Advanced Heterogeneous Catalytic Route for 2-Trifluoromethyl Benzimidazole Commercial Production
Advanced Heterogeneous Catalytic Route for 2-Trifluoromethyl Benzimidazole Commercial Production
The pharmaceutical and agrochemical industries continuously demand robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups to enhance metabolic stability and lipophilicity. A significant breakthrough in this domain is detailed in patent CN113429349A, which discloses a novel preparation method for 2-trifluoromethyl substituted benzimidazole compounds utilizing a heterogeneous copper-doped carbon nitride catalyst. This technology represents a paradigm shift from traditional homogeneous catalysis, addressing critical pain points regarding catalyst recovery and product purity that have long plagued the manufacturing of these valuable intermediates. By leveraging a recyclable solid-state catalyst system, this invention not only streamlines the synthetic workflow but also aligns with modern green chemistry principles, offering a sustainable pathway for producing high-value scaffolds used in drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted benzimidazoles has relied on several conventional strategies, each fraught with significant operational and economic drawbacks for large-scale manufacturers. Traditional routes often involve the condensation of o-phenylenediamine with trifluoroacetic acid derivatives or the reductive cyclization of o-nitroanilines, processes that frequently require harsh reaction conditions, strong acids, or expensive reducing agents. Furthermore, earlier copper-catalyzed methods reported in literature typically employed homogeneous copper salts, which, while effective, present severe challenges in downstream processing. The removal of trace copper residues from the final active pharmaceutical ingredient (API) is a costly and tedious endeavor, often requiring specialized scavengers or multiple recrystallization steps to meet stringent regulatory limits. Additionally, homogeneous catalysts cannot be easily recovered or reused, leading to increased waste generation and higher raw material costs per kilogram of product produced.
The Novel Approach
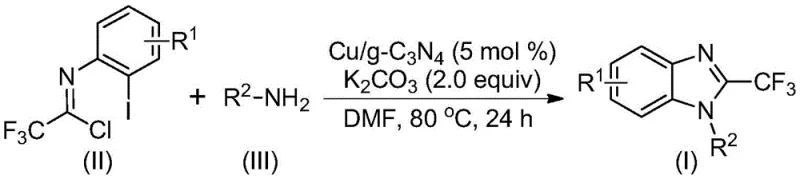
In stark contrast, the methodology outlined in the referenced patent introduces a highly efficient heterogeneous catalytic system that circumvents these legacy issues through the use of copper-doped graphitic carbon nitride (Cu/g-C3N4). This innovative approach facilitates a cascade cyclization reaction between trifluoroacetimidoyl chloride and various amines under remarkably mild conditions, typically ranging from 70°C to 90°C in polar aprotic solvents like DMF. The true brilliance of this system lies in its heterogeneity; the solid catalyst can be simply filtered off upon reaction completion, drastically simplifying the workup procedure and eliminating the need for complex metal scavenging protocols. As illustrated in the reaction scheme below, this single-pot transformation efficiently constructs the benzimidazole core with high atom economy and exceptional yields, often exceeding 90% for a wide range of substrates, thereby establishing a new benchmark for process efficiency in heterocyclic synthesis.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal-mediated C-N bond formation, driven by the unique electronic properties of the copper-doped carbon nitride surface. Initially, the amine nucleophile attacks the electrophilic carbon of the trifluoroacetimidoyl chloride, undergoing a nucleophilic addition-elimination sequence to generate an amidine intermediate in situ. Subsequently, the copper species embedded within the carbon nitride lattice activates the aryl carbon-iodine bond of the intermediate, forming a transient organocopper complex. This activation is crucial as it lowers the energy barrier for the subsequent intramolecular cyclization step. Under the basic conditions provided by potassium carbonate, a molecule of hydrogen iodide is eliminated, prompting the formation of a bivalent or trivalent cyclic copper intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted benzimidazole product and regenerates the active copper catalytic site on the heterogeneous support, ready for the next turnover.
From a quality control perspective, the heterogeneous nature of the catalyst plays a pivotal role in impurity management and product consistency. Unlike soluble copper salts that can coordinate non-specifically with various functional groups leading to side reactions or difficult-to-remove complexes, the immobilized copper sites on the g-C3N4 support offer a more defined steric environment. This confinement effect helps suppress unwanted homocoupling side reactions or over-alkylation, resulting in a cleaner crude reaction profile. Moreover, the physical separation of the catalyst via filtration ensures that the bulk of the copper remains in the solid waste stream rather than contaminating the organic phase. This inherent advantage significantly reduces the burden on purification teams, allowing for simpler chromatographic separations or even direct crystallization in optimized scenarios, ultimately delivering a high-purity intermediate suitable for sensitive downstream coupling reactions in API synthesis.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
The execution of this synthesis protocol is designed for operational simplicity, making it accessible for both laboratory scale optimization and pilot plant campaigns without requiring specialized high-pressure equipment. The process begins with the preparation of the catalyst itself, where copper sulfate pentahydrate is doped onto pre-treated graphitic carbon nitride, creating a stable and active solid reagent. In the main reaction vessel, the trifluoroacetimidoyl chloride substrate is combined with the desired amine partner, a stoichiometric amount of potassium carbonate base, and the Cu/g-C3N4 catalyst in a solvent such as DMF. The mixture is then heated to a moderate temperature, typically around 80°C, and stirred for a period of 18 to 30 hours to ensure complete conversion. Following the reaction, the heterogeneous catalyst is removed by simple filtration, and the filtrate is concentrated and purified, usually via silica gel column chromatography, to isolate the target benzimidazole derivative in high yield.
- Prepare the heterogeneous catalyst by doping copper sulfate onto graphitic carbon nitride support.
- Mix trifluoroacetimidoyl chloride, amine substrate, potassium carbonate, and the Cu/g-C3N4 catalyst in DMF solvent.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heterogeneous catalytic technology translates into tangible strategic benefits that extend far beyond mere chemical yield improvements. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of manufacturing by eliminating the need for expensive ligand systems and reducing the consumption of copper salts, as the catalyst can be potentially recycled multiple times with only partial loss of activity. This durability means that the effective cost per mole of catalyst utilized drops significantly over time, contributing to a leaner bill of materials. Furthermore, the reliance on readily available commodity chemicals such as copper sulfate, carbon nitride precursors, and common amines ensures a robust supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents, thereby securing production continuity for long-term projects.
- Cost Reduction in Manufacturing: The implementation of this process drives down manufacturing expenses primarily through the simplification of the downstream processing workflow. By removing the necessity for rigorous heavy metal clearance steps, which often involve costly scavenger resins or additional aqueous washes, the overall processing time and consumable usage are drastically reduced. The ability to operate at atmospheric pressure and moderate temperatures also lowers energy consumption compared to high-pressure carbonylation or harsh acidic cyclization methods. Additionally, the high reaction efficiency minimizes the formation of by-products, leading to better mass balance and reduced waste disposal costs, which is a critical factor in maintaining profitability in competitive intermediate markets.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including the amine substrates and the trifluoroacetimidoyl chloride building blocks, are structurally simple and widely sourced from global chemical suppliers, mitigating the risk of single-source dependency. The robustness of the catalyst system means that production batches are less likely to fail due to catalyst deactivation or sensitivity to trace moisture and oxygen, common issues with sensitive homogeneous palladium or copper complexes. This reliability allows for more accurate forecasting and inventory planning, ensuring that critical intermediates can be delivered to API manufacturers without unexpected delays caused by complex synthesis failures or extended purification bottlenecks.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is straightforward due to the absence of hazardous reagents and the use of standard stirred-tank reactor configurations. The heterogeneous catalyst can be contained within fixed-bed reactors or easily filtered in large-scale centrifuges, facilitating continuous processing opportunities that further enhance throughput. From an environmental standpoint, the reduced copper leaching and the potential for catalyst reuse align perfectly with increasingly strict environmental regulations regarding heavy metal discharge. This eco-friendly profile not only simplifies permitting for new manufacturing lines but also enhances the sustainability credentials of the final pharmaceutical products, a growing priority for end-user pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heterogeneous catalytic method for benzimidazole synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the operational parameters and scope limitations for potential licensees or manufacturing partners. Understanding these nuances is essential for R&D teams evaluating this route for their specific pipeline candidates and for supply chain professionals assessing the feasibility of technology transfer.
Q: What are the advantages of using Cu/g-C3N4 over homogeneous copper catalysts?
A: The heterogeneous Cu/g-C3N4 catalyst offers superior recyclability, maintaining significant catalytic efficiency even after three cycles, whereas homogeneous catalysts are difficult to separate and often result in heavy metal contamination in the final API.
Q: What is the substrate scope for this benzimidazole synthesis?
A: The method demonstrates broad substrate tolerance, successfully accommodating various amines including aliphatic chains (n-butyl, cyclohexyl) and aromatic groups (benzyl, phenyl), as well as substituted aryl rings with halogens or trifluoromethyl groups.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes cheap, commercially available starting materials and mild reaction conditions (70-90°C), combined with a simple filtration workup, making it highly scalable for industrial production without requiring complex pressure vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and functional materials. Our technical team has thoroughly analyzed the potential of the Cu/g-C3N4 catalytic system described in CN113429349A and is fully prepared to leverage this technology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl benzimidazole meets the exacting standards required for GMP API manufacturing.
We invite you to collaborate with us to optimize this synthetic route for your specific molecular targets, taking advantage of the cost and efficiency benefits offered by this heterogeneous catalysis. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your project volume, demonstrating how this method can reduce your overall COGS. Please contact our technical procurement team today to request specific COA data for related analogues and to discuss route feasibility assessments for your proprietary compounds, ensuring a reliable supply of these valuable building blocks for your future success.
