Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Global Pharmaceutical Manufacturing
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Global Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is documented in patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These heterocyclic scaffolds are ubiquitous in modern medicinal chemistry, serving as critical core structures for a wide array of bioactive molecules. As illustrated in the structural diversity of current market drugs, the 1,2,4-triazole motif is found in prominent therapeutics such as Maraviroc, Sitagliptin, and Deferasirox, highlighting its immense value in treating HIV, diabetes, and iron overload disorders respectively. The introduction of trifluoromethyl groups into these scaffolds further enhances their pharmacokinetic profiles, improving metabolic stability and membrane permeability, which makes the development of efficient synthesis methods for these specific derivatives a high priority for R&D teams globally.
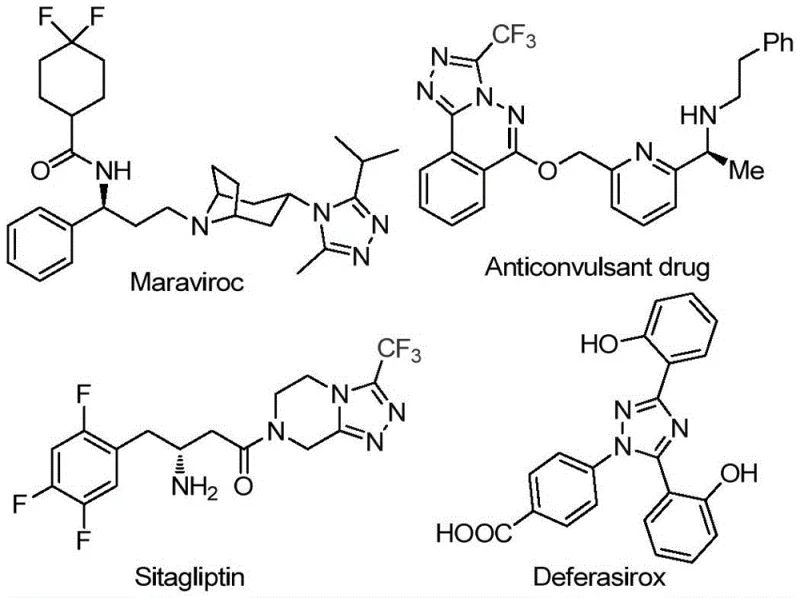
The strategic importance of this patent lies in its ability to address long-standing bottlenecks in heterocycle synthesis. Traditional methods for constructing polysubstituted triazoles often rely on harsh conditions or expensive transition metal catalysts that complicate downstream processing. By leveraging a metal-free, iodine-promoted pathway, this technology offers a streamlined approach that aligns perfectly with the goals of green chemistry and industrial efficiency. For procurement managers and supply chain directors, this represents a tangible opportunity to optimize the sourcing of high-value intermediates. The method utilizes cheap and readily available starting materials, such as aryl ethyl ketones and trifluoroethylimide hydrazides, ensuring that the supply chain remains robust and less susceptible to the volatility often associated with precious metal catalysts. This foundational shift in synthetic strategy paves the way for more reliable production of complex pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4,5-trisubstituted 1,2,4-triazoles, particularly those bearing both acyl and trifluoromethyl groups, has been fraught with technical challenges that impact commercial viability. Conventional protocols frequently depend on transition metal catalysis, involving elements like copper or palladium, which introduce significant cost burdens and environmental liabilities. The presence of these heavy metals necessitates rigorous purification steps to meet the stringent residual metal limits imposed by regulatory bodies such as the FDA and EMA for active pharmaceutical ingredients (APIs). Furthermore, many traditional routes require strictly anhydrous and oxygen-free conditions, demanding specialized equipment and inert atmosphere handling that drastically increases operational complexity and capital expenditure. These factors collectively contribute to longer lead times and higher manufacturing costs, creating friction in the supply chain for high-purity pharmaceutical intermediates. Additionally, the substrate scope in older methods is often limited, struggling to tolerate diverse functional groups without significant yield degradation.
The Novel Approach
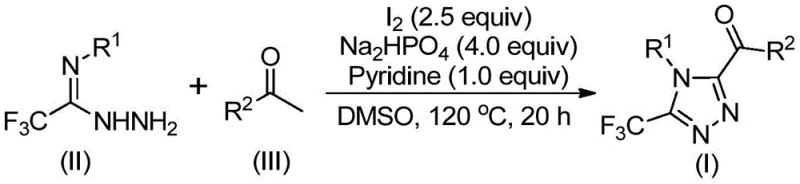
In stark contrast, the methodology outlined in patent CN113105402B presents a paradigm shift by employing a non-metallic iodine promotion system in dimethyl sulfoxide (DMSO). This innovative route eliminates the need for toxic heavy metal catalysts entirely, thereby removing the costly and time-consuming heavy metal scavenging steps from the downstream processing workflow. The reaction proceeds under relatively mild conditions without the strict requirement for anhydrous or oxygen-free environments, significantly simplifying the operational procedure and reducing the barrier to entry for scale-up. The use of elemental iodine, a cheap and abundant reagent, coupled with inexpensive bases like sodium dihydrogen phosphate and pyridine, ensures that the raw material costs remain low. This approach not only enhances the economic feasibility of producing these complex heterocycles but also broadens the functional group tolerance, allowing for the synthesis of a diverse library of derivatives with varying electronic and steric properties. The simplicity of the work-up, involving basic filtration and column chromatography, further underscores the practical utility of this method for industrial applications.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The elegance of this synthesis lies in its tandem reaction mechanism, which seamlessly integrates oxidation and cyclization steps into a single pot. The process initiates with the iodination and subsequent Kornblum oxidation of the aryl ethyl ketone substrate, facilitated by the synergistic action of elemental iodine and the DMSO solvent. This transformation generates an reactive aryl diketone intermediate in situ, which serves as the electrophilic partner for the subsequent condensation. The trifluoroethylimide hydrazide then attacks this activated species, undergoing dehydration to form a hydrazone intermediate. This step is crucial as it sets the stage for the final ring closure. The presence of pyridine and sodium dihydrogen phosphate acts as a buffer system, promoting the intramolecular cyclization that constructs the 1,2,4-triazole core. The entire sequence is driven by the thermodynamic stability of the aromatic triazole ring and the formation of stable byproducts, ensuring high conversion rates even with electron-deficient or sterically hindered substrates.
From an impurity control perspective, this mechanism offers distinct advantages over multi-step sequences. By avoiding the isolation of unstable intermediates, the potential for decomposition or side reactions is minimized. The use of DMSO as both solvent and reactant in the oxidation step ensures a homogeneous reaction environment, which promotes consistent heat and mass transfer, critical for maintaining product quality during scale-up. The specific stoichiometry, typically employing a molar ratio of trifluoroethylimide hydrazide to aryl ethyl ketone of 1:2, ensures that the ketone is in excess, driving the equilibrium towards the desired product and suppressing the formation of symmetric byproducts. This precise control over reaction parameters allows for the generation of high-purity 3,4,5-trisubstituted 1,2,4-triazoles with well-defined impurity profiles, a key requirement for R&D directors focusing on regulatory compliance and process robustness. The ability to fine-tune the electronic nature of the R1 and R2 groups further allows chemists to optimize the physicochemical properties of the final molecule without compromising the efficiency of the core synthesis.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature control to maximize yield and purity. The process begins by dissolving the aryl ethyl ketone and a portion of elemental iodine in DMSO, followed by heating to initiate the oxidation phase. Once the intermediate is formed, the remaining reagents including the hydrazide, base, and additional iodine are introduced to trigger the cyclization. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and work-up procedures, are critical for reproducibility and are outlined in the technical guide below.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis method translates into immediate and tangible strategic benefits. The elimination of precious metal catalysts removes a major cost driver and supply risk, as the market for palladium and copper can be volatile and subject to geopolitical constraints. By switching to elemental iodine and commodity chemicals like acetophenones, the raw material basket becomes significantly more stable and cost-effective. This shift not only lowers the direct cost of goods sold (COGS) but also simplifies the vendor qualification process, as these starting materials are widely available from multiple global suppliers. Furthermore, the simplified downstream processing reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media, contributing to overall operational efficiency and waste reduction.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the complete avoidance of transition metal catalysts. In traditional pharmaceutical manufacturing, the cost of catalysts combined with the expensive purification technologies required to remove trace metals to ppm levels can be substantial. By utilizing a metal-free protocol, manufacturers can bypass these costly unit operations entirely. Additionally, the use of DMSO, a high-boiling polar aprotic solvent, allows for reactions to be run at elevated temperatures without the need for high-pressure equipment, further reducing capital and energy expenditures. The high atom economy of the tandem reaction also means less waste generation, lowering disposal costs and aligning with sustainability goals. These factors combine to create a leaner, more cost-efficient manufacturing process that enhances margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commoditized raw materials. Aryl ethyl ketones and elemental iodine are produced on a massive industrial scale for various sectors, ensuring a steady and reliable supply flow that is less prone to the shortages often seen with specialized organometallic reagents. The robustness of the reaction conditions, which do not require stringent exclusion of moisture or oxygen, reduces the risk of batch failures due to environmental fluctuations. This operational forgiveness is crucial for maintaining consistent delivery schedules to downstream API manufacturers. Moreover, the scalability of the process from gram to kilogram levels without significant re-optimization ensures that supply can be ramped up quickly to meet market demand, providing a competitive edge in fast-moving therapeutic areas.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method offers a cleaner profile. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. DMSO is a recyclable solvent, and the benign nature of the byproducts facilitates easier waste management. The process is inherently safer as it avoids the use of pyrophoric reagents or highly toxic gases often associated with alternative triazole synthesis routes. This alignment with green chemistry principles not only helps in meeting increasingly strict environmental regulations but also enhances the corporate social responsibility profile of the manufacturing entity. The ease of scale-up, demonstrated by the successful expansion to gram-level synthesis in the patent data, indicates a clear path to multi-ton production, making it a viable candidate for commercial-scale API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on the practical implementation and advantages of this novel route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production pipelines.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metallic promoter, completely eliminating the need for costly palladium or copper catalysts and the associated heavy metal removal steps.
Q: What are the optimal reaction conditions for scaling this triazole synthesis?
A: The reaction operates effectively in dimethyl sulfoxide (DMSO) at temperatures between 110°C and 130°C, utilizing a molar ratio of ketone to hydrazide of approximately 2:1 for optimal conversion.
Q: Is this method suitable for producing trifluoromethyl-substituted drug intermediates?
A: Yes, the method is specifically designed to incorporate trifluoromethyl groups efficiently, which are critical for enhancing the metabolic stability and lipophilicity of pharmaceutical candidates like sitagliptin analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from early-stage discovery through to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, backed by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in heterocyclic chemistry, particularly in the synthesis of fluorinated triazoles, positions us as a strategic partner capable of navigating the complexities of modern pharmaceutical synthesis while maintaining cost efficiency and supply reliability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you accelerate your timeline to market with a supply chain partner dedicated to excellence and innovation.
