Advanced Chiral Imidazole Sulfide Amines: Scalable Synthesis for Asymmetric Catalysis
The landscape of asymmetric synthesis is undergoing a paradigm shift towards sustainable, metal-free methodologies, driven by stringent regulatory requirements and the need for greener manufacturing processes. Patent CN101041638B introduces a groundbreaking class of chiral amines featuring a unique imidazole sulfide structure, designed to bridge the gap between the high activity of metal complexes and the safety profile of organocatalysts. These novel compounds integrate pyrrolidinyl, thioether, and imidazole functional groups into a single chiral scaffold, creating a synergistic environment that stabilizes transition states through non-covalent interactions. For R&D directors seeking robust catalytic systems, this technology offers a compelling alternative to traditional proline-based catalysts, providing enhanced stereocontrol and broader substrate tolerance. As a reliable chiral amine supplier, understanding the structural nuances of these molecules is critical for optimizing downstream synthetic routes in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, asymmetric catalysis has relied heavily on transition metal complexes or simple natural amino acids like proline, both of which present significant operational challenges in large-scale pharmaceutical production. Metal-based catalysts, while highly active, introduce the persistent risk of heavy metal residues in the final Active Pharmaceutical Ingredient (API), necessitating expensive and complex purification steps to meet strict ppm limits. Furthermore, many metal ligands are air-sensitive or require anhydrous conditions, complicating process engineering and increasing operational costs. On the other hand, simple organocatalysts such as L-proline, while safe and inexpensive, often suffer from limited solubility in non-polar solvents and moderate stereoselectivity in certain transformations. As illustrated in the prior art structures below, early generations of catalysts lacked the multifunctional design required to activate diverse electrophiles and nucleophiles simultaneously with high precision.
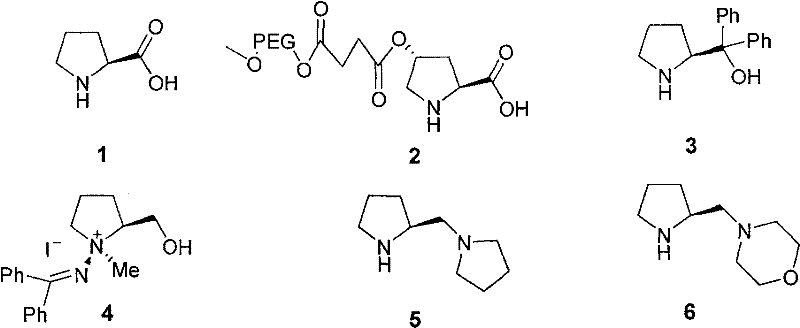
The Novel Approach
The innovation disclosed in CN101041638B overcomes these historical bottlenecks by engineering a hybrid molecular architecture that combines the nucleophilicity of a secondary amine with the hydrogen-bonding capability of an imidazole ring and the polarizability of a sulfur atom. This tri-functional design allows the catalyst to activate substrates through multiple modes simultaneously, such as enamine formation and hydrogen bonding, leading to superior reaction rates and selectivity. Unlike the rigid structures of previous generations, these imidazole sulfide amines offer tunable steric and electronic properties through variation of the R1, R2, and R3 substituents. This modularity enables chemists to fine-tune the catalyst for specific transformations, ensuring high yields and enantiomeric excess across a wide range of asymmetric Michael, Aldol, and Mannich reactions without the need for toxic metals.
Mechanistic Insights into Synergistic Organocatalysis
The exceptional performance of these chiral amines stems from the precise spatial arrangement of their functional groups, which creates a well-defined chiral pocket for substrate binding. The pyrrolidine nitrogen acts as the primary nucleophilic site, forming transient enamine or iminium intermediates with carbonyl substrates, while the adjacent thioether group enhances the nucleophilicity of the nitrogen through electron-donating effects. Crucially, the imidazole ring serves as a hydrogen bond donor/acceptor module, stabilizing the developing charge in the transition state and directing the approach of the electrophile to one specific face of the enamine. This cooperative catalysis mechanism mimics the efficiency of enzymatic active sites, where multiple residues work in concert to lower activation energy barriers. The general structures I and II shown below highlight the core scaffold responsible for this high level of stereoinduction, where the chirality at the alpha-carbon is effectively transmitted to the newly formed bond.
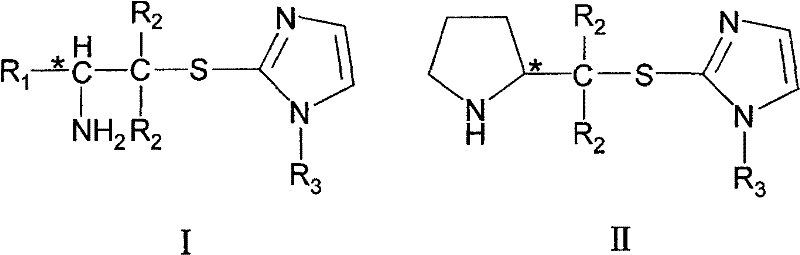
From an impurity control perspective, the robustness of this mechanistic pathway minimizes the formation of side products commonly associated with uncatalyzed background reactions. The strong coordination between the catalyst and the substrate ensures that the reaction proceeds through a single, dominant transition state, thereby suppressing the formation of undesired diastereomers or racemic byproducts. This high degree of selectivity is vital for pharmaceutical applications, where impurity profiles must be tightly controlled to ensure patient safety and regulatory approval. Furthermore, the absence of metal ions eliminates the risk of metal-catalyzed decomposition pathways that can degrade sensitive intermediates during storage or processing. For procurement teams, this translates to a more consistent product quality and reduced batch-to-batch variability, streamlining the quality assurance process.
How to Synthesize Chiral Imidazole Sulfide Amines Efficiently
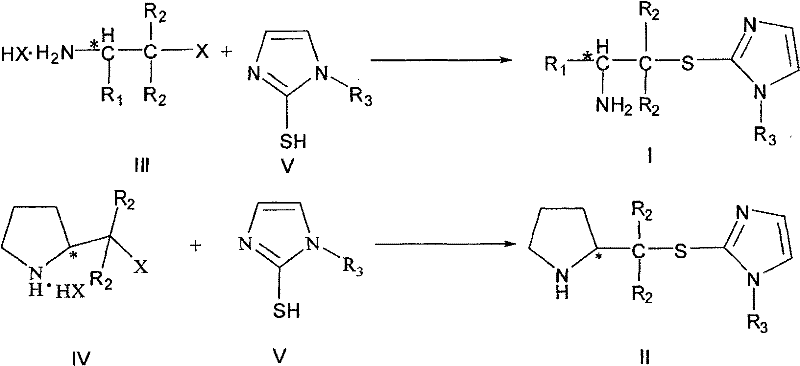
The preparation of these high-value chiral intermediates relies on a straightforward nucleophilic substitution strategy that leverages readily available chiral pool materials. The process involves reacting a halogenated aliphatic amine hydrohalide salt, derived directly from natural amino acids, with an N-substituted mercaptoimidazole in a common organic solvent. This convergent synthesis allows for the rapid assembly of the complex scaffold in a single step, avoiding the need for lengthy protection-deprotection sequences typical of peptide synthesis. The reaction conditions are mild, typically requiring reflux in solvents like ethanol or acetonitrile, making the process energy-efficient and safe for operation. Detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Prepare the reaction mixture by combining chiral amino acid-derived halogenated aliphatic amine hydrohalide salt with N-R3 substituted mercaptoimidazole in a suitable organic solvent such as ethanol or acetonitrile.
- Heat the mixture to reflux conditions for approximately 8 to 12 hours to ensure complete substitution reaction while maintaining the stereochemical integrity of the chiral center.
- Upon completion, neutralize the reaction solution, remove solvents under reduced pressure, wash with ethyl acetate, and purify the crude product via column chromatography to obtain the high-purity target chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology represents a strategic opportunity to optimize costs and mitigate supply risks associated with traditional catalytic methods. The elimination of precious metals such as palladium, rhodium, or iridium from the process removes a significant variable cost component and shields the supply chain from the volatility of the precious metals market. Additionally, the use of amino acids as starting materials ensures a sustainable and renewable feedstock base, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing process. The simplicity of the workup procedure, which involves standard distillation and washing rather than complex scavenging or filtration steps, further drives down operational expenditures and shortens cycle times.
- Cost Reduction in Manufacturing: The primary economic driver for this technology is the complete removal of expensive transition metal catalysts and the associated ligands, which often account for a substantial portion of raw material costs in fine chemical synthesis. By replacing these with inexpensive, bio-based amino acid derivatives and simple imidazole reagents, the overall material cost is drastically simplified. Furthermore, the high yields reported in the patent examples, often exceeding 90%, minimize raw material waste and maximize throughput per batch. The absence of metal removal steps also reduces the consumption of specialized scavengers and filtration media, leading to substantial cost savings in downstream processing.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral catalysts can often be a bottleneck due to the limited number of specialized suppliers and the complexity of their synthesis. However, the precursors for these imidazole sulfide amines are commodity chemicals available from multiple global vendors, ensuring a robust and diversified supply base. The synthetic route itself is short and high-yielding, allowing for rapid production scaling to meet sudden spikes in demand without long lead times. This resilience is critical for maintaining continuous API production schedules and avoiding costly delays caused by catalyst shortages.
- Scalability and Environmental Compliance: The process operates in common, recyclable solvents like ethanol and acetonitrile, avoiding the use of chlorinated solvents or toxic reagents that complicate waste management. The metal-free nature of the catalyst means that wastewater streams do not require expensive heavy metal treatment before discharge, significantly lowering environmental compliance costs. Moreover, the reaction is exothermic but controllable under reflux conditions, posing minimal safety risks during scale-up from laboratory to pilot and commercial plant scales. This ease of scale-up facilitates the commercial production of complex organocatalysts at multi-ton levels.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral amine technology in industrial settings. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing a factual basis for feasibility assessments. Understanding these details helps stakeholders evaluate the fit of this catalyst for their specific synthetic challenges.
Q: What are the primary advantages of these imidazole sulfide amines over traditional metal catalysts?
A: Unlike traditional metal complexes, these organocatalysts eliminate the risk of toxic heavy metal contamination in pharmaceutical products. They operate effectively under mild conditions, often in air or wet solvents, significantly simplifying downstream purification and reducing environmental compliance costs associated with metal waste disposal.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patented process utilizes standard nucleophilic substitution reactions with commercially available starting materials like amino acid derivatives and mercaptoimidazoles. The use of common solvents like ethanol and straightforward workup procedures involving distillation and chromatography makes the process highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Q: What types of asymmetric reactions can these catalysts facilitate?
A: These chiral amines exhibit broad substrate scope and have been demonstrated to effectively catalyze key carbon-carbon bond-forming reactions including asymmetric Michael additions, Aldol condensations, Mannich reactions, and tandem Michael-Aldol sequences, delivering products with high enantiomeric excess (ee) and diastereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free organocatalysis in modern drug discovery and process chemistry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral intermediate meets the highest standards of optical purity and chemical integrity required by the global pharmaceutical industry.
We invite you to collaborate with our technical procurement team to explore how these novel imidazole sulfide amines can enhance your synthetic routes. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific project needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timeline and achieve your commercial goals.
