Advanced Chiral Quinoline-Imidazoline Ligands for High-Performance Asymmetric Catalysis and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust solutions for the construction of chiral scaffolds, particularly gamma-lactams which serve as critical cores in numerous bioactive molecules. Patent CN111925356B introduces a significant advancement in this domain through the development of a novel class of chiral quinoline-imidazoline ligands. These ligands are designed to overcome the limitations of traditional organophosphine systems by offering superior stability and tunable steric environments. The core innovation lies in the unique combination of a rigid quinoline backbone with a flexible chiral imidazoline moiety, creating a privileged scaffold for transition metal catalysis. This technology enables the efficient asymmetric difunctionalization of olefin-tethered amine chlorides, a transformation that has historically been challenging due to issues with regioselectivity and over-alkylation. By providing a reliable pathway to high-purity chiral intermediates, this invention addresses a critical bottleneck in the synthesis of complex nitrogen-containing heterocycles.
Historically, the synthesis of chiral gamma-lactams has relied heavily on stoichiometric chiral auxiliaries or expensive noble metal catalysts paired with air-sensitive phosphine ligands. Conventional methods often suffer from poor atom economy, requiring multiple protection and deprotection steps that inflate production costs and extend lead times. Furthermore, many existing catalytic systems struggle to control the stereochemistry when dealing with monosubstituted olefins, frequently leading to racemic mixtures or low yields that are unacceptable for commercial API manufacturing. The reliance on palladium or rhodium in traditional cross-coupling approaches also introduces significant supply chain vulnerabilities and cost volatility. These legacy processes often necessitate rigorous exclusion of moisture and oxygen, demanding specialized equipment and increasing the operational complexity for process chemists aiming for scale-up.
In contrast, the novel approach detailed in the patent utilizes a nickel-catalyzed system supported by the new quinoline-imidazoline ligands, which are remarkably stable to air and moisture. The synthesis of the ligand itself is streamlined, starting from readily available quinoline-8-carboxylic acid and chiral beta-amino alcohols. This route avoids the use of cryogenic conditions or exotic reagents, facilitating a more sustainable and cost-effective production model. The ligand design allows for extensive modulation of electronic and steric properties through the variation of substituents on the imidazoline nitrogen and the quinoline ring. This modularity ensures that the catalyst system can be finely tuned to accommodate a broad range of substrates, from simple alkyl halides to complex functionalized aryl groups, thereby expanding the scope of accessible chemical space for drug discovery teams.
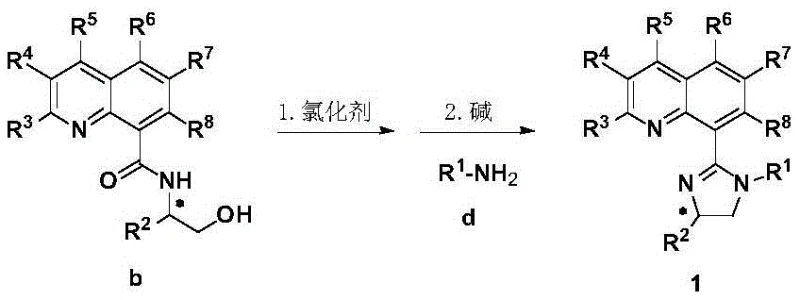
The mechanistic efficacy of this system stems from the precise coordination geometry imposed by the bidentate N,N-ligand on the nickel center. During the catalytic cycle, the ligand stabilizes the low-valent nickel species necessary for the oxidative addition of the alkyl halide, while simultaneously directing the migratory insertion of the tethered olefin. The chiral environment created by the tert-butyl or other bulky groups on the imidazoline ring effectively shields one face of the reacting intermediate, ensuring high facial selectivity during the bond-forming event. This results in the preferential formation of one enantiomer of the cyclic lactam product, often with enantiomeric excess values exceeding 90%. The ability to utilize inexpensive manganese as a reductant further enhances the economic viability of the process, replacing more costly zinc or magnesium alternatives often seen in similar reductive couplings.
Crucially, the reaction tolerates a wide array of functional groups, including esters, nitriles, and ethers, without compromising the integrity of the chiral center. The patent data indicates that the reaction proceeds efficiently at mild temperatures, typically ranging from 0°C to room temperature, which minimizes thermal degradation of sensitive intermediates. The use of polar aprotic solvents like DMF or NMP ensures good solubility of the ionic intermediates while maintaining a homogeneous reaction mixture. This level of control over the reaction parameters is essential for maintaining consistent product quality in a GMP environment. The robustness of the catalyst system reduces the need for extensive purification steps post-reaction, directly contributing to improved overall process mass intensity and reduced waste generation.
How to Synthesize Chiral Quinoline-Imidazoline Ligand Efficiently
The preparation of these high-performance ligands follows a logical and scalable two-step sequence that is well-suited for kilogram-scale production. The process begins with the activation of quinoline-8-carboxylic acid using a carbonyl activator such as isobutyl chloroformate in the presence of a tertiary amine base. This activated species is then coupled with a chiral amino alcohol to form the key amide intermediate. Subsequent cyclization is achieved by treating the amide with a chlorinating agent like thionyl chloride to generate an imidoyl chloride intermediate in situ, which immediately reacts with a primary amine to close the imidazoline ring. Detailed standardized synthesis protocols ensuring optimal yield and purity are provided in the structured guide below.
- Condense quinoline-8-carboxylic acid with a chiral beta-amino alcohol using a carbonyl activator like isobutyl chloroformate and a base such as N-methylmorpholine in dichloromethane.
- Activate the resulting amide intermediate using a chlorinating agent such as thionyl chloride or phosphorus pentachloride under reflux conditions.
- React the activated intermediate with a primary amine in the presence of a base like triethylamine to form the final quinoline-imidazoline ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand technology offers substantial strategic benefits beyond mere technical performance. The shift from precious metal catalysts to nickel-based systems represents a significant opportunity for cost reduction in API manufacturing. Nickel is abundant and significantly cheaper than palladium or rhodium, reducing the raw material cost burden and mitigating risks associated with the volatility of precious metal markets. Furthermore, the stability of the quinoline-imidazoline ligands eliminates the need for specialized inert atmosphere storage and handling during transportation, simplifying logistics and reducing packaging costs. This robustness translates directly into lower total cost of ownership for the catalytic system.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals and the use of commodity starting materials like quinoline carboxylic acid drastically lowers the bill of materials. The high turnover number of the catalyst means that lower loading levels are required to achieve full conversion, further driving down costs. Additionally, the simplified workup procedures reduce solvent consumption and waste disposal fees, contributing to a leaner and more economical production process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Since the ligand precursors are commercially available bulk chemicals, the risk of supply disruption is minimized compared to sourcing proprietary or complex chiral phosphines. The synthetic route is short and high-yielding, allowing for rapid replenishment of catalyst stocks if demand spikes. This reliability ensures continuous production schedules for downstream API synthesis, preventing costly delays in drug development timelines and securing the supply chain against external market fluctuations.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize standard industrial solvents, making the scale-up from gram to ton scale straightforward without the need for specialized reactor modifications. The use of manganese as a reductant generates less hazardous waste compared to other metal reductants, aligning with increasingly stringent environmental regulations. This green chemistry profile facilitates easier regulatory approval and supports corporate sustainability goals by reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this ligand system in industrial settings. Understanding these aspects is crucial for process development teams evaluating the technology for integration into existing pipelines. The answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and relevance.
Q: What are the stability characteristics of the chiral quinoline-imidazoline ligand?
A: According to patent CN111925356B, the synthesized ligands exhibit excellent stability against water and oxygen, making them significantly easier to store and handle compared to sensitive phosphine ligands.
Q: Which transition metals are compatible with this ligand system?
A: The patent demonstrates successful complexation and catalytic activity with various nickel salts including nickel perchlorate, nickel bromide, and bis(1,5-cyclooctadiene)nickel, as well as cobalt and copper compounds.
Q: What level of enantioselectivity can be achieved in lactam synthesis?
A: Experimental data in the patent shows that using these ligands in nickel-catalyzed asymmetric difunctionalization reactions can achieve enantiomeric excess (ee) values up to 95% with high chemical yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quinoline-Imidazoline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands in accelerating drug discovery and process optimization. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications for complex chiral intermediates, guaranteeing that every batch meets the highest industry standards. We are committed to supporting your R&D efforts with reliable supply and technical expertise.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific synthesis requirements, demonstrating how this ligand system can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
