Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
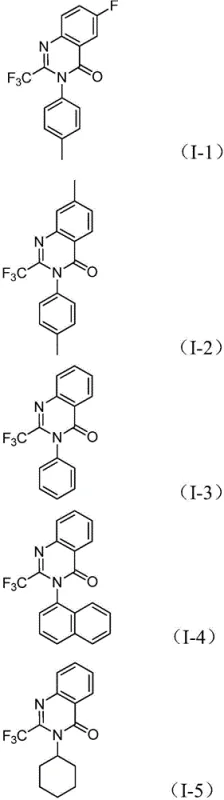
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those with enhanced metabolic stability. Patent CN112480015B introduces a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a privileged scaffold found in numerous bioactive molecules ranging from antifungals to anticancer agents. This technology leverages a sophisticated palladium-catalyzed carbonylation cascade that transforms inexpensive nitro compounds and trifluoroethylimidoyl chlorides into high-value intermediates. By integrating the installation of the trifluoromethyl group directly into the ring-forming step, this process addresses critical pain points in modern drug discovery, offering a streamlined pathway that bypasses the need for hazardous gaseous reagents and complex multi-step sequences. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for quinazolinone-based active pharmaceutical ingredients (APIs) while maintaining stringent purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on synthetic routes that are fraught with operational hazards and economic inefficiencies. Traditional methods often necessitate the use of high-pressure carbon monoxide gas in conjunction with ruthenium or platinum catalysts to effect the reductive N-heterocyclization of nitro-substituted benzamides, requiring specialized autoclave equipment that increases capital expenditure and safety protocols. Alternative approaches involving iron-catalyzed condensation or palladium-catalyzed cyclization of 2-bromoformylaniline derivatives frequently suffer from narrow substrate scope and the requirement for expensive, pre-activated starting materials that are not readily available on a metric-ton scale. Furthermore, many existing protocols exhibit poor atom economy and generate substantial waste streams due to low yields and the necessity for extensive purification steps to remove metal residues and side products. These limitations create bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, leading to extended lead times and inflated manufacturing costs that ultimately impact the affordability of the final therapeutic agents.
The Novel Approach
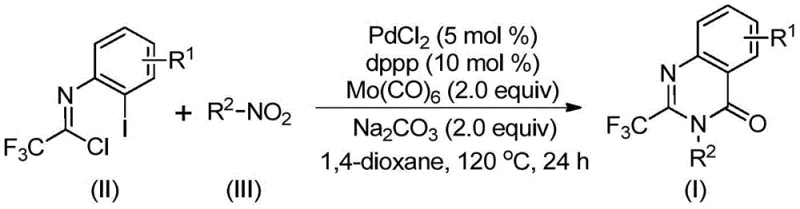
In stark contrast to these legacy techniques, the methodology disclosed in CN112480015B utilizes a transition metal palladium-catalyzed carbonylation cascade that operates under remarkably mild and accessible conditions. By employing molybdenum hexacarbonyl as a solid carbon monoxide surrogate, the process eliminates the dangers associated with handling toxic CO gas while ensuring a steady, controlled release of the carbonyl source in situ. The reaction seamlessly couples readily available nitro compounds with trifluoroethylimidoyl chlorides in a single pot, effectively merging the reduction of the nitro group, the formation of the carbon-nitrogen bond, and the cyclization event into one continuous operation. This telescoped approach not only simplifies the workflow but also dramatically improves the overall yield, with specific embodiments demonstrating conversion efficiencies reaching up to 96 percent. The versatility of this system is evident in its ability to tolerate a wide array of functional groups, allowing for the rapid generation of diverse chemical libraries essential for structure-activity relationship (SAR) studies without the need for protecting group strategies.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its intricate catalytic cycle which orchestrates multiple bond-forming events with high precision. The reaction is initiated by the reduction of the nitro compound to the corresponding amine species mediated by molybdenum hexacarbonyl, which simultaneously serves as the source of carbon monoxide for the subsequent carbonylation step. Following this reduction, a base-promoted intermolecular coupling occurs between the generated amine and the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. The palladium catalyst, activated by the dppp ligand, then undergoes oxidative addition into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive divalent palladium species. As the temperature is maintained at 120 °C, the molybdenum complex releases carbon monoxide which inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This key species then undergoes intramolecular nucleophilic attack by the nitrogen atom, facilitated by the base, to close the seven-membered palladacycle before final reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. Because the reaction proceeds through a concerted cascade in a single vessel, the exposure of reactive intermediates to external environments is minimized, thereby reducing the formation of hydrolysis byproducts or oxidation impurities that often plague multi-step processes. The use of sodium carbonate as a mild base ensures that sensitive functional groups on the aromatic rings remain intact, preventing unwanted side reactions such as dehalogenation or ester hydrolysis. Furthermore, the choice of 1,4-dioxane as the solvent provides an optimal balance of polarity and boiling point, ensuring that all organic components remain in solution throughout the 16 to 30 hour reaction window, which promotes homogeneous catalysis and consistent heat transfer. This controlled environment is critical for achieving the high purity profiles required for pharmaceutical intermediates, as it limits the generation of complex impurity profiles that are difficult to separate during downstream processing.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and thermal parameters to maximize yield and reproducibility. The process begins by charging a reaction vessel with the palladium chloride catalyst, the dppp ligand, sodium carbonate base, and the molybdenum hexacarbonyl CO source in 1,4-dioxane solvent. Once the mixture is homogenized, the trifluoroethylimidoyl chloride and the specific nitro compound substrate are added, typically with a slight excess of the nitro component to drive the reaction to completion. The detailed standardized synthesis steps, including specific workup procedures and purification protocols, are outlined in the guide below.
- Charge a reaction vessel with palladium chloride (5 mol %), dppp ligand (10 mol %), sodium carbonate (2.0 equiv), and molybdenum hexacarbonyl (2.0 equiv) in 1,4-dioxane solvent.
- Add the trifluoroethylimidoyl chloride substrate and the corresponding nitro compound (1.2 equiv) to the mixture under stirring.
- Heat the reaction mixture to 120 °C and maintain for 16 to 30 hours to ensure complete conversion, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical transformation. The primary driver of value is the substitution of expensive, specialty starting materials with commodity-grade nitro compounds, which are produced globally in massive volumes and offer exceptional price stability. By eliminating the dependency on pre-functionalized anilines or acid anhydrides, manufacturers can decouple their supply chains from niche vendors and source raw materials from a broader, more competitive market. Additionally, the one-pot nature of the reaction significantly reduces the consumption of solvents and energy, as there is no need for intermediate isolation, drying, or solvent swaps between steps. This consolidation of unit operations leads to a drastic simplification of the manufacturing workflow, allowing for higher throughput in existing reactor fleets without the need for significant capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of low-cost nitro aromatics and the avoidance of high-pressure equipment. By using solid Mo(CO)6 instead of gaseous CO, facilities can operate in standard glass-lined or stainless steel reactors rather than expensive high-pressure autoclaves, resulting in substantial capital expenditure savings. Furthermore, the high reaction efficiency and yields exceeding 90 percent in optimized examples mean that less raw material is wasted per kilogram of product, directly lowering the cost of goods sold (COGS). The reduction in processing steps also minimizes labor costs and utility consumption, creating a leaner, more cost-effective production profile that enhances margin potential for high-volume API intermediates.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved because the key building blocks, such as nitrobenzenes and substituted nitro compounds, are ubiquitous in the global chemical market with multiple qualified suppliers. This diversity of supply sources mitigates the risk of shortages or price spikes that often affect specialized reagents. The robustness of the reaction conditions, which tolerate a wide range of substituents including halogens and alkyl groups, allows for flexibility in raw material specifications, meaning that commercial grade reagents can often be used without rigorous upgrading. This flexibility ensures continuous production schedules and reduces the likelihood of batch failures due to minor variations in raw material quality, thereby securing a steady flow of intermediates to downstream formulation sites.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its successful demonstration on gram scales with straightforward translation to kilogram and metric-ton levels. The use of a closed system with a solid CO source inherently improves workplace safety and environmental compliance by eliminating fugitive emissions of toxic carbon monoxide gas. Waste generation is minimized through the high atom economy of the cascade reaction and the reduced volume of solvent required for a one-pot process compared to multi-step alternatives. This aligns perfectly with modern green chemistry initiatives and regulatory expectations, simplifying the permitting process for new manufacturing lines and reducing the burden on waste treatment facilities, which is a critical factor for long-term sustainable operations in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: What are the advantages of using nitro compounds over traditional aniline substrates?
A: Nitro compounds are significantly cheaper and more readily available commodity chemicals compared to pre-functionalized anilines or acid anhydrides. This method eliminates the need for expensive starting materials and reduces the overall raw material cost for large-scale production.
Q: How does this method improve safety regarding carbon monoxide usage?
A: Instead of using hazardous high-pressure carbon monoxide gas, this protocol utilizes molybdenum hexacarbonyl (Mo(CO)6) as a solid CO surrogate. This releases CO in situ under heating, drastically reducing safety risks and eliminating the need for specialized high-pressure autoclaves.
Q: What is the substrate scope for this quinazolinone synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as halogens (F, Cl, Br), alkyl groups (methyl), and trifluoromethyl groups on the aromatic ring, as well as diverse amines including aryl, naphthyl, and cycloalkyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, having a partner with deep technical expertise in C-H activation and carbonylation chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs capable of detecting trace metal residues to meet stringent purity specifications. We understand that the transition from bench-scale discovery to commercial supply requires not just chemical knowledge but also project management excellence, ensuring that your timelines are met without compromising on quality or safety standards.
We invite you to engage with our technical team to explore how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule. We encourage potential partners to contact our technical procurement team to obtain specific COA data for related analogues and comprehensive route feasibility assessments. Let us help you accelerate your drug development timeline with a supply chain that is as robust and efficient as the chemistry we employ.
