Revolutionizing Benzimidazole Production: A Green One-Pot Catalytic Oxidation Strategy for Commercial Scale
Revolutionizing Benzimidazole Production: A Green One-Pot Catalytic Oxidation Strategy for Commercial Scale
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and cost-effective pathways for synthesizing privileged heterocyclic scaffolds. Patent CN102442973A introduces a transformative methodology for the synthesis of benzimidazole and benzothiazole compounds via the catalytic oxidation of primary alcohols. This technology represents a significant leap forward from traditional condensation reactions, utilizing a robust TEMPO-based catalytic system coupled with transition metal nitrates. By employing molecular oxygen or air as the terminal oxidant, this process achieves a true "green chemistry" status, producing water as the sole byproduct. For R&D directors and procurement managers alike, this approach offers a compelling alternative to legacy methods, promising enhanced purity profiles and streamlined supply chains for high-value intermediates.
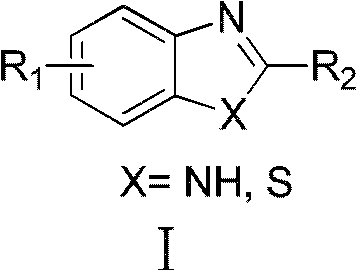
This innovative one-pot protocol effectively bypasses the need for isolating unstable aldehyde intermediates. Instead, it generates the reactive aldehyde species in situ from stable and inexpensive primary alcohol precursors. The reaction conditions are remarkably mild, operating effectively between 0°C and 150°C, and accommodate a wide range of solvents including dichloromethane, acetonitrile, and acetic acid. The versatility of this method allows for the incorporation of diverse substituents on both the alcohol and the diamine/thiophenol components, making it an invaluable tool for the commercial scale-up of complex heterocyclic compounds required in modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of benzimidazoles and benzothiazoles has relied heavily on two primary strategies, both of which suffer from significant drawbacks that impact cost reduction in API manufacturing. The first conventional route involves the condensation of carboxylic acids or their derivatives with o-phenylenediamines. This process typically demands harsh reaction conditions, including strong acidic environments and excessively high temperatures exceeding 180°C. Such extreme parameters not only pose safety risks during commercial scale-up but also lead to the formation of numerous side products, complicating downstream purification and reducing overall yield. The second traditional method utilizes aldehydes as starting materials, reacting them with diamines to form Schiff bases followed by oxidation.
While the aldehyde route operates at lower temperatures, it is plagued by the necessity of using stoichiometric amounts of strong oxidants such as manganese dioxide (MnO2), lead tetraacetate, or DDQ. These reagents are not only expensive but also generate substantial quantities of toxic heavy metal waste, creating severe environmental compliance issues and increasing waste disposal costs. Furthermore, aldehyde starting materials are often chemically unstable, prone to oxidation upon storage, and significantly more costly than their corresponding alcohol counterparts. These factors collectively result in a cumbersome post-treatment process and a less sustainable manufacturing footprint, driving the urgent need for the novel approach detailed in the patent data.
The Novel Approach
The methodology disclosed in CN102442973A fundamentally reengineers the synthesis landscape by shifting the feedstock from unstable aldehydes or expensive acids to robust primary alcohols. This novel approach leverages a catalytic aerobic oxidation system that converts the alcohol to the requisite aldehyde intermediate directly within the reaction vessel. By integrating the oxidation and cyclization steps into a seamless one-pot operation, the process eliminates the need for intermediate isolation, thereby reducing solvent consumption and processing time. The use of air or oxygen as the oxidant is a game-changer, replacing toxic chemical oxidants with a benign, abundant, and cost-free reagent. This shift dramatically simplifies the workup procedure, as the primary byproduct is merely water, aligning perfectly with modern green chemistry principles and regulatory expectations for reduced environmental impact.
Mechanistic Insights into TEMPO-Mediated Aerobic Oxidation
The core of this technological breakthrough lies in the sophisticated interplay between nitroxyl radical catalysts and transition metal co-catalysts. The mechanism initiates with the TEMPO derivative, such as 4-OH-TEMPO or 4-MeO-TEMPO, which acts as the primary hydrogen abstractor. In the presence of a transition metal nitrate (e.g., Iron(III) nitrate, Copper(II) nitrate, or Cerium ammonium nitrate), the TEMPO radical facilitates the selective dehydrogenation of the primary alcohol to form the corresponding aldehyde. Crucially, the transition metal serves to regenerate the active oxoammonium species of the TEMPO catalyst using molecular oxygen, closing the catalytic loop. This synergistic cycle ensures that only catalytic amounts of the expensive organic radical are needed, while the bulk of the oxidation potential is derived from cheap atmospheric oxygen.
Once the aldehyde is generated in situ, it immediately undergoes nucleophilic attack by the o-phenylenediamine or o-aminothiophenol present in the mixture. This forms a Schiff base intermediate, which is subsequently oxidized by the same catalytic system to effect ring closure, yielding the final benzimidazole or benzothiazole scaffold. From an impurity control perspective, this mechanism offers superior selectivity compared to stoichiometric oxidants. The mild nature of the TEMPO/metal nitrate system minimizes over-oxidation of sensitive functional groups, such as sulfides or electron-rich aromatics, which are common in pharmaceutical intermediates. This high chemoselectivity translates directly to higher crude purity, reducing the burden on crystallization and chromatography steps during the production of high-purity OLED material or pharmaceutical intermediates.
How to Synthesize 2-Phenyl-1H-benzimidazole Efficiently
Implementing this synthesis route requires precise control over catalyst loading and oxygen pressure to maximize efficiency. The patent outlines a standardized procedure where the primary alcohol, TEMPO catalyst, and metal nitrate are dissolved in a suitable organic solvent such as acetonitrile or acetic acid. The mixture is then pressurized with air or oxygen, typically between 0.1 to 1.0 MPa, and heated to initiate the oxidation phase. Once the alcohol conversion is confirmed, the diamine component is introduced to trigger the cyclization. Detailed standard operating procedures for scaling this reaction from gram to kilogram levels are critical for ensuring reproducibility and safety. The detailed standardized synthesis steps see the guide below.
- Oxidize the primary alcohol substrate to the corresponding aldehyde in situ using a TEMPO catalyst and transition metal nitrate under air or oxygen pressure.
- Without isolation, introduce the o-phenylenediamine or o-aminothiophenol coupling partner directly into the reaction mixture.
- Continue the oxidative cyclization at elevated temperatures (60-150°C) to form the final benzimidazole or benzothiazole ring structure with high selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic oxidation technology offers profound strategic benefits beyond mere technical elegance. The shift to primary alcohol starting materials addresses a critical vulnerability in the supply chain: the stability and availability of raw materials. Unlike aldehydes, which often require cold storage and have limited shelf lives due to auto-oxidation, primary alcohols are chemically stable, easy to transport, and widely available from bulk chemical suppliers. This stability significantly reduces the risk of raw material degradation during storage and logistics, ensuring a more reliable pharmaceutical intermediate supplier capability. Furthermore, the elimination of stoichiometric heavy metal oxidants removes a major cost center associated with hazardous waste disposal and regulatory compliance.
- Cost Reduction in Manufacturing: The economic argument for this process is robust, driven primarily by the substitution of expensive reagents with catalytic systems and air. Traditional methods relying on DDQ or metal oxides incur high material costs and generate tons of solid waste that must be treated. In contrast, this method uses catalytic amounts of inexpensive metal nitrates and TEMPO derivatives, with oxygen serving as the free oxidant. This drastic reduction in reagent consumption, combined with the simplified aqueous workup (since the byproduct is water), leads to substantial cost savings in raw material procurement and waste management. Additionally, the one-pot nature of the reaction reduces solvent usage and energy consumption by eliminating intermediate isolation steps.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity-grade starting materials. Primary alcohols like benzyl alcohol and its derivatives are produced on a massive global scale for various industries, ensuring a steady and competitive supply. This contrasts with specialized aldehyde intermediates that may have fewer suppliers and longer lead times. By decoupling the synthesis from unstable aldehyde feedstocks, manufacturers can reduce lead time for high-purity intermediates and mitigate the risk of production stoppages due to raw material shortages. The robustness of the catalyst system also means that minor variations in raw material quality are less likely to derail the entire batch, enhancing overall process reliability.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this aerobic oxidation method is inherently scalable. The use of air or low-pressure oxygen is manageable in standard stainless steel reactors, avoiding the need for specialized equipment required for high-pressure hydrogenation or cryogenic conditions. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding heavy metal discharge and volatile organic compound (VOC) emissions. The generation of water as the primary byproduct simplifies effluent treatment, allowing facilities to meet eco-friendly standards more easily. This compliance advantage is crucial for maintaining operational licenses and meeting the sustainability goals of multinational corporate partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: Why is the primary alcohol route superior to traditional aldehyde condensation for benzimidazole synthesis?
A: Traditional aldehyde routes often require stoichiometric amounts of hazardous oxidants like MnO2 or DDQ, generating significant waste. The primary alcohol route described in CN102442973A utilizes stable, cheaper alcohol starting materials and employs catalytic aerobic oxidation, where the only byproduct is water, drastically simplifying purification and reducing environmental impact.
Q: What represents the key catalytic system in this green synthesis method?
A: The core innovation lies in the synergistic use of nitroxyl radicals, specifically 4-OH-TEMPO or its analogues, combined with transition metal nitrates such as iron, copper, or cerium nitrates. This dual-catalyst system enables efficient activation of molecular oxygen (air) to drive the oxidation of alcohols to aldehydes and subsequent cyclization without needing toxic heavy metal oxidants.
Q: Can this methodology be applied to heteroatom-substituted substrates?
A: Yes, the patent data explicitly demonstrates high compatibility with aromatic primary alcohols containing nitrogen, oxygen, and sulfur heteroatoms, as well as various substituted o-phenylenediamines. This broad substrate scope makes it highly suitable for synthesizing diverse pharmaceutical intermediates and agrochemical building blocks with complex functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic oxidation technologies described in CN102442973A for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle aerobic oxidation reactions safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. We understand that consistency and purity are paramount for your downstream applications, whether in oncology drug development or advanced material synthesis.
We invite you to collaborate with us to leverage this green synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating how this methodology can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our benchmark compounds and comprehensive route feasibility assessments. Let us help you build a more sustainable and cost-effective supply chain for your critical pharmaceutical intermediates.
