to generate carbon monoxide in situ, effectively acting as a liquid surrogate that simplifies reactor design and enhances operator safety. This novel approach allows for the simultaneous formation of multiple chemical bonds, including C-C and C-N linkages, in a single operational step starting from readily available trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. The reaction proceeds at a mild temperature of 30°C, demonstrating exceptional compatibility with sensitive functional groups such as halogens, nitro groups, and trifluoromethyl moieties. This level of chemoselectivity ensures that complex molecular architectures can be built without the need for extensive protecting group strategies, thereby streamlining the overall synthetic route and improving the speed of lead optimization cycles.
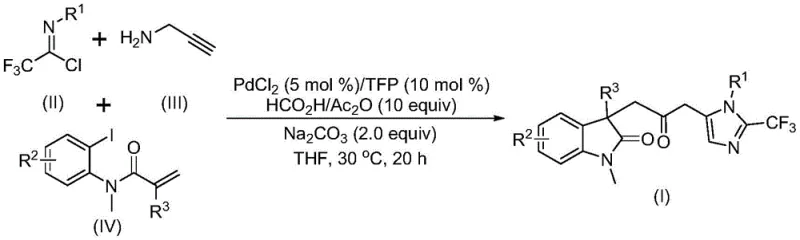
Mechanistically, this transformation is a masterpiece of organometallic chemistry, orchestrated by a palladium catalyst system that facilitates a complex cascade of events. The reaction likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type cyclization that constructs the indolinone core, establishing the first heterocyclic ring with high stereocontrol. Subsequently, the in-situ generated carbon monoxide inserts into the alkyl-palladium bond to form an acyl-palladium species, effectively installing the crucial carbonyl bridge. In parallel, the trifluoroethylimidoyl chloride reacts with propargylamine under basic conditions to form a trifluoroacetamidine intermediate, which then undergoes isomerization. The final stage involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active catalyst.
From an impurity control perspective, the robustness of this catalytic cycle offers distinct advantages for manufacturing high-purity intermediates. The use of a well-defined palladium/TFP (tris(2-furyl)phosphine) catalyst system minimizes the formation of homocoupling byproducts that are common in less selective cross-coupling reactions. The mild reaction temperature of 30°C further suppresses thermal degradation pathways and polymerization side reactions that often plague acrylamide chemistry. Additionally, the stoichiometric balance of reagents, particularly the slight excess of propargylamine and acrylamide relative to the imidoyl chloride, drives the reaction to completion while ensuring that unreacted starting materials can be easily removed during the workup. The resulting crude mixtures are amenable to standard purification techniques like silica gel column chromatography, yielding products with purity profiles suitable for rigorous biological testing and subsequent process development.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility. The protocol involves dissolving the palladium catalyst, ligand, and base in an aprotic solvent such as tetrahydrofuran (THF), which has been identified as the optimal medium for solubilizing all components and facilitating the catalytic turnover. The sequential addition of the three key building blocks—trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative—must be managed to ensure proper mixing before the carbonylation cascade begins. The reaction mixture is then maintained at 30°C for a duration of 12 to 20 hours, allowing sufficient time for the multi-step cyclization to reach completion. Detailed standardized operating procedures for scaling this reaction are outlined below.
- Combine palladium chloride catalyst, TFP ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12-20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of toxic carbon monoxide gas removes the need for specialized gas handling infrastructure, cylinder storage, and associated safety monitoring systems, resulting in substantial capital expenditure savings and reduced regulatory compliance burdens. Furthermore, the starting materials, including trifluoroethylimidoyl chloride and propargylamine, are commercially available and cost-effective, ensuring a stable and reliable supply chain for large-scale production campaigns. The ability to run the reaction at ambient pressure and low temperature significantly lowers energy consumption compared to high-pressure carbonylation processes, contributing to a more sustainable and cost-efficient manufacturing footprint.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction drastically reduces the number of unit operations required, eliminating the need for intermediate isolation and purification steps that typically drive up processing costs. By avoiding the use of expensive noble metal catalysts in favor of economical palladium chloride and simple phosphine ligands, the overall catalyst cost per kilogram of product is significantly minimized. The high atom economy of the multi-component approach ensures that a greater proportion of raw material mass is incorporated into the final product, reducing waste disposal fees and maximizing raw material utilization efficiency.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard organic solvents like THF mitigates the risk of supply chain disruptions often associated with exotic or proprietary reagents. The robustness of the reaction conditions allows for flexible scheduling and batch processing, enabling manufacturers to respond quickly to fluctuating demand without lengthy changeover times. Additionally, the scalability demonstrated in the patent data, extending from milligram to gram scales with consistent performance, provides confidence in the technology's readiness for ton-scale commercial production.
- Scalability and Environmental Compliance: The absence of high-pressure gases and the use of mild temperatures simplify the engineering requirements for scale-up, allowing the process to be transferred to standard glass-lined or stainless steel reactors without major modifications. The simplified workup procedure, involving filtration and chromatography, generates less hazardous waste compared to traditional methods that might require quenching of reactive intermediates or extensive aqueous extractions. This aligns with green chemistry principles, facilitating easier environmental permitting and reducing the overall ecological impact of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, aiming to clarify feasibility for potential licensees or contract manufacturing partners. Understanding these nuances is critical for assessing the fit of this technology within existing production portfolios.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This method utilizes a formic acid and acetic anhydride mixture as a safe carbon monoxide substitute, eliminating the need for handling toxic, high-pressure CO gas cylinders, thereby significantly reducing operational hazards and infrastructure costs.
Q: What is the substrate scope for the R1, R2, and R3 groups in this synthesis?
A: The process demonstrates excellent functional group tolerance, accommodating alkyl, substituted aryl (with methyl, methoxy, halogens, trifluoromethyl, or nitro groups), and benzyl substituents, allowing for the diversification of the final biheterocyclic scaffold.
Q: Is this synthesis scalable for industrial production?
A: Yes, the patent explicitly mentions successful expansion to gram-scale reactions with high efficiency and simple post-treatment procedures, indicating strong potential for commercial scale-up in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN115353511A for accelerating drug discovery pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop innovation to market-ready supply is seamless and secure. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize this carbonylation process for your specific target molecules. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and purity requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can drive efficiency and reliability in your supply chain.
