Advanced Chiral Quinoline-Imidazoline Ligands for Efficient Asymmetric Lactam Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust solutions for the construction of chiral heterocyclic scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN111925356B introduces a significant advancement in this domain by disclosing a novel class of chiral quinoline-imidazoline ligands and their efficient synthetic methodology. These ligands are engineered to overcome the limitations of traditional chiral phosphine or oxazoline systems, offering superior stability and tunable steric environments for transition metal catalysis. The core innovation lies in the unique fusion of a quinoline backbone with a chiral imidazoline moiety, creating a rigid yet adaptable coordination sphere that effectively controls stereochemistry during complex bond-forming events. This technology is particularly relevant for the synthesis of chiral gamma-lactams, a ubiquitous motif in medicinal chemistry, via nickel-catalyzed asymmetric difunctionalization reactions.
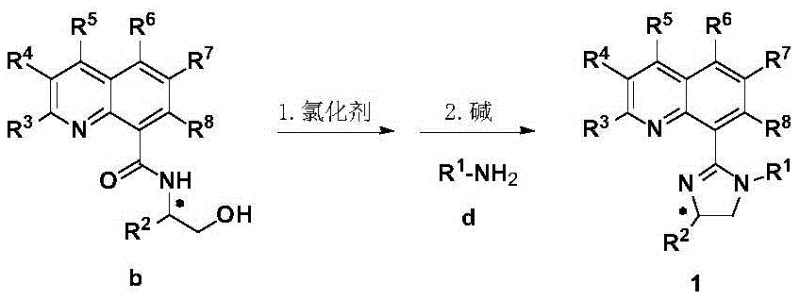
Traditional methods for synthesizing asymmetric alpha-monosubstituted gamma-lactams often suffer from significant drawbacks, including the need for stoichiometric chiral auxiliaries, harsh reaction conditions, or the use of expensive and air-sensitive noble metal catalysts like palladium or rhodium. Furthermore, conventional approaches frequently struggle with controlling regioselectivity and preventing over-alkylation side reactions, leading to complex purification processes and reduced overall yields. The reliance on 1,1-disubstituted olefins in many existing protocols also limits the structural diversity of accessible products, restricting the chemical space available for drug discovery teams. These inefficiencies translate directly into higher production costs and longer lead times for bringing new therapeutic candidates to clinical trials, creating a bottleneck in the supply chain for high-value pharmaceutical intermediates.
The novel approach detailed in the patent addresses these challenges through a concise and economically viable synthetic route for the ligand itself, starting from readily available quinoline-8-carboxylic acid and chiral beta-amino alcohols. This methodology eliminates the need for complex multi-step sequences often associated with privileged ligand synthesis. More importantly, when applied in catalysis, these ligands enable the use of abundant and inexpensive nickel sources to drive the asymmetric difunctionalization of olefin-tethered amine chlorides with various halides. The reaction proceeds under remarkably mild conditions, typically at temperatures ranging from -20°C to 50°C, and demonstrates exceptional tolerance to functional groups. This represents a paradigm shift towards more sustainable and cost-effective manufacturing processes for complex chiral nitrogen-containing heterocycles.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Difunctionalization
The efficacy of the chiral quinoline-imidazoline ligand stems from its ability to form a well-defined chiral pocket around the nickel center, facilitating a controlled radical pathway. The catalytic cycle likely initiates with the reduction of the nickel(II) precatalyst to a low-valent nickel(0) or nickel(I) species by manganese powder, which then undergoes oxidative addition with the alkyl halide substrate. The chiral ligand coordinates to the metal, imposing strict stereochemical constraints that dictate the facial selectivity of the subsequent migratory insertion of the tethered olefin. This step is critical for establishing the new stereocenter with high enantiomeric excess, as evidenced by the patent data showing ee values consistently above 90% for optimized substrates. The rigidity of the quinoline-imidazoline framework prevents unfavorable conformational rotations that could lead to racemization, ensuring high fidelity in chirality transfer throughout the catalytic turnover.
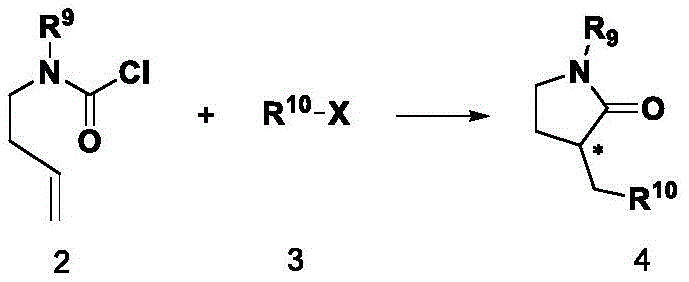
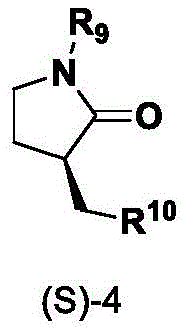
Impurity control is another crucial aspect where this ligand system excels. The specific electronic properties of the quinoline nitrogen and the imidazoline nitrogens modulate the electron density at the metal center, suppressing competing side reactions such as beta-hydride elimination or homocoupling of the halide. The patent highlights that the reaction can be performed in polar aprotic solvents like DMF or NMP, which stabilize the charged intermediates without decomposing the catalyst. Furthermore, the resulting chiral lactam products are formed with high diastereoselectivity, minimizing the formation of unwanted diastereomers that are notoriously difficult to separate. This high level of chemoselectivity and stereoselectivity simplifies downstream processing, reducing the burden on purification teams and improving the overall mass balance of the manufacturing process.
How to Synthesize Chiral Quinoline-Imidazoline Ligands Efficiently
The preparation of these high-performance ligands is designed for scalability and operational simplicity, avoiding the need for cryogenic conditions or inert atmosphere techniques during the ligand synthesis itself. The process involves an initial amidation step followed by a cyclization dehydration, both of which utilize common industrial reagents. The robustness of the synthetic route ensures that the ligand can be produced in multi-kilogram quantities with consistent quality, which is essential for supporting commercial-scale catalytic applications. For detailed operational parameters and specific workup procedures, please refer to the standardized synthesis guide below.
- Condense quinoline-8-carboxylic acid with a chiral beta-amino alcohol using a carbonyl activator like isobutyl chloroformate to form the amide intermediate.
- Activate the amide hydroxyl group using thionyl chloride or phosphorus pentachloride under reflux conditions to generate the reactive imidoyl chloride species.
- React the activated intermediate with a primary amine in the presence of a base such as triethylamine to close the imidazoline ring and yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this technology offers substantial strategic benefits by decoupling production from the volatility of precious metal markets. The substitution of palladium or rhodium with nickel drastically reduces the raw material cost profile of the catalytic system, while the use of manganese as a terminal reductant further enhances economic efficiency. The ligand synthesis itself relies on commodity chemicals like quinoline carboxylic acid and simple amino alcohols, ensuring a stable and diversified supply base that mitigates the risk of single-source dependency. This resilience is critical for maintaining continuous manufacturing operations in the face of global supply chain disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals and the use of ambient or near-ambient reaction temperatures significantly lower the energy consumption and capital expenditure associated with reactor infrastructure. The high catalytic activity allows for lower catalyst loading, which reduces the total amount of ligand and metal required per kilogram of product. Additionally, the simplified purification process resulting from high selectivity minimizes solvent usage and waste disposal costs, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: The stability of the quinoline-imidazoline ligands towards air and moisture simplifies logistics and storage requirements, removing the need for specialized cold-chain shipping or glovebox handling. This robustness ensures that the catalyst system remains active and effective even after extended storage periods, providing flexibility in inventory management. The broad substrate scope demonstrated in the patent means that a single ligand platform can be utilized for the synthesis of diverse intermediates, streamlining the procurement of specialized reagents.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer and more environmentally friendly, utilizing less toxic solvents and generating fewer hazardous byproducts compared to traditional methods. The high atom economy of the difunctionalization reaction aligns with green chemistry principles, facilitating regulatory approval and reducing the environmental footprint of the manufacturing site. The process is readily scalable from gram to ton scale without significant re-optimization, ensuring a smooth transition from process development to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this ligand system in industrial settings. The answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability for process engineers.
Q: What are the stability characteristics of the chiral quinoline-imidazoline ligand?
A: According to patent CN111925356B, the synthesized ligands exhibit excellent stability against water and oxygen, making them easier to store and handle compared to sensitive phosphine ligands.
Q: What is the primary application of this ligand in organic synthesis?
A: The ligand is specifically designed for transition metal-catalyzed asymmetric difunctionalization of olefin-tethered amine chlorides with halides to produce chiral gamma-lactams with high enantioselectivity.
Q: Does this method require expensive noble metal catalysts?
A: No, the protocol utilizes cost-effective nickel catalysts (such as Ni(COD)2 or nickel perchlorate) combined with manganese powder as a reductant, significantly reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quinoline-Imidazoline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands in accelerating drug discovery and process optimization. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand meets the highest standards of quality and consistency required for sensitive asymmetric catalysis applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific synthetic challenges. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you integrate this efficient nickel-catalyzed methodology into your manufacturing portfolio.
